Abstract
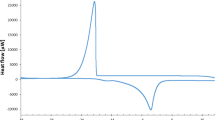
Based on thermograms obtained by differential scanning calorimetry (DSC), melted extruded matrices that contained icing sucrose, maltodextrin and waxy pregelatinized corn starch plasticized with 1,2-propanediol were not totally amorphous. An amorphous phase, which was characterized by a glassy transition of 46.3 °C ± 3.2, coexisted with another crystallized phase, with a fusion temperature of 159.6 °C ± 3.2. Considering the powder X-ray diffractograms of each ingredient, this amorphous phase was only based on sucrose. Increases in the 1,2-propanediol rate did not decrease the fusion enthalpy measured by DSC during the first heating cycle. However, this value increased before stabilizing. Simultaneously, the glassy transition drops showed a greater molecular mobility. Adding water when 1,2-propanediol had a stable low rate during the extrusion process did not improve the amorphization of sucrose, which was mainly crystallized. For an increase in 1,2-propanediol, the fusion enthalpy increased before remaining stable. A comparative assay with a stable water addition in the presence and absence of 1,2-propanediol was characterized by a decrease in the fusion enthalpy. Utilizing water alone as a plasticizer induced a lower proportion of crystalline phase. These results must be linked with the interactions between 1,2-propanediol, a chemical molecule that is highly hygroscopic, and water; propylene glycol captures a part of the water linked with polysaccharides, inducing a more rigid structure. This solvent, which is then hydrated, moves between saccharide aggregates. For the first time, a molecular model was proposed for an extruded carbohydrate matrix containing a crystalline sucrose plasticized by propylene glycol.















Similar content being viewed by others
References
Barra J, Bouquerand P-E, Zampieri D (2012) Extruded particles. EP2440071 (A2). [Online]. Available: https://worldwide.espacenet.com/publicationDetails/biblio?FT=D&date=20120418&DB=&locale=fr_EP&CC=EP&NR=2440071A2&KC=A2&ND=4. Accessed 10 May 2021
Toth AJ, Henson L, Popplewell LM (2007) Non-hygroscopic flavor particles. US2007184163 (A1). [Online]. Available: https://worldwide.espacenet.com/publicationDetails/biblio?FT=D&date=20070809&DB=&locale=fr_EP&CC=US&NR=2007184163A1&KC=A1&ND=4. Accessed 10 May 2021
Lou WC, Popplewell LM (2003) Hydroxypropyl cellulose encapsulation material. EP1304044 (A2). [Online]. Available: https://worldwide.espacenet.com/publicationDetails/biblio?FT=D&date=20030423&DB=&locale=fr_EP&CC=EP&NR=1304044A2&KC=A2&ND=4. Accessed 10 May 2021
Normand V, Cantergiani E, Bouquerand P-E, Barra J, Benczedi D (2005) Non-crystalline perfume or flavour delivery system. EP1515681 (A2). [Online]. Available: https://worldwide.espacenet.com/publicationDetails/biblio?FT=D&date=20050323&DB=&locale=fr_EP&CC=EP&NR=1515681A2&KC=A2&ND=4. Accessed 10 May 2021
Van Lengerich BH, Leung L, Robie SC, Lakkis J, Kang Y, Jarl TM (2004) Encapsulation of sensitive components using pre-emulsification. WO2004009054 (A2). [Online]. Available: https://worldwide.espacenet.com/publicationDetails/biblio?FT=D&date=20040129&DB=&locale=fr_EP&CC=WO&NR=2004009054A2&KC=A2&ND=4. Accessed 10 May 2021
Van Lengerich BH (1998) Embedding and encapsulation of controlled release particles. WO9818610 (A1). [Online]. Available: https://worldwide.espacenet.com/publicationDetails/biblio?FT=D&date=19980507&DB=&locale=fr_EP&CC=WO&NR=9818610A1&KC=A1&ND=4. Accessed 10 May 2021
Aleksovski A, Vervaet C, Dreu R (2016) Hot-melt extrusion and prilling as contemporary and promising techniques in the solvent free production of solid oral dosage forms, based on solid dispersions. Maced Pharm Bull 62(1):3–24. https://doi.org/10.33320/maced.pharm.bull.2016.62.01.001
Castro N, Durrieu V, Raynaud C, Rouilly A, Rigal L, Quellet C (2016) Melt extrusion encapsulation of flavors: a review. Polym Rev 56(1):137–186. https://doi.org/10.1080/15583724.2015.1091776
Reineccius GA, Yan C (2016) Factors controlling the deterioration of spray dried flavourings and unsaturated lipids. Flavour Fragr J 31(1):5–21. https://doi.org/10.1002/ffj.3270
Ubbink J, Krüger J (2006) Physical approaches for the delivery of active ingredients in foods. Trends Food Sci Technol 17(5):244–254. https://doi.org/10.1016/j.tifs.2006.01.007
Kilburn D, Claude J, Schweizer T, Alam A, Ubbink J (2005) Carbohydrate polymers in amorphous states: an integrated thermodynamic and nanostructural investigation. Biomacromolecules 6(2):864–879. https://doi.org/10.1021/bm049355r
Zasypkin D, Paranjpe S, Reick M, Shen C (2018) Natural encapsulation flavor products. WO2018009532 (A1). [Online]. Available: https://worldwide.espacenet.com/publicationDetails/biblio?FT=D&date=20180111&DB=&locale=fr_EP&CC=WO&NR=2018009532A1&KC=A1&ND=4. Accessed 10 May 2021
Subramaniam A, Mciver R, Van Sleeuwen R (2007) Process for the incorporation of a flavor or fragrance ingredient or composition into a carbohydrate matrix. US2007128234 (A1). [Online]. Available: https://worldwide.espacenet.com/publicationDetails/biblio?FT=D&date=20070607&DB=&locale=fr_EP&CC=US&NR=2007128234A1&KC=A1&ND=4. Accessed 10 May 2021
Snyder D, Zhang J (2017) High load flavor particles. WO2017203006 (A1). [Online]. Available: https://worldwide.espacenet.com/publicationDetails/biblio?FT=D&date=20171130&DB=&locale=fr_EP&CC=WO&NR=2017203006A1&KC=A1&ND=4. Accessed 10 May 2021
Poshadri A, Kuna A (2010) Microencapsulation technology: a review. J Res Angrau 38(1):86–102
Porzio M (2008) Melt Extrusion and Melt Injection. Perfum Flavor 33(6):48–53
Benczedi D, Bouquerand P-E (2001) Process for the preparation of granules for the controlled release of volatile compounds. WO0117372 (A1). [Online]. Available: https://worldwide.espacenet.com/publicationDetails/biblio?FT=D&date=20010315&DB=&locale=fr_EP&CC=WO&NR=0117372A1&KC=A1&ND=4. Accessed 10 May 2021
Leusner SJ, Lakkis J, Van Lengerich BH, Jarl TM (2002) Oligosaccharide encapsulated mineral and vitamin ingredients. WO0205667A2. [Online]. Available: https://worldwide.espacenet.com/patent/search/family/024382403/publication/WO0205667A2?q=WO0205667%20A2
Renz K-H, Schleifenbaum B, Uhlemann J, Saleeb F (2003) Aroma particle. WO03092412A2. [Online]. Available: https://worldwide.espacenet.com/patent/search/family/029224905/publication/WO03092412A2?q=WO03092412A2
Hilary LN, Sultana S, Islam Z, Sarker MdKU, Abedin MdJ, Haque MM (2021) Recycling of waste poly(vinyl chloride) fill materials to produce new polymer composites with propylene glycol plasticizer and waste sawdust of Albizia lebbeck wood. Curr Res Green Sustain Chem 4:100221. https://doi.org/10.1016/j.crgsc.2021.100221
Jost V, Langowski H-C (2015) Effect of different plasticisers on the mechanical and barrier properties of extruded cast PHBV films. Eur Polym J 68:302–312. https://doi.org/10.1016/j.eurpolymj.2015.04.012
Orliac O, Rouilly A, Silvestre F, Rigal L (2003) Effects of various plasticizers on the mechanical properties, water resistance and aging of thermo-moulded films made from sunflower proteins. Ind Crops Prod 18(2):91–100. https://doi.org/10.1016/S0926-6690(03)00015-3
Jagadeesh D, Prem Kumar B, Sudhakara P, Venkata Prasad C, Varada Rajulu A, Song JI (2013) Preparation and properties of propylene glycol plasticized wheat protein isolate novel green films. J Polym Environ 21(4):930–936. https://doi.org/10.1007/s10924-013-0572-4
Ekielski A, Żelaziński T, Siwek A, Sharma V, Kumar Mishra P (2020) Formulation and characterization of corn grits- propylene glycol extrudates. Mater Today Proc. 21:1772–1780. https://doi.org/10.1016/j.matpr.2020.01.230
Salah AM (2015) Mechanical grinding effects on physical state of amorphous pharmaceutical materials.’, Materials Science, University of Lille I, France [Online]. Available: http://www.theses.fr/2015LIL10098
Goupy J (2017) Les plans d’expériences, optimisation du choix des essais et de l’interprétation des résultats, 5e ed. Dunod
Russo L, Gražulis S, Bagdziunas G (2013) Structure of Sucrose. http://www.crystallography.net/cod/3500015.html. Accessed 16 Oct 2021
Tackenberg MW, Thommes M, Schuchmann HP, Kleinebudde P (2014) Solid state of processed carbohydrate matrices from maltodextrin and sucrose. J Food Eng 129:30–37. https://doi.org/10.1016/j.jfoodeng.2014.01.003
Dujardin N (2009) Solid state vitrification of glucose by mechanical milling and the control of its mutarotation. Sciences et Technologies, University of Lille I, Lille [Online]. Available: https://pepite-depot.univ-lille.fr/LIBRE/Autres/2009/50376-2009-Dujardin.pdf
Magoń A et al (2015) Reprint of “Heat capacity and transition behavior of sucrose by standard, fast scanning and temperature-modulated calorimetry.” Thermochim Acta 603:149–161. https://doi.org/10.1016/j.tca.2014.12.013
Ronkart SN, Blecker CS, Deroanne C, Paquot M (2015) Phénomène de la transition vitreuse appliquée aux glucides alimentaires amorphes à l’état de poudre. Thermochim Acta 603:149–161
Hurtta M, Pitkänen I, Knuutinen J (2004) Melting behaviour of d-sucrose, d-glucose and d-fructose. Carbohydr Res 339(13):2267–2273. https://doi.org/10.1016/j.carres.2004.06.022
Zasypkin D, Paranjpe S, Reick M, Johnson S (2014) Encapsulation compositions comprising of spices, herbs, fruit, and vegetable powders. US2014272011 (A1). [Online]. Available: https://worldwide.espacenet.com/publicationDetails/biblio?FT=D&date=20140918&DB=&locale=fr_EP&CC=US&NR=2014272011A1&KC=A1&ND=4. Accessed 10 May 2021
Zasypkin D, Paranjpe S, Reick M, Johnson S (2013) Extrusion encapsulation of actives at an increased load, using surface active plant. US2013243851 (A1). [Online]. Available: https://worldwide.espacenet.com/publicationDetails/biblio?FT=D&date=20130919&DB=&locale=fr_EP&CC=US&NR=2013243851A1&KC=A1&ND=4. Accessed 10 May 2021
Castro N, Durrieu V, Raynaud C, Rouilly A (2016) Influence of DE-value on the physicochemical properties of maltodextrin for melt extrusion processes. Carbohydr Polym 144:464–473. https://doi.org/10.1016/j.carbpol.2016.03.004
Mathlouthi M, Rogé B (2003) Water vapour sorption isotherms and the caking of food powders. Food Chem 82(1):61–71. https://doi.org/10.1016/S0308-8146(02)00534-4
Ahlqvist MUA, Taylor LS (2002) Water diffusion in hydrated crystalline and amorphous sugars monitored using H/D exchange. J Pharm Sci 91(3):690–698. https://doi.org/10.1002/jps.10068
Roudaut G, Simatos D, Champion D, Contreras-Lopez E, Le Meste M (2004) Molecular mobility around the glass transition temperature: a mini review. Innov Food Sci Emerg Technol. 5(2):127–134. https://doi.org/10.1016/j.ifset.2003.12.003
Zhao YH, Abraham MH, Zissimos AM (2003) Fast calculation of van der Waals volume as a sum of atomic and bond contributions and its application to drug compounds. J Org Chem 68(19):7368–7373. https://doi.org/10.1021/jo034808o
Matveev Y (2000) The plasticizing effect of water on proteins, polysaccharides and their mixtures. Glassy state of biopolymers, food and seeds. Food Hydrocoll 14(5):425–437. https://doi.org/10.1016/S0268-005X(00)00020-5
BeMiller JN (2019) Polysaccharides: properties. In Carbohydrate chemistry for food Scientists, 3rd Edition. WoodHead Publishing-Elsevier-ACCS International, pp. 103–156. [Online]. Available: https://www.elsevier.com/books/carbohydrate-chemistry-for-food-scientists/bemiller/978-0-12-812069-9
Hatakeyama H, Hatakeyama T (1988) Interaction between water and hydrophilic polymers. Thermochim Acta 308(1–2):3–22. https://doi.org/10.1016/S0040-6031(97)00325-0
Ferreira ESC, Voroshylova IV, Koverga VA, Pereira CM, Cordeiro MNDS (2017) New force field model for propylene glycol: insight to local structure and dynamics. J Phys Chem B 121(48):10906–10921. https://doi.org/10.1021/acs.jpcb.7b08251
Rhys NH, Gillams RJ, Collins LE, Callear SK, Lawrence MJ, McLain SE (2016) On the structure of an aqueous propylene glycol solution. J Chem Phys 145(22):1–12. https://doi.org/10.1063/1.4971208
Fey MW, Weil CM, Segur JB (1951) Solubility of sucrose in aqueous glycerol and propylene glycol. Ind Eng Chem 43(6):1435–1436. https://doi.org/10.1021/ie50498a048
Acknowledgements
This study was financed by Expressions Aromatiques with the agreement of Eric Girone, CEO of this company. We thank Théo Schroth, engineer at Expressions Aromatiques graduate of the “École Nationale Supérieure de Chimie de Lille (ENSCL)”, for the treatment of Differential Scanning Calorimeter raw data.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Vincent Carpentier, Régis Goumont, Nathalie Guillou and Bénédicte Ravel. The first draft of the manuscript was written by Vincent Carpentier, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
This subject is not funded but benefited from CNRS credits from the ILV laboratory. This study is part of a joint research program between the University of Versailles and the company Expressions Aromatiques.
Conflicts of interests/competing interests
Therefore, all authors of this publication confirm that there are no financial interests or conflicts of interest. They also confirm that this research respects perfectly ethics aspects, the absence of the use of animal and human as test subjects and the perfect integrity of chemists while creating the experiences.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Carpentier, V., Goumont, R., Guillou, N. et al. Anti-plasticizing effect of 1,2-propanediol in melt-extruded polysaccharide/sucrose blends. J Polym Res 29, 498 (2022). https://doi.org/10.1007/s10965-022-03352-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-022-03352-4