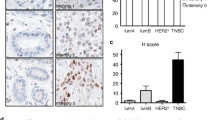
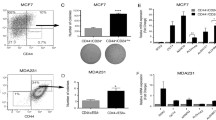
Abstract
Pax-5, an essential transcription factor in B cell development, is aberrantly expressed in various B cell cancer lesions and solid tumors such as breast carcinoma. We have recently shown that Pax-5 regulates NF-κB activity which lead to the modulation of breast cancer phenotypic features (EMT-MET). NF-κB is known as a central mediator in inflammation, stress response as well as being a gatekeeper of pro-tumorigenic activity. However, little is known as to how Pax-5 affects this modulation. We thus turned our attention to microRNAs as potential regulatory effectors. In this study, we set out to elucidate the regulatory network between differential Pax-5 expression and NF-κB activity which dictate breast cancer malignancy. Through next-generation sequencing (NGS) of breast cancer cells conditionally expressing Pax-5, we profile significantly upregulated microRNAs; including microRNA-155, a known regulator of pathological processes and suppressor of malignant growth. Through the conditional expression of microRNA-155 in breast cancer models, we identify and validate IKKε (IKBKE) as a downstream target and an essential effector of Pax-5-mediated suppression of NF-κB signaling. Using rescue experiments, we also confirm that Pax-5 modulates NF-κB activity via IKKε downregulation. Interestingly, we also show that microRNA-155, in turn, supresses Pax-5 expression, indicative of an auto-regulatory feedback loop. Altogether, we demonstrate that Pax-5 inhibits NF-κB signalling through the regulation of microRNA-155 and its downstream target IKKε. The elucidation of this signaling network is relevant as Pax-5 and NF-κB are potent transcriptional regulators of breast cancer aggressivity. In addition, IKKε is relevant oncogene aberrantly expressed in 30% of breast carcinomas. Further insight into the regulatory pathways of breast cancer progression will eventually identify strategic therapeutic and prognostic targets to improve cancer patient outcome.






Similar content being viewed by others
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65(2):87–108. https://doi.org/10.3322/caac.21262.
Miller KD, Siegel RL, Lin CC, Mariotto AB, Kramer JL, Rowland JH, et al. Cancer treatment and survivorship statistics, 2016. CA Cancer J Clin. 2016;66(4):271–89. https://doi.org/10.3322/caac.21349.
Strachan T, Read AP. PAX genes. Curr Opin Genet Dev. 1994;4(3):427–38.
Tremblay P, Gruss P. Pax: genes for mice and men. Pharmacol Ther. 1994;61(1–2):205–26.
Busslinger M. Transcriptional control of early B cell development. Annu Rev Immunol. 2004;22:55–79. https://doi.org/10.1146/annurev.immunol.22.012703.104807.
Rolink AG, Schaniel C, Busslinger M, Nutt SL, Melchers F. Fidelity and infidelity in commitment to B-lymphocyte lineage development. Immunol Rev. 2000;175:104–11.
Adams B, Dorfler P, Aguzzi A, Kozmik Z, Urbanek P, Maurer-Fogy I, et al. Pax-5 encodes the transcription factor BSAP and is expressed in B lymphocytes, the developing CNS, and adult testis. Genes Dev. 1992;6(9):1589–607.
Hamada T, Yonetani N, Ueda C, Maesako Y, Akasaka H, Akasaka T, et al. Expression of the PAX5/BSAP transcription factor in haematological tumour cells and further molecular characterization of the t(9;14)(p13;q32) translocation in B-cell non-Hodgkin’s lymphoma. Br J Haematol. 1998;102(3):691–700.
O'Brien P, Morin P, Ouellette R, Robichaud G. The Pax-5 gene: a pluripotent regulator of B-cell differentiation and cancer disease. Cancer Res. 2011;71(24):7345–50. https://doi.org/10.1158/0008-5472.CAN-11-1874.
Vidal L, Perry J, Vouyovitch C, Pandey V, Brunet-Dunand S, Mertani H, et al. PAX5alpha enhances the epithelial behavior of human mammary carcinoma cells. Molecular Cancer Research : MCR. 2010;8(3):444–56. https://doi.org/10.1158/1541-7786.MCR-09-0368.
Vouyovitch C, Vidal L, Borges S, Raccurt M, Arnould C, Chiesa J, et al. Proteomic analysis of autocrine/paracrine effects of human growth hormone in human mammary carcinoma cells. Adv Exp Med Biol. 2008;617:493–500. https://doi.org/10.1007/978-0-387-69080-3_49.
Benzina S, Beauregard AP, Guerrette R, Jean S, Faye MD, Laflamme M, et al. Pax-5 is a potent regulator of E-cadherin and breast cancer malignant processes. Oncotarget. 2017;8:12052–66. https://doi.org/10.18632/oncotarget.14511.
Crapoulet N, O'Brien P, Ouellette RJ, Robichaud GA. Coordinated expression of Pax-5 and FAK1 in metastasis. Anti Cancer Agents Med Chem. 2011;11(7):643–9.
Schebesta A, McManus S, Salvagiotto G, Delogu A, Busslinger GA, Busslinger M. Transcription factor Pax5 activates the chromatin of key genes involved in B cell signaling, adhesion, migration, and immune function. Immunity. 2007;27(1):49–63. https://doi.org/10.1016/j.immuni.2007.05.019.
Hoesel B, Schmid JA. The complexity of NF-kappaB signaling in inflammation and cancer. Mol Cancer. 2013;12:86. https://doi.org/10.1186/1476-4598-12-86.
Huber MA, Azoitei N, Baumann B, Grunert S, Sommer A, Pehamberger H, et al. NF-kappaB is essential for epithelial-mesenchymal transition and metastasis in a model of breast cancer progression. J Clin Invest. 2004;114(4):569–81. https://doi.org/10.1172/JCI21358.
Guttridge DC, Albanese C, Reuther JY, Pestell RG, Baldwin AS Jr. NF-kappaB controls cell growth and differentiation through transcriptional regulation of cyclin D1. Mol Cell Biol. 1999;19(8):5785–99.
Bours V, Bentires-Alj M, Hellin AC, Viatour P, Robe P, Delhalle S, et al. Nuclear factor-kappa B, cancer, and apoptosis. Biochem Pharmacol. 2000;60(8):1085–9.
Hayden MS, Ghosh S. Shared principles in NF-kappaB signaling. Cell. 2008;132(3):344–62. https://doi.org/10.1016/j.cell.2008.01.020.
Boehm JS, Zhao JJ, Yao J, Kim SY, Firestein R, Dunn IF, et al. Integrative genomic approaches identify IKBKE as a breast cancer oncogene. Cell. 2007;129(6):1065–79. https://doi.org/10.1016/j.cell.2007.03.052.
Shen RR, Zhou AY, Kim E, Lim E, Habelhah H, Hahn WC. IkappaB kinase epsilon phosphorylates TRAF2 to promote mammary epithelial cell transformation. Mol Cell Biol. 2012;32(23):4756–68. https://doi.org/10.1128/MCB.00468-12.
Hutti JE, Shen RR, Abbott DW, Zhou AY, Sprott KM, Asara JM, et al. Phosphorylation of the tumor suppressor CYLD by the breast cancer oncogene IKKepsilon promotes cell transformation. Mol Cell. 2009;34(4):461–72. https://doi.org/10.1016/j.molcel.2009.04.031.
Benzina S, Harquail J, Guerrette R, O'Brien P, Jean S, Crapoulet N, et al. Breast cancer malignant processes are regulated by Pax-5 through the disruption of FAK signaling pathways. J Cancer. 2016;7(14):2035–44. https://doi.org/10.7150/jca.15200.
Ma J, Dong C, Ji C. MicroRNA and drug resistance. Cancer Gene Ther. 2010;17(8):523–31. https://doi.org/10.1038/cgt.2010.18.
Price C, Chen J. MicroRNAs in cancer biology and therapy: current status and perspectives. Genes Dis. 2014;1(1):53–63. https://doi.org/10.1016/j.gendis.2014.06.004.
Ha TY. MicroRNAs in human diseases: from Cancer to cardiovascular disease. Immune Netw. 2011;11(3):135–54. https://doi.org/10.4110/in.2011.11.3.135.
Harquail J, Benzina S, Robichaud GA. MicroRNAs and breast cancer malignancy: an overview of miRNA-regulated cancer processes leading to metastasis. Cancer Biomark. 2012;11(6):269–80. https://doi.org/10.3233/CBM-120291.
Garzon R, Calin GA, Croce CM. MicroRNAs in Cancer. Annu Rev Med. 2009;60:167–79. https://doi.org/10.1146/annurev.med.59.053006.104707.
Arima Y, Inoue Y, Shibata T, Hayashi H, Nagano O, Saya H, et al. Rb depletion results in deregulation of E-cadherin and induction of cellular phenotypic changes that are characteristic of the epithelial-to-mesenchymal transition. Cancer Res. 2008;68(13):5104–12. https://doi.org/10.1158/0008-5472.CAN-07-5680.
Lo HW, Hsu SC, Xia W, Cao X, Shih JY, Wei Y, et al. Epidermal growth factor receptor cooperates with signal transducer and activator of transcription 3 to induce epithelial-mesenchymal transition in cancer cells via up-regulation of TWIST gene expression. Cancer Res. 2007;67(19):9066–76. https://doi.org/10.1158/0008-5472.CAN-07-0575.
Robichaud GA, Nardini M, Laflamme M, Cuperlovic-Culf M, Ouellette RJ. Human Pax-5 C-terminal isoforms possess distinct transactivation properties and are differentially modulated in normal and malignant B cells. J Biol Chem. 2004;279(48):49956–63. https://doi.org/10.1074/jbc.M407171200.
Picot N, Guerrette R, Beauregard AP, Jean S, Michaud P, Harquail J, et al. Mammaglobin 1 promotes breast cancer malignancy and confers sensitivity to anticancer drugs. Mol Carcinog. 2016;55(7):1150–62. https://doi.org/10.1002/mc.22358.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method. Methods. 2001;25(4):402–8. https://doi.org/10.1006/meth.2001.1262.
Hua K, Jin J, Zhao J, Song J, Song H, Li D, et al. miR-135b, upregulated in breast cancer, promotes cell growth and disrupts the cell cycle by regulating LATS2. Int J Oncol. 2016;48(5):1997–2006. https://doi.org/10.3892/ijo.2016.3405.
Comité consultatif de la Société canadienne du cancer : Statistiques canadiennes sur le cancer 2016. Toronto (Ontario) : Société canadienne du cancer, 2016.
Jiang S, Zhang HW, Lu MH, He XH, Li Y, Gu H, et al. MicroRNA-155 functions as an OncomiR in breast cancer by targeting the suppressor of cytokine signaling 1 gene. Cancer Res. 2010;70(8):3119–27. https://doi.org/10.1158/0008-5472.CAN-09-4250.
Gironella M, Seux M, Xie MJ, Cano C, Tomasini R, Gommeaux J, et al. Tumor protein 53-induced nuclear protein 1 expression is repressed by miR-155, and its restoration inhibits pancreatic tumor development. Proc Natl Acad Sci U S A. 2007;104(41):16170–5. https://doi.org/10.1073/pnas.0703942104.
Faraoni I, Antonetti FR, Cardone J, Bonmassar E. miR-155 gene: a typical multifunctional microRNA. Biochim Biophys Acta. 2009;1792(6):497–505. https://doi.org/10.1016/j.bbadis.2009.02.013.
Song C, Liu LZ, Pei XQ, Liu X, Yang L, Ye F, et al. miR-200c inhibits breast cancer proliferation by targeting KRAS. Oncotarget. 2015;6(33):34968–78. https://doi.org/10.18632/oncotarget.5198.
Chang BP, Wang DS, Xing JW, Yang SH, Chu Q, Yu SY. miR-200c inhibits metastasis of breast cancer cells by targeting HMGB1. J Huazhong Univ Sci Technolog Med Sci. 2014;34(2):201–6. https://doi.org/10.1007/s11596-014-1259-3.
Korpal M, Lee ES, Hu G, Kang Y. The miR-200 family inhibits epithelial-mesenchymal transition and cancer cell migration by direct targeting of E-cadherin transcriptional repressors ZEB1 and ZEB2. J Biol Chem. 2008;283(22):14910–4. https://doi.org/10.1074/jbc.C800074200.
Hong H, Yu H, Yuan J, Guo C, Cao H, Li W, et al. MicroRNA-200b impacts breast cancer cell migration and invasion by regulating Ezrin-Radixin-Moesin. Med Sci Monit. 2016;22:1946–52.
Kong X, Ding X, Li X, Gao S, Yang Q. 53BP1 suppresses epithelial-mesenchymal transition by downregulating ZEB1 through microRNA-200b/429 in breast cancer. Cancer Sci. 2015;106(8):982–9. https://doi.org/10.1111/cas.12699.
Tili E, Michaille JJ, Cimino A, Costinean S, Dumitru CD, Adair B, et al. Modulation of miR-155 and miR-125b levels following lipopolysaccharide/TNF-alpha stimulation and their possible roles in regulating the response to endotoxin shock. J Immunol. 2007;179(8):5082–9.
Lossner C, Meier J, Warnken U, Rogers MA, Lichter P, Pscherer A, et al. Quantitative proteomics identify novel miR-155 target proteins. PLoS One. 2011;6(7):e22146. https://doi.org/10.1371/journal.pone.0022146.
Lu F, Weidmer A, Liu CG, Volinia S, Croce CM, Lieberman PM. Epstein-Barr virus-induced miR-155 attenuates NF-kappaB signaling and stabilizes latent virus persistence. J Virol. 2008;82(21):10436–43. https://doi.org/10.1128/JVI.00752-08.
Marsolier J, Pineau S, Medjkane S, Perichon M, Yin Q, Flemington E, et al. OncomiR addiction is generated by a miR-155 feedback loop in Theileria-transformed leukocytes. PLoS Pathog. 2013;9(4):e1003222. https://doi.org/10.1371/journal.ppat.1003222.
Hu JY, Zhang J, Ma JZ, Liang XY, Chen GY, Lu R, et al. MicroRNA-155-IFN-gamma feedback loop in CD4(+)T cells of erosive type oral lichen planus. Sci Rep. 2015;5:16935. https://doi.org/10.1038/srep16935.
Chen Y, Siegel F, Kipschull S, Haas B, Frohlich H, Meister G, et al. miR-155 regulates differentiation of brown and beige adipocytes via a bistable circuit. Nat Commun. 2013;4:1769. https://doi.org/10.1038/ncomms2742.
Van Roosbroeck K, Fanini F, Setoyama T, Ivan C, Rodriguez-Aguayo C, Fuentes-Mattei E, et al. Combining anti-Mir-155 with chemotherapy for the treatment of lung cancers. Clin Cancer Res. 2017;23(11):2891–904. https://doi.org/10.1158/1078-0432.CCR-16-1025.
O'Donnell KA, Wentzel EA, Zeller KI, Dang CV, Mendell JT. C-Myc-regulated microRNAs modulate E2F1 expression. Nature. 2005;435(7043):839–43. https://doi.org/10.1038/nature03677.
Chang TC, Wentzel EA, Kent OA, Ramachandran K, Mullendore M, Lee KH, et al. Transactivation of miR-34a by p53 broadly influences gene expression and promotes apoptosis. Mol Cell. 2007;26(5):745–52. https://doi.org/10.1016/j.molcel.2007.05.010.
Feng R, Dong L. Inhibitory effect of miR-184 on the potential of proliferation and invasion in human glioma and breast cancer cells in vitro. Int J Clin Exp Pathol. 2015;8(8):9376–82.
Wu H, Wang G, Wang Z, An S, Ye P, Luo S. A negative feedback loop between miR-200b and the nuclear factor-kappaB pathway via IKBKB/IKK-beta in breast cancer cells. FEBS J. 2016;283(12):2259–71. https://doi.org/10.1111/febs.13543.
Park SM, Gaur AB, Lengyel E, Peter ME. The miR-200 family determines the epithelial phenotype of cancer cells by targeting the E-cadherin repressors ZEB1 and ZEB2. Genes Dev. 2008;22(7):894–907. https://doi.org/10.1101/gad.1640608.
Li ZW, Chu W, Hu Y, Delhase M, Deerinck T, Ellisman M, et al. The IKKbeta subunit of IkappaB kinase (IKK) is essential for nuclear factor kappaB activation and prevention of apoptosis. J Exp Med. 1999;189(11):1839–45.
Gibcus JH, Tan LP, Harms G, Schakel RN, de Jong D, Blokzijl T, et al. Hodgkin lymphoma cell lines are characterized by a specific miRNA expression profile. Neoplasia. 2009;11(2):167–76.
Zhu M, Wang M, Yang F, Tian Y, Cai J, Yang H, et al. miR-155-5p inhibition promotes the transition of bone marrow mesenchymal stem cells to gastric cancer tissue derived MSC-like cells via NF-kappaB p65 activation. Oncotarget. 2016;7(13):16567–80. https://doi.org/10.18632/oncotarget.7767.
Long L, Yu P, Liu Y, Wang S, Li R, Shi J, et al. Upregulated microRNA-155 expression in peripheral blood mononuclear cells and fibroblast-like synoviocytes in rheumatoid arthritis. Clin Dev Immunol. 2013;2013:296139. https://doi.org/10.1155/2013/296139.
Gerloff D, Grundler R, Wurm AA, Brauer-Hartmann D, Katzerke C, Hartmann JU, et al. NF-kappaB/STAT5/miR-155 network targets PU.1 in FLT3-ITD-driven acute myeloid leukemia. Leukemia. 2015;29(3):535–47. https://doi.org/10.1038/leu.2014.231.
O'Dea E, Hoffmann A. The regulatory logic of the NF-kappaB signaling system. Cold Spring Harb Perspect Biol. 2010;2(1):a000216. https://doi.org/10.1101/cshperspect.a000216.
Higgs G, Slack F. The multiple roles of microRNA-155 in oncogenesis. J Clin Bioinf. 2013;3(1):17. https://doi.org/10.1186/2043-9113-3-17.
He XH, Zhu W, Yuan P, Jiang S, Li D, Zhang HW, et al. miR-155 downregulates ErbB2 and suppresses ErbB2-induced malignant transformation of breast epithelial cells. Oncogene. 2016;35:6015–25. https://doi.org/10.1038/onc.2016.132.
Qin W, Ren Q, Liu T, Huang Y, Wang J. MicroRNA-155 is a novel suppressor of ovarian cancer-initiating cells that targets CLDN1. FEBS Lett. 2013;587(9):1434–9. https://doi.org/10.1016/j.febslet.2013.03.023.
Kim S, Song JH, Kim S, Qu P, Martin BK, Sehareen WS, et al. Loss of oncogenic miR-155 in tumor cells promotes tumor growth by enhancing C/EBP-beta-mediated MDSC infiltration. Oncotarget. 2016;7(10):11094–112. https://doi.org/10.18632/oncotarget.7150.
Mattiske S, Suetani RJ, Neilsen PM, Callen DF. The oncogenic role of miR-155 in breast cancer. Cancer Epidemiol Biomark Prev. 2012;21(8):1236–43. https://doi.org/10.1158/1055-9965.EPI-12-0173.
Lei C, Wang Y, Huang Y, Yu H, Huang Y, Wu L, et al. Up-regulated miR155 reverses the epithelial-mesenchymal transition induced by EGF and increases chemo-sensitivity to cisplatin in human Caski cervical cancer cells. PLoS One. 2012;7(12):e52310. https://doi.org/10.1371/journal.pone.0052310.
Mathelier A, Fornes O, Arenillas DJ, Chen CY, Denay G, Lee J, et al. JASPAR 2016: a major expansion and update of the open-access database of transcription factor binding profiles. Nucleic Acids Res. 2016;44(D1):D110–5. https://doi.org/10.1093/nar/gkv1176.
Lu D, Nakagawa R, Lazzaro S, Staudacher P, Abreu-Goodger C, Henley T, et al. The miR-155-PU.1 axis acts on Pax5 to enable efficient terminal B cell differentiation. J Exp Med. 2014;211(11):2183–98. https://doi.org/10.1084/jem.20140338.
Beishline K, Azizkhan-Clifford J. Sp1 and the ‘hallmarks of cancer’. FEBS J. 2015;282(2):224–58. https://doi.org/10.1111/febs.13148.
Acknowledgements
We gladly acknowledge the support by grants from the New Brunswick (NB) Innovation Foundation; the Canadian Breast Cancer Foundation; the Beatrice Hunter Cancer Research Institute; the Canadian Breast Cancer Society/QEII Foundation; and, the NB Health Research Foundation. Salaries for JH and NL are supported by the Beatrice Hunter Cancer Research Institute with funds provided by the Terry Fox Strategic Health Research Training Program in Cancer Research at CIHR in partnership with the NB Health Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Supplementary Table 1
(PPTX 50 kb)
Rights and permissions
About this article
Cite this article
Harquail, J., LeBlanc, N., Landry, C. et al. Pax-5 Inhibits NF-κB Activity in Breast Cancer Cells Through IKKε and miRNA-155 Effectors. J Mammary Gland Biol Neoplasia 23, 177–187 (2018). https://doi.org/10.1007/s10911-018-9404-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10911-018-9404-4