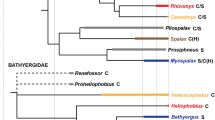
Abstract
All evolutionary modifications of morphology in adult animals presuppose occurrence of changes in developmental programming. While some developmental changes affect rates of trait growth during the entire ontogeny, other developmental changes modify timing and growth rates during limited stages, usually in early development. Identifying which kind of these alterations are more frequent during evolution is crucial for understanding processes influencing the emergence of phenotypic diversity and specializations. Here, we used an allometric approach to assess the relative impact of these two kinds of ontogenetic alterations in the emergence of specialized skull morphology in fossorial spiny rats, comparing them with closely related, more generalist, terrestrial species. Univariate and multivariate analyses of adult shape consistently showed that fossorial spiny rats remarkably differed from terrestrial species, mainly by showing shorter and lower rostrum and more expanded auditory bullae, a set of traits usually considered specializations for life underground. Slopes and elevations of allometric trajectories of cranial traits were estimated for each species and compared with Analysis of Covariance, Likelihood-ratio tests, and Analysis of Variance based on Burnaby-corrected data. These tests showed that changes in allometric elevations were more recurrent during evolution and more congruent with the change in adult morphology than change in allometric slopes. These findings indicated that developmental changes modifying timing and growth rates during limited stages of early development were more frequent than alterations of trait covariation patterns along the entire ontogeny. This kind of developmental change accounts for a large effect on diversification of adult morphology and emergence of burrowing specializations in spiny rats.




Similar content being viewed by others
References
Alberch P, Gould SJ, Oster GF, Wake DB (1997) Size and shape in ontogeny and phylogeny size. Paleobiology 5:296–317
Álvarez A, Perez SI, Verzi DH (2013) Ecological and phylogenetic dimensions of cranial shape diversification in South American caviomorph rodents (Rodentia: Hystricomorpha). Biol J Linn Soc 110:898–913. https://doi.org/10.1111/bij.12164
Álvarez A, Perez SI, Verzi DH (2015) The role of evolutionary integration in the morphological evolution of the skull of caviomorph rodents (Rodentia: Hystricomorpha). Evol Biol 42:312–327. https://doi.org/10.1007/s11692-015-9326-7
Bezerra AMR, Bonvicino CR (2015) Genus Clyomys Thomas, 1916. In: Patton JL, Pardiñas UFJ, D’Elía G (eds) Mammals of South America, Volume 2 (Rodents). The University of Chicago Press, Chicago, pp 935–937
Bezerra AMR, de Oliveira JA, Bonvicino CR (2016) Clyomys laticeps (Rodentia: Echimyidae). Mammal Species 48:83–90. https://doi.org/10.1093/mspecies/sew009
Bonvicino CR, Bezerra AMR (2015) Genus Euryzygomatomys Goeldi, 1901. In: Patton JL, Pardiñas UFJ, D’Elía G (eds) Mammals of South America, Volume 2 (Rodents). The University of Chicago Press, Chicago, pp 937–940
Burnaby TP (1966) Growth-invariant discriminant functions and generalized distances. Biometrics 22:96–110. https://doi.org/10.2307/2528217
Carroll SB (2008) Evo-Devo and an expanding evolutionary synthesis: a genetic theory of morphological evolution. Cell 134:25–36. https://doi.org/10.1016/j.cell.2008.06.030
Carvalho GAS, Salles LO (2004) Relationships among extant and fossil echimyids (Rodentia: Hystricognathi). Zool J Linn Soc 142:445–477. https://doi.org/10.1111/j.1096-3642.2004.00150.x
Cheverud JM (1982) Phenotypic, genetic, and environmental morphological integration in the cranium. Evolution 36:499-516. https://doi.org/10.2307/2408096
Emmons LH, Leite YLR, Patton JL (2015) Family Echimyidae Gray, 1825. In: Patton JL, Pardiñas UFJ, Elía G (eds) Mammals of South America, Volume 2 (Rodents). The University of Chicago Press, Chicago, pp 878–880
Fabre P-H, Galewski T, Tilak M, Douzery EJP (2013) Diversification of South American spiny rats (Echimyidae): a multigene phylogenetic approach. Zool Scr 42:117–134. https://doi.org/10.1111/j.1463-6409.2012.00572.x
Fabre P-H, Upham NS, Emmons LH, Justy F, Leite YLR, Loss AC, Orlando L, Tilak M-K, Patterson BD, Douzery EJP (2016) Mitogenomic phylogeny, diversification, and biogeography of South American spiny rats. Mol Biol Evol 34:msw261. https://doi.org/10.1093/molbev/msw261
Feldhamer GA, Drickamer LC, Vessey SH, Merritt JF, Carey K (2014) Mammalogy: Adaptation, Diversity, Ecology, 4th Editio. Johns Hopkins University Press, Baltimore
Felsenstein J (1988) Phylogenies and quantitative characters. Annu Rev Ecol Syst 19:445–471. https://doi.org/10.1146/annurev.es.19.110188.002305
Felsenstein J (1985) Phylogenies and the comparative method. Am Nat 125:1–15. https://doi.org/10.1086/284325
Giannini NP (2014) Quantitative developmental data in a phylogenetnic framework. J Exp Zool Part B Mol Dev Evol 322:558–566. https://doi.org/10.1002/jez.b.22588
Gomes Rodrigues H, Šumbera R, Hautier L (2016) Life in burrows channelled the morphological evolution of the skull in rodents: the case of African mole-rats (Bathyergidae, Rodentia). J Mammal Evol 23:175–189. https://doi.org/10.1007/s10914-015-9305-x
Goswami A, Polly PD, Mock OB, Sánchez-Villagra MR (2012) Shape, variance and integration during craniogenesis: contrasting marsupial and placental mammals. J Evol Biol 25:862–872. https://doi.org/10.1111/j.1420-9101.2012.02477.x
Goswami A, Randau M, Polly PD, Weisbecker V, Bennett CV, Hautier L, Sánchez-Villagra MR (2016) Do developmental constraints and high integration limit the evolution of the marsupial oral apparatus? Integr Comp Biol icw039. https://doi.org/10.1093/icb/icw039
Gould SJ (1977) Ontogeny and Phylogeny. Harvard University Press, Cambridge
Isaeva VV (2015) Heterochronies, heterotopies, and cell resources of development in ontogenetic and evolutionary transformations. Paleontol J 49:1–8. https://doi.org/10.1134/S0031030115140051
Jolicoeur P (1963) The multivariate generalization of the allometry equation. Biometrics 19:497–499. https://doi.org/10.2307/2527939
Klingenberg CP (1998) Heterochrony and allometry: the analysis of evolutionary change in ontogeny. Biol Rev Camb Phil Soc 73:79–123
Klingenberg CP (1996) Multivariate allometry. In: Marcus LF, Corti M, Loy A, Nalyor GJP, Slice DE (eds) Advances in Morphometrics. Plenum Press, New York, pp 23–49
Klingenberg CP (2008) Morphological integration and developmental modularity. Annu Rev Ecol Evol Syst 39:115–132. https://doi.org/10.1146/annurev.ecolsys.37.091305.110054
Klingenberg CP, Spence JR (1993) Heterochrony and allometry: lessons from the water strider genus Limnoporus. Evolution 47:1834-1853. https://doi.org/10.2307/2410225
Lacey EA, Patton JL, Cameron GN (eds) (2000) Life Underground: the Biology of Subterranean Rodents. The University of Chicago Press, Chicago
Mallarino R, Abzhanov A (2012) Paths less traveled: Evo-Devo approaches to investigating animal morphological evolution. Annu Rev Cell Dev Biol 28:743–763. https://doi.org/10.1146/annurev-cellbio-101011-155732
Marcy AE, Hadly EA, Sherratt E, Garland K, Weisbecker V (2016) Getting a head in hard soils: convergent skull evolution and divergent allometric patterns explain shape variation in a highly diverse genus of pocket gophers (Thomomys). BMC Evol Biol 16:207. https://doi.org/10.1186/s12862-016-0782-1
Marroig G, Shirai LT, Porto A, Oliveira FB, Conto V (2009) The evolution of modularity in the mammalian skull II: evolutionary consequences. Evol Biol 36:136–148. https://doi.org/10.1007/s11692-009-9051-1
Mason MJ (2006) Middle ear structures in fossorial mammals: a comparison with non-fossorial species. J Zool 255:467–486. https://doi.org/10.1017/S0952836901001558
McCoy MW, Bolker BM, Osenberg CW, Miner BG, Vonesh JR (2006) Size correction: comparing morphological traits among populations and environments. Oecologia 148:547–554. https://doi.org/10.1007/s00442-006-0403-6
Mcintosh AF, Cox PG (2016) The impact of digging on craniodental morphology and integration. J Evol Biol 29:2383–2394. https://doi.org/10.1111/jeb.12962
McKinney ML (1988) Classifying heterochrony: allometry, size and time. In: McKinney ML (ed) Heterochrony in Evolution: A Multidisciplinary Approach. Plenum Press, New York, pp 17 – 34
Monteiro LR, Duarte LC, dos Reis SF (2003) Environmental correlates of geographical variation in skull and mandible shape of the punaré rat Thrichomys apereoides (Rodentia: Echimyidae). J Zool 261:47–57. https://doi.org/10.1017/S0952836903003893
Mora M, Olivares AI, Vassallo AI (2003) Size, shape and structural versatility of the skull of the subterranean rodent Ctenomys (Rodentia, Caviomorpha): functional and morphological analysis. Biol J Linn Soc 78:85–96. https://doi.org/10.1046/j.1095-8312.2003.00133.x
Morgan CC, Verzi DH (2011) Carpal-metacarpal specializations for burrowing in South American octodontoid rodents. J Anat 219:167–175. https://doi.org/10.1111/j.1469-7580.2011.01391.x
Mosimann JE (1970) Size allometry: size and shape variables with characterizations of the lognormal and generalized gamma distributions. J Am Stat Assoc 65:930. https://doi.org/10.2307/2284599
Nevo E (1979) Adaptive convergence and divergence of subterranean mammals. Annu Rev Ecol Syst 10:269–308. https://doi.org/10.1146/annurev.es.10.110179.001413
Nevo E (1995) Mammalian evolution underground. The ecological-genetic-phenetic interfaces. Acta Theriol Suppl 3:9–31
Nicola PA, Monteiro LR, Pessôa LM, von Zuben FJ, Rohlf FJ, dos Reis SF (2003) Congruence of hierarchical, localized variation in cranial shape and molecular phylogenetic structure in spiny rats, genus Trinomys (Rodentia: Echimyidae). Biol J Linn Soc 80:385–396. https://doi.org/10.1046/j.1095-8312.2003.00245.x
Patton JL, Leite RN (2015) Genus Proechimys J. A. Allen, 1899. In: Patton JL, Pardiñas UFJ, D’Elía G (eds) Mammals of South America, Volume 2 (Rodents). The University of Chicago Press, Chicago, pp 950–989
Perez SI, Diniz-Filho JAF, Rohlf FJ, dos Reis SF (2009) Ecological and evolutionary factors in the morphological diversification of South American spiny rats. Biol J Linn Soc 98:646–660. https://doi.org/10.1111/j.1095-8312.2009.01307.x
Pessôa LM, Tavares WC, Neves ACA, Silva ALG (2015a) Genus Thrichomys E.- L. Trouessart, 1880. In: Patton JL, Pardiñas UFJ, D’Elía G (eds) Mammals of South America, Volume 2 (Rodents). The University of Chicago Press, Chicago, pp 989–999
Pessôa LM, Tavares WC, Oliveira JA, Patton JL (2015b) Genus Trinomys Thomas, 1921. In: Patton JL, Pardiñas UFJ, D’Elía G (eds) Mammals of South America, Volume 2 (Rodents). The University of Chicago Press, Chicago, pp 999–1019
R Core Team (2016) R: A Language and Environment for Statistical Computing.
Revell LJ (2012) phytools: an R package for phylogenetic comparative biology (and other things). Methods Ecol Evol 3:217–223. https://doi.org/10.1111/j.2041-210X.2011.00169.x
Santos JWA, Lacey EA (2011) Burrow sharing in the desert-adapted torch-tail spiny rat, Trinomys yonenagae. J Mammal 92:3–11. https://doi.org/10.1644/09-MAMM-S-389.1
Schleich CE, Vassallo AI (2003) Bullar volume in subterranean and surface-dwelling caviomorph rodents. J Mammal 84:185–189. https://doi.org/10.1644/1545-1542(2003)084<0185:BVISAS>2.0.CO;2
Sidlauskas B (2008) Continuous and arrested morphological diversification in sister clades of characiform fishes: a phylomorphospace approach. Evolution 62:3135–3156. https://doi.org/10.1111/j.1558-5646.2008.00519.x
Tavares WC, Pessôa LM, Seuánez HN (2016a) Phylogenetic and size constraints on cranial ontogenetic allometry of spiny rats (Echimyidae, Rodentia). J Evol Biol 29:1752–1765. https://doi.org/10.1111/jeb.12905
Tavares WC, Pessôa LM, Seuánez HN (2016b) Stability and acceleration of phenotypic evolution in spiny rats (Trinomys, Echimyidae) across different environments. Zool J Linn Soc 178:149–162. https://doi.org/10.1111/zoj.12406
Upham NS, Patterson BD (2012) Diversification and biogeography of the Neotropical caviomorph lineage Octodontoidea (Rodentia: Hystricognathi). Mol Phylogenet Evol 63:417–429. https://doi.org/10.1016/j.ympev.2012.01.020
Van Valen L (1973) Festschrift. Science 180:488. https://doi.org/10.1111/j.1467-8411.2010.01252.x
Vassallo AI, Mora MS (2007) Interspecific scaling and ontogenetic growth patterns of the skull in living and fossil ctenomyid and octodontid rodents (Caviomorpha: Octodontoidea). In: Kelt DA, Lessa EP, Salazar-Bravo J, Patton JL (eds). The Quintessential Naturalist: Honoring the Life and Legacy of Oliver P. Pearson. University of California Press, Berkeley, pp 944–968
Verzi DH, Álvarez A, Olivares AI, Morgan CC, Vassallo AI (2010) Ontogenetic trajectories of key morphofunctional cranial traits in South American subterranean ctenomyid rodents. J Mammal 91:1508–1516. https://doi.org/10.1644/09-MAMM-A-411.1.Key
Verzi DH, Olivares AI, Morgan CC, Álvarez A (2016) Contrasting phylogenetic and diversity patterns in octodontoid rodents and a new definition of the Family Abrocomidae. J Mammal Evol 23:93–115. https://doi.org/10.1007/s10914-015-9301-1
Voje KL, Hansen TF, Egset CK, Bolstad GH, Pélabon C (2014) Allometric constraints and the evolution of allometry. Evolution 68:866–885. https://doi.org/10.1111/evo.12312
Warton DI, Duursma RA, Falster DS, Taskinen S (2012) Smatr 3- an R package for estimation and inference about allometric lines. Methods Ecol Evol 3:257–259. https://doi.org/10.1111/j.2041-210X.2011.00153.x
Warton DI, Wright IJ, Falster DS, Westoby M (2006) Bivariate line-fitting methods for allometry. Biol Rev Camb Phil Soc 81:259–291. https://doi.org/10.1017/S1464793106007007
Watanabe A, Slice DE (2014) The utility of cranial ontogeny for phylogenetic inference: a case study in crocodylians using geometric morphometrics. J Evol Biol 27:1078–1092. https://doi.org/10.1111/jeb.12382
Webster DB, Plassmann W (1992) Parallel evolution of low-frequency sensitivity in Old World and New World desert rodents. In: Webster DB, Fay RR, Popper AN (eds). The Evolutionary Biology of Hearing. Springer, New York, pp 633–636
Weston EM (2003) Evolution of ontogeny in the hippopotamus skull: using allometry to dissect developmental change. Biol J Linn Soc 80:625–638. https://doi.org/10.1111/j.1095-8312.2003.00263.x
Wilson LAB (2013) Allometric disparity in rodent evolution. Ecol Evol 3:971–984. https://doi.org/10.1002/ece3.521
Wilson LAB, Sánchez-Villagra MR (2010) Diversity trends and their ontogenetic basis: an exploration of allometric disparity in rodents. Proc R Soc B 277:1227–1234. https://doi.org/10.1098/rspb.2009.1958
Zelditch ML, Bookstein FL, Lundrigan BL (1992) Ontogeny of integrated skull growth in the Cotton Rat Sigmodon fulviventer. Evolution 46: 1164-1180. https://doi.org/10.2307/2409763
Acknowledgments
We are grateful to the following curators for access to collections: J.A. de Oliveira (Museu Nacional - Universidade Federal do Rio de Janeiro) and M. de Vivo (Museu de Zoologia da Universidade de São Paulo. WCT initiated this study during his M.Sc. training in Programa de Pós-Graduação em Zoologia (Museu Nacional, Universidade Federal do Rio de Janeiro), with a fellowship from Conselho Nacional de Pesquisa de Desenvolvimento Tecnológico (CNPq). WCT concluded this study in the Department of Genetics (Instituto de Biologia, Universidade Federal do Rio de Janeiro), with a postdoctoral fellowship from Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro – FAPERJ/CAPES grant 209101/E_44/2014.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Online Resource 1
Bonferroni correction of ANOVA for identifying inter-specific variation in log-shape-corrected measurements (PDF 406 kb)
Online Resource 2
Parameters of allometric trajectories of each trait per species (PDF 412 kb)
Online Resource 3
Results of pairwise tests of heterogeneity in allometric slopes and elevations (PDF 533 kb)
Appendix
Appendix
List of specimens used in morphometric analysis. Specimens are deposited in the following institutions: Museu Nacional, Universidade Federal do Rio de Janeiro, Rio de Janeiro, Brazil (MN and CR), and Museu de Zoologia da Universidade de São Paulo, São Paulo, Brazil (MZUSP).
Clyomys Thomas, 1916 (Code: CLY).
Clyomys laticeps (Thomas, 1909).
BRAZIL. DISTRITO FEDERAL: Brasília: MN 22237 and 22,234. Parque Nacional de Brasília, Brasília: MN 22236 and 24,146. GOIÁS: Parque Nacional das Emas: MN 68162, 68,166, and 68,170. MATO GROSSO DO SUL: Aquidauana, Fazenda Rio Negro, Pantanal: MN 68786. Camapuã: MN 24156, 24,159, 24,156, 24,157, and 31,565. Corumbá, Fazenda Alegria: MN 63872, 63,891, 63,850, 63,863, 63,872, 63,897, 63,943 and 63,945. SÃO PAULO: Brotas, Estação Ecológica do Instituto Florestal de Itirapina: MN 68964, 68,971, 68,964, 68,965, 68,968 and 68,971. Campinas: MN 24137. Itapetininga: MZUSP 10743, 10,751, 10,752, 10,753, 10,754, 107,540, 10,798, 25,880, 26,660, and 26,748. Itirapina, Instituto Florestal, Cerrado do Valério: MN 43068, 43,069, 43,070, 43,073 and 68,966.
Euryzygomatomys Goeldi, 1901 (Code: EUR).
Euryzygomatomys spinosus (G. Fischer, 1814).
BRAZIL: ESPÍRITO SANTO: Santa Teresa: MN 4029, 6235 and 31,531. MINAS GERAIS: Caratinga: MN 24147 and 24,149. Passos: MN 31557. PARANÁ: Jacarezinho: MZUSP 26679. RIO DE JANEIRO: Itatiaia, Ponte da Maromba: MZUSP 8078. Levy Gasparian: MN 70164. Silva Jardim: MN 29411 and 29,423. SANTA CATARINA: Corupá: MZUSP 385. SÃO PAULO: Araçoiaba da Serra: MZUSP 10199. Boracéia, Ribeirão da Lagoa: 9803, 10,755, 10,821 and 25,875. Itapetininga: MN 24150 and 24,152. Salesópolis: MN 24153 and 24,154. Salto de Pirapora, Bairro da Ilha: MZUSP 26556, 26,558, 26,559, 26,560, 26,564, 26,566, 26,567, 26,570, 26,570, 26,571, 26,573, 26,575, 26,578, 26,580, 26,582, 26,584, 26,586, 26,625, 26,626, 26,627, 26,628, 26,632, 26,636, 26,637, 26,638, 26,704, 26,829 and 26,830. São Paulo: MZUSP 6684. Ubatuba: MN 31560. Unknown localities: MN 7284, 31,519, 31,525 and 31,528; MZUSP 22407 and 22,408.
Trinomys Thomas, 1921.
Trinomys albispinus (I. Geoffroy St.-Hilaire, 1838) (Code: TRA).
BRAZIL: BAHIA: Jaguaquara: MN 13870, 13,871, 13,872, 13,874, 13,875, 13,878, 13,879, 13,880, 13,881, 13,882, 13,883, 13,884, 13,885, 13,886, 13,887, 13,888, 13,890, 13,892, 13,895, 13,896, 13,897, 13,898, 13,899, 13,900, 13,901, 13,902, 13,932, 13,933, 13,934, 13,936, 13,937, 13,940, 13,944, 13,947, 13,955, 13,956, 13,970, 13,972, 13,973, 13,975, 13,976, 13,981, 13,982, 13,986, 13,990, 13,995, 13,998, 14,000, 33,911, 33,913, 33,914, 33,915, 33,917, 33,919, 33,922, 33,923, 33,931, 33,934, 33,935, 33,936, 34,032, 34,046, and 34,051.
Trinomys yonenagae (Rocha, 1995) (Code: TRY).
BRAZIL: BAHIA: Ibiraba: MZUSP 28883, 28,896, 28,897, 28,898, 28,899, 28,900, 28,901, 28,902, 28,903, 28,904, 28,905, 28,906, 28,907, 28,908, 28,909, 28,910, 28,911, 28,912, 28,913, 28,914, 28,915, 28,916, 28,917, 28,918, 28,919, 28,920, 28,922, 28,923, 28,925, 28,926, 28,927, 28,928, 28,929, 28,930, 28,931, 28,933, 28,934, 28,935, 28,936, 28,938, 28,941, 28,943, 28,944 and 28,945.
Proechimys J. A. Allen, 1899.
Proechimys guyannensis (É. Geoffroy St.-Hilaire, 1803) (Code: PRO).
BRAZIL: AMAPÁ: Serra do Navio: MN 20304, 20,305, 20,307, 20,308, 20,309, 20,312, 20,314, 20,316, 20,320, 20,321, 20,323, 20,325, 20,326, 20,327, 20,328, 20,329, 20,330, 20,331, 20,332, 20,333, 20,335, 20,336, 20,338, 20,340, 20,343, 20,344, 20,345, 20,346, 20,347, 20,348, 20,349, 20,350, 20,351, 20,352, 20,353, 20,354, 20,356, 20,357, 20,358, 20,359, 20,364, 20,381, 20,382, 20,421, 20,424, 20,429, 20,430, 20,435, 20,438, 20,441, 20,443 and 20,447.
Thrichomys E.-L. Trouessart, 1880.
Thrichomys laurentius Thomas, 1904 (Code: THR).
BRAZIL: PERNAMBUCO: Bodocó: MN 5289, 26,816, 26,615, 26,523, 26,313, 26,308, 26,307, 26,306, 26,298, 26,296, 26,295, 26,293, 26,292, 26,265, 26,259, 26,257, 26,256, 26,255, 26,255, 26,254, 26,242, 26,241, 26,240, 26,239, 26,238, 26,237, 26,235, 26,234, 26,232, 26,219, 26,216, 26,214, 26,213, 26,210, 26,209, 26,207, 26,206, 26,197, 26,192, 26,191, 26,190, 26,189, 26,187, 26,185, 26,183, 26,182, 26,178, 26,168, 26,162, 26,160, 26,157, 26,156, 26,153, 26,152, 26,150, 26,148, 26,147, 26,146, 26,145, 26,088, 26,087, 26,086, 26,085, 26,084, 26,083, 26,082, 26,081, 26,037, 26,036, 26,035, CR 992, 988, 2784, 1422, 1404 and 1250.
Rights and permissions
About this article
Cite this article
Tavares, W.C., Pessôa, L.M. & Seuánez, H.N. Changes in Ontogenetic Allometry and their Role in the Emergence of Cranial Morphology in Fossorial Spiny Rats (Echimyidae, Hystricomorpha, Rodentia). J Mammal Evol 26, 575–585 (2019). https://doi.org/10.1007/s10914-018-9433-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10914-018-9433-1