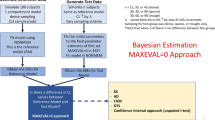
In nonlinear mixed-effects models, estimation methods based on a linearization of the likelihood are widely used although they have several methodological drawbacks. Kuhn and Lavielle (Comput. Statist. Data Anal. 49:1020–1038 (2005)) developed an estimation method which combines the SAEM (Stochastic Approximation EM) algorithm, with a MCMC (Markov Chain Monte Carlo) procedure for maximum likelihood estimation in nonlinear mixed-effects models without linearization. This method is implemented in the Matlab software MONOLIX which is available at http://www.math.u-psud.fr/~lavielle/monolix/logiciels. In this paper we apply MONOLIX to the analysis of the pharmacokinetics of saquinavir, a protease inhibitor, from concentrations measured after single dose administration in 100 HIV patients, some with advance disease. We also illustrate how to use MONOLIX to build the covariate model using the Bayesian Information Criterion. Saquinavir oral clearance (CL/F) was estimated to be 1.26 L/h and to increase with body mass index, the inter-patient variability for CL/F being 120%. Several methodological developments are ongoing to extend SAEM which is a very promising estimation method for population pharmacockinetic/pharmacodynamic analyses.
Similar content being viewed by others
References
Beal S.L., Sheiner L.B. (1980) The NONMEM System. Am. Stat. 34:118–119
Sheiner L.B., Rosenberg B., Marathe V.V. (1977). Estimation of population characteristics of pharmacokinetic parameters from routine clinical data. J. Pharmacokinet. Biopharm. 5:445–479
Davidian M., Giltinan D.M. (2003). Non linear models for repeated measurement data: an overview and update. J. Agric. Biol. Environ. Stat. 8:387–419
Ramos R.Q., Pantula S.G. (1995). Estimation of nonlinear random coefficient models. Statist. Probab. Lett. 24:49–56
Vonesh E.F. (1996) A note on the use of Laplace’s approximation for nonlinear mixed-effects models. Biometrika 83:447–452
Ko H., Davidian M. (2000). Correcting for measurement error in individual-level covariates in nonlinear mixed effect models. Biometrics 56:368–375
Dempster A.P., Laird N.M., Rubin D.B. (1977). Maximum likelihood from incomplete data via the EM algorithm. J. R. Stat. Soc. Ser. B. Stat. Methodol. 1:1–38
Lindstrom M.J., Bates D.M. (1988). Newton-Raphson and EM algorithms for linear mixed-effects models for repeated-measures data. J. Am. Stat. Assoc. 83:1014–1022
Mentré F., Gomeni R. (1995). A two-step algorithm for estimation on non-linear mixed-effects with an evaluation in population pharmacokinetics. J. Biopharm. Stat. 5:141–158
Wei G.C., Tanner M.Z. (1991). Applications of multiple imputation to the analysis of censored regression data. Biometrics 47:1297–1309
Walker G. (1996). An EM algorithm for non-linear random effects models. Biometrics 52:934–944
Wu L. (2002). A joint model for nonlinear mixed-effects models with censoring and covariates measured with error, with application to AIDS studies. J. Am. Statist. Assoc. 97:955–964
Wu L. (2004). Exact and approximate inferences for nonlinear mixed-effects models with missing covariates. J. Am. Statist. Assoc. 99: 700–709
Delyon B., Lavielle M., Moulines E. (1999). Convergence os a stochastic approximation version of the EM procedure. Ann. Stat. 27:94–128
Kuhn E., Lavielle M. (2005). Maximum likelihood estimation in nonlinear mixed effects models. Comput. Statist. Data. Anal. 49:1020–1038
Trout H., Mentré F., Panhard X., Kodjo A., Escaut L., Pernet P., Gobert J.G., Vittecoq D., Knellwolf A.L., Caulin C., Bergmann J.F. (2004). Enhanced saquinavir exposure in HIV1-infected patients with diarrhea and/or wasting syndrome. Antimicrob. Agents Chemother. 48:538–545
P. Girard and F. Mentré. A comparison of estimation methods in nonlinear mixed effects models using a blind analysis. PAGE 14 (2005); Abstr 834 [www.page-meeting. org/?abstract=834].
Louis T.A. (1982). Finding the observed information matrix when using EM algorithm. J. R. Stat. Soc. B. 44:226–233
Kowalski K.G., Hutmacher M.M. (2001). Efficient screening of covariates in population models using Wald’s approximation to the likelihood ratio test. J. Pharmacokinet. Pharmacodyn. 28:253–275
D. O. Stram and J. W. Lee. Variance components testing in the longitudinal mixed effects model. Biometrics 50(4):1171–1177 (1994). Erratum in: Biometrics 51(3):1196 (1995).
Jonsson E.N., Karlsson M.O. (1998) Automated covariate model building within NONMEM. Pharm. Res. 15:1463–1468
Kass R.E., Raftery A.E. (1995). Bayes factors. J. Am. Stat. Assoc. 90:773–795
Verbeke G., Molenberghs G. (2004). Linear Mixed Effect Models for Longitudinal Data. New York, Springer
Burnham K.P. Anderson D.R. (2004). Multimodel Inference: Understanding AIC and BIC in model selection. Sociol. Methods Res. 33:261–304
Ramos J.I. (1999). Linearized methods for ordinary differential equations. Appl. Math. Comput. 104:109–129
S. Donnet and A. Samson. Estimation of parameters in incomplete data models defined by dynamical systems. J. Stat. Plan. Infer. to appear (2007).
S. Retout, E. Comets, A. Samson, and F. Mentré. Designs in nonlinear mixed effects models: application to HIV viral load decrease with evaluation, optimization and determination of the power of the test of a treatment effect. PAGE 14 (2005); Abstr 775 [.page-meeting.org/?abstract=775]
Retout S., Mentré F. (2003). Optimization of individual and population designs using Splus. J. Pharmacokinet. Pharmacodyn. 30:417–443
Pillai G.C., Mentré F F., Steimer J.L. (2005). Non-linear mixed effects modeling – from methodology and software development to driving implementation in drug development science. J. Pharmacokinet. Pharmacodyn. 32:161–183
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lavielle, M., Mentré, F. Estimation of Population Pharmacokinetic Parameters of Saquinavir in HIV Patients with the MONOLIX Software. J Pharmacokinet Pharmacodyn 34, 229–249 (2007). https://doi.org/10.1007/s10928-006-9043-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10928-006-9043-z