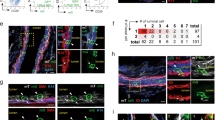
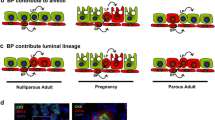
Abstract
Androgens influence mammary gland development but the specific role of the androgen receptor (AR) in mammary function is largely unknown. We identified cell subsets that express AR in vivo and determined the effect of AR activation and transgenic AR inhibition on sub-populations of the normal mouse mammary epithelium by flow cytometry and immunohistochemistry. Immunolocalisation of AR with markers of lineage identity was also performed in human breast tissues. AR activation in vivo significantly decreased the proportion of basal cells, and caused an accumulation of cells that expressed a basal cell marker but exhibited morphological features of luminal identity. Conversely, in AR null mice the proportion of basal mammary epithelial cells was significantly increased. Inhibition of AR increased basal but not luminal progenitor cell activity in vitro. A small population of AR-positive cells in a basal-to-luminal phenotype transition was also evident in human breast lobules. Collectively, these data support a role for AR in promoting a luminal phenotype in mammary epithelial cells.




Similar content being viewed by others
Abbreviations
- AR:
-
Androgen receptor
- DHT:
-
Dihydrotestosterone
- ECM:
-
Extracellular matrix
- ER:
-
Estrogen receptor-alpha
- FACS:
-
Fluorescence-activated cell sorting
- HF:
-
Hydroxyflutamide
- HS-MEC:
-
Hormone-sensing mammary epithelial cell
- MEC:
-
Mammary epithelial cell
- PR:
-
Progesterone receptor
References
Brisken C, Duss S. Stem cells and the stem cell niche in the breast: an integrated hormonal and developmental perspective. Stem Cell Rev. 2007;3(2):147–56.
Rios AC, Fu NY, Lindeman GJ, Visvader JE. In situ identification of bipotent stem cells in the mammary gland. Nature. 2014;506(7488):322–7.
Van Keymeulen A, Rocha AS, Ousset M, Beck B, Bouvencourt G, Rock J, et al. Distinct stem cells contribute to mammary gland development and maintenance. Nature. 2011;479(7372):189–93.
Wuidart A, Ousset M, Rulands S, Simons BD, Van Keymeulen A, Blanpain C. Quantitative lineage tracing strategies to resolve multipotency in tissue-specific stem cells. Genes Dev. 2016;30(11):1261–77.
Colvin CW, Abdullatif H. Anatomy of female puberty: the clinical relevance of developmental changes in the reproductive system. Clin Anat. 2013;26:115–29.
Misiti S, Stigliano A, Borro M, Gentile G, Michienzi S, Cerquetti L, et al. Proteomic profiles in hyperandrogenic syndromes. J Endocrinol Investig. 2010;33(3):156–64.
Hickey TE, Robinson JLL, Carroll JS, Tilley WD. Minireview: the androgen receptor in breast tissues: growth inhibitor, tumor suppressor, oncogene? Mol Endocrinol. 2012;26(8):1252–67.
Tarulli GA, Butler LM, Tilley WD, Hickey TE. Bringing androgens up a NOTCH in breast cancer. Endocr Relat Cancer. 2014;4:T183–202.
Peters AA, Ingman WV, Tilley WD, Butler LM. Differential effects of exogenous androgen and an androgen receptor antagonist in the Peri- and Postpubertal murine mammary gland. Endocrinology. 2011;152(10):3728–37.
Gao YR, Walters KA, Desai R, Zhou H, Handelsman DJ, Simanainen U. Androgen receptor inactivation resulted in acceleration in pubertal mammary gland growth, upregulation of ERα expression, and Wnt/β-catenin signaling in female mice. Endocrinology. 2014;155(12):4951–63.
Barton VN, D’Amato NC, Gordon MA, Christenson JL, Elias A, Richer JK. Androgen receptor biology in triple negative breast Cancer: a case for classification as AR+ or quadruple negative disease. Hormones Cancer. 2015;6:206–13.
Dubois V, Simitsidellis I, Laurent MR, Jardi F, Saunders PT, Vanderschueren D, et al. Enobosarm (GTx-024) modulates adult skeletal muscle mass independently of the androgen receptor in the satellite cell lineage. Endocrinology. 2015;156(12):4522–33.
Barbareschi M, Pecciarini L, Cangi MG, Macrì E, Rizzo A, Viale G, et al. p63, a p53 homologue, is a selective nuclear marker of myoepithelial cells of the human breast. Am J Surg Pathol. 2001;25(8):1054–60.
Yalcin-Ozuysal O, Fiche M, Guitierrez M, Wagner KU, Raffoul W, Brisken C. Antagonistic roles of Notch and p63 in controlling mammary epithelial cell fates. Cell Death Differ. 2010;17(10):1600–12.
Oakes SR, Naylor MJ, Asselin-Labat ML, Blazek KD, Gardiner-Garden M, Hilton HN, et al. The Ets transcription factor Elf5 specifies mammary alveolar cell fate. Genes Dev. 2008;22(5):581–6.
Regan JL, Kendrick H, Magnay F-A, Vafaizadeh V, Groner B, Smalley MJ. C-kit is required for growth and survival of the cells of origin of Brca1-mutation-associated breast cancer. Oncogene. 2012;31(7):869–83.
Kendrick H, Regan JL, Magnay F-A, Grigoriadis A, Mitsopoulos C, Zvelebil M, et al. Transcriptome analysis of mammary epithelial subpopulations identifies novel determinants of lineage commitment and cell fate. BMC Genomics. 2008;9(1):591.
Sflomos G, Dormoy V, Metsalu T, Jeitziner R, Battista L, Scabia V, et al. A preclinical model for ERα-positive breast Cancer points to the epithelial microenvironment as determinant of luminal phenotype and hormone response. Cancer Cell. 2016;29(3):407–22.
Xie Q, Liu Y, Cai T, Horton C, Stefanson J, Wang ZA. Dissecting cell-type-specific roles of androgen receptor in prostate homeostasis and regeneration through lineage tracing. Nat Commun. 2017;8:14284.
Dall GV, Vieusseux JL, Korach KS, Arao Y, Hewitt SC, Hamilton KJ, et al. SCA-1 labels a subset of estrogen-responsive Bipotential repopulating cells within the CD24+ CD49fhi mammary stem cell-enriched compartment. Stem Cell Rep. 2017;8(2):417–31.
Hilton HN, Graham JD, Kantimm S, Santucci N, Cloosterman D, Huschtscha LI, et al. Progesterone and estrogen receptors segregate into different cell subpopulations in the normal human breast. Mol Cell Endocrinol. 2012;361(1–2):191–201.
van Amerongen R, Bowman AN, Nusse R. Developmental stage and time dictate the fate of Wnt/β-catenin-responsive stem cells in the mammary gland. Cell Stem Cell. 2012;11(3):387–400.
Zeng YA, Nusse R. Wnt proteins are self-renewal factors for mammary stem cells and promote their long-term expansion in culture. Cell Stem Cell. 2012;6(6):568–77.
Lindley LE, Curtis KM, Sanchez-Mejias A, Rieger ME, Robbins DJ, Briegel KJ. The WNT-controlled transcriptional regulator LBH is required for mammary stem cell expansion and maintenance of the basal lineage. Development. 2015;142(5):893–904.
Blakemore J, Naftolin F. Aromatase: contributions to physiology and disease in women and men. Physiology. 2016;31(4):258–69.
Bocchinfuso WP, Lindzey JK, Hewitt SC, Clark JA, Myers PH, Cooper R, et al. Induction of mammary gland development in estrogen receptor-alpha knockout mice. Endocrinology. 2000;141(8):2982–94.
Ciarloni L, Mallepell S, Brisken C. Amphiregulin is an essential mediator of estrogen receptor alpha function in mammary gland development. Proc Natl Acad Sci U S A. 2007;104(13):5455–60.
Cunha GR, Young P, Hom YK, Cooke PS, Taylor JA, Lubahn DB. Elucidation of a role for stromal steroid hormone receptors in mammary gland growth and development using tissue recombinants. J Mammary Gland Biol Neoplasia. 1997;2(4):393–402.
Katzenellenbogen BS, Norman MJ. Multihormonal regulation of the progesterone receptor in mcf-7 human breast cancer cells: interrelationships among insulin/insulin- like growth factor-i, serum, and estrogen. Endocrinology. 1990;126(2):891–8.
Lain AR, Creighton CJ, Conneely OM. Research resource: progesterone receptor targetome underlying mammary gland branching morphogenesis. Mol Endocrinol. 2013;27(10):1743–61.
Petz LN, Ziegler YS, Schultz JR, Kim H, Kemper JK, Nardulli AM. Differential regulation of the human progesterone receptor gene through an estrogen response element half site and Sp1 sites. J Steroid Biochem Mol Biol. 2004;88(2):113–22.
Tarulli GA, De Silva D, Ho V, Kunasegaran K, Ghosh K, Tan BC, et al. Hormone- sensing cells require Wip1 for paracrine stimulation in normal and premalignant mammary epithelium. Breast Cancer Res: BCR. 2013;15(1):R10.
Shehata M, Teschendorff A, Sharp G, Novcic N, Russell IA, Avril S, et al. Phenotypic and functional characterisation of the luminal cell hierarchy of the mammary gland. Breast Cancer Res. 2012;14(5):R134.
Mohammed H, Russell IA, Stark R, Rueda OM, Hickey TE, Tarulli GA, et al. Progesterone receptor modulates ERα action in breast cancer. Nature. 2015;523(7560):313–7.
Acknowledgements
The authors wish to acknowledge the support of the University of Adelaide Laboratory Animal Services, University of Adelaide Microscopy for imaging support, Dr. Randall Grose from the South Australian Heath and Medical Research Institute for providing cell sorting services, Ms. Marie Pickering and Ms. Zoya Khityak for assistance with histological experiments, and Ms. Tamara Crittenden from Flinders Medical Centre for recruitment and consent of patients for primary breast tissue collection.
Funding
This work was supported by funding from the National Breast Cancer Foundation (ID PS-15-041 to GAT and WDT), the National Health and Medical Research Council of Australia (ID APP1130077 and ID 1084416 to WDT and TEH), and Cancer Australia (ID 1043497). IMD is supported by a National Breast Cancer Foundation Fellowship ID PF-17-006. The laboratory of WDT & TEH is supported by research funds from GTx Inc., the manufacturers and suppliers of GTx-024 used in this study.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interests
The authors declare no competing or financial interests.
Independent Author Contributions
GAT: Devised concepts & hypotheses, performed experiments, interpreted data, devised conclusions, prepared manuscript. GLL: Performed experiments and analyzed data. MS: Performed single-cell RNA experiments and analyzed data. IMD: Assisted in performing mammary digestion and cell labelling. MMR: Assisted in performing mammary digestion and cell labelling. KAW & DJH: Design and development of AR null mice and supply of their mammary tissue. WDT: Reviewed data, manuscript preparation and editing. TEH: Experimental conceptual design, data revision, study supervision, manuscript editing.
Electronic Supplementary Material
Supplemental Fig. S1
- Treatment with GTx-024 does not alter the proportion of alveolar compared to hormone-sensing MECs when assessed by flow cytometry. (PNG 64 kb)
Supplemental Fig. S2
- A) AR-positive primary human breast cancer tissue labeled with secondary antibodies alone. B) AR+ prostate cancer sample labeled with anti-AR. Scale bar = 50 μm. (PNG 197 kb)
Supplemental Fig. S3
- A-B) Confocal microscopy images of normal breast tissue immunolabeled for p63 (green) and AR (red), and nuclei labeled with DAPI (blue). Dashed box in “i” denotes magnified image in “ii”. Individual channels from magnified image in “ii”, found in panel “iii” (DAPI), “iv” (p63) and “v” (AR). Circles denote cells expressing p63 and AR. Scale bar = 25 μm. (PNG 208 kb)
Supplemental Fig. S4
– A-C) Confocal microscopy images of normal breast tissue immunostained for AR (blue), ER (green) and PR (red). “i” = All channels overlaid, “ii-iv” = same confocal image in “i”, with pairs of channels segregated (AR & PR - “ii”, AR & ER - “iii”, PR & ER - “iv”). Cells that are positive for all three nuclear receptors are observed (arrowheads), as are cells that are only AR-positive (arrows). Scale bar = 25 μm. (PNG 218 kb)
Supplemental Fig. S5
- Gating hierarchy for flow cytometry and cell sorting experiments. Debris and cell clumps were excluded by FSC vs. SSC (A), followed by exclusion of doublets using FSC-H vs. FSC-W followed by SSC-H vs. SSC-W (B). Dead/dying cells were excluded by gating on DAPI-negative cells (C), and lymphocytes excluded by gating on CD45-negative cells (D). The proportion of epithelial cells was determined by gating on CD24/CD49f-positive cells (E). CD24 and CD49f allows for the distinction between basal and luminal epithelial cells (F). Luminal subsets were segregated based on CD49b (G). (PNG 206 kb)
Table S1.
(PNG 131 kb)
Rights and permissions
About this article
Cite this article
Tarulli, G.A., Laven-Law, G., Shehata, M. et al. Androgen Receptor Signalling Promotes a Luminal Phenotype in Mammary Epithelial Cells. J Mammary Gland Biol Neoplasia 24, 99–108 (2019). https://doi.org/10.1007/s10911-018-9406-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10911-018-9406-2