Abstract
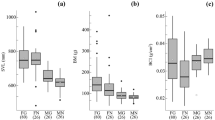
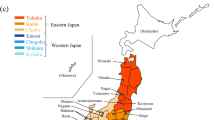
The Japanese natricine snake Rhabdophis tigrinus sequesters cardiotonic steroids, bufadienolides (BDs), from ingested toads in the nuchal glands as defensive toxins. A previous study showed that R. tigrinus in captivity converts dietary BDs when it sequesters them. However, it is unknown whether the dietary BDs are actually converted and the modified products accumulated under natural conditions. It is also unknown to what extent the BD profile of ingested toads is reflected in that of the snake. We collected 123 snakes from throughout Japan, analyzed their BD profiles by liquid chromatography/mass spectrometry, and identified 15 BDs from R. tigrinus by nuclear magnetic resonance analyses. We also compared their BD profiles using hierarchical cluster analysis (HCA). HCA exhibited two main clusters associated with their collection locations: eastern and western regions of the Japanese main islands. These results, coupled with previous findings on the BDs of Japanese toads, suggest that 1) R. tigrinus converts toad-derived BDs into other compounds under natural conditions; 2) there are both universal and regionally-specific conversions of dietary BDs by R. tigrinus; and 3) geographic variation in toad BD profiles is partially reflected in the variation of snake BD profiles.







Similar content being viewed by others
Data Availability
Uploading and archiving of auxiliary data files is not required as all data related to this manuscript is contained in the supplementary material.
Code Availability
Not applicable.
References
Akizawa T, Yasuhara T, Kano R, Nakajima T (1985) Novel polyhydroxylated cardiac steroids in the nuchal glands of the snake, Rhabdophis tigrinus. Biomed Res 6:437–444. https://doi.org/10.2220/biomedres.6.437
de Sousa LQ, da Conceição MK, de Carvalho Oliveira SF, da Silva AL, dos Santos M-F, de Carvalho Melo-Cavalcante AA, Veira-Júnior GM, Pinheiro Ferreira PM (2017) Bufadienolides from amphibians: A promising source of anticancer prototypes for radical innovation, apoptosis triggering and Na+K+ ATPase inhibition. Toxicon 127:63–76. https://doi.org/10.1016/j.toxicon.2017.01.004
Duffey SS (1980) Sequestration of plant natural products by insects. Ann Rev Entomol 25:447–477. https://doi.org/10.1146/annurev.en.25.010180.002311
Dumbacher JP, Wako A, Derrickson SR, Samuelson A, Spande TF, Daly JW (2004) Melyrid beetles (Choresine): A putative source for the batrachotoxin alkaloids found in poison-dart frogs and toxic passerine birds. Proc Natl Acad Sci USA 101:15857–15860. https://doi.org/10.1073/pnas.0407197101
González A, Schroeder FC, Attygalle AB, Svatoš A, Meinwald J, Eisner T (1999) Metabolic transformations of acquired lucibufagins by firefly ‘“femmes fatales.”’ Chemoecology 9:105–112. https://doi.org/10.1007/s000490050040
Green B, Crane RI, Khaidem IS, Leighton RS, Newaz SS, Smyser TE (1985) Synthesis of steroidal 16, 17-fused unsaturated–lactones. J Org Chem 50:640–644. https://doi.org/10.1021/jo00205a016
Hayes RA, Crossland MR, Hagman M, Capon RJ, Shine R (2009) Ontogenetic variation in the chemical defenses of cane toads (Bufo marinus): Toxin profiles and effects on predators. J Chem Ecol 35:391–399. https://doi.org/10.1007/s10886-009-9608-6
Hutchinson DA, Mori A, Savitzky AH, Burghardt GM, Wu X, Meinwald J, Schroeder FC (2007) Dietary sequestration of defensive steroids in nuchal glands of the Asian snake Rhabdophis tigrinus. Proc Natl Acad Sci USA 104:2265–2270. https://doi.org/10.1073/pnas.0610785104
Hutchinson DA, Savitzky AH (2004) Vasculature of the parotoid glands of four species of toads (Bufonidae: Bufo). J Morphol 260:247–254. https://doi.org/10.1002/jmor.10219
Hutchinson DA, Savitzky AH, Mori A, Burghardt GM, Meinwald J, Schroeder FC (2012) Chemical investigations of defensive steroid sequestration by the Asian snake Rhabdophis tigrinus. Chemoecology 22:199–206. https://doi.org/10.1007/s00049-011-0078-2
Igawa T, Kurabayashi A, Nishioka M, Sumida M (2006) Molecular phylogenetic relationship of toads distributed in the Far East and Europe inferred from the nucleotide sequences of mitochondrial DNA genes. Mol Phylogenet Evol 38:250–260. https://doi.org/10.1016/j.ympev.2005.09.003
Inoue T, Nakata R, Savitzky AH, Yoshinaga N, Mori A, Mori N (2020) Variation in bufadienolide composition of parotoid gland secretion from three taxa of Japanese toads. J Chem Ecol 46:997–1009. https://doi.org/10.1007/s10886-020-01217-y
Jones PL, Petschenka G, Flacht L, Agrawal AA (2019) Cardenolide intake, sequestration, and excretion by the Monarch Butterfly along gradients of plant toxicity and larval ontogeny. J Chem Ecol 45:264–277. https://doi.org/10.1007/s10886-019-01055-7
Kamano Y, Kotake A, Hashima H, Inoue M, Morita H, Takeya K, Itokawa H, Nandachi N, Segawa T, Yukita A, Saitou K, Katsuyama M, Pettit GR (1998) Structure–cytotoxic activity relationship for the toad poison bufadienolides. Bioorg Med Chem 6:1103–1115. https://doi.org/10.1016/S0968-0896(98)00067-4
Matsukawa M, Akizawa T, Ohigashi M, Morris JF, Butler VP Jr, Yoshioka M (1997) A novel bufadienolide, marinosin, in the skin of the giant toad, Bufo marinus. Chem Pharm Bull 45:249–254. https://doi.org/10.1248/cpb.45.249
Matsukawa M, Mukai T, Akizawa T, Miyatake S, Yoshioka M, Morris JF, Butler VP Jr (1998) Isolation and characterization of novel endogenous digitalis-like factors in the ovary of the giant toad, Bufo marinus. J Nat Prod 61:1476–1481. https://doi.org/10.1021/np980189g
McGugan JA, Byrd GD, Roland AB, Caty SN, Kabir N, Tapia EE, Trauger SA, Coloma LA, O’Connell LA (2016) Ant and mite diversity drives toxin variation in the Little Devil poison frog. J Chem Ecol 42:537–551. https://doi.org/10.1007/s10886-016-0715-x
Meng Q, Yau L-F, Lu J-G, Wu Z-Z, Zhang B-X, Wang J-R, Jiang Z-H (2016) Chemical profiling and cytotoxicity assay of bufadienolides in toad venom and toad skin. J Ethnopharmacol 187:74–82. https://doi.org/10.1016/j.jep.2016.03.062
Mohammadi S, Gompert Z, Gonzalez J, Takeuchi H, Mori A, Savitzky AH (2016) Toxin-resistant isoforms of Na+/K+-ATPase in snakes do not closely track dietary specialization on toads. Proc R Soc B 283:20162111. https://doi.org/10.1098/rspb.2016.2111
Mohammadi S, Petschenka G, French SS, Mori A, Savitzky AH (2018) Snakes exhibit tissue-specific variation in cardiotonic steroid sensitivity of Na+/K+-ATPase. Comp Biochem Physiol B 217:21–26. https://doi.org/10.1016/j.cbpb.2017.11.014]
Mori A, Burghardt GM (2017) Do tiger keelback snakes (Rhabdophis tigrinus) recognize how toxic they are? J Comp Psychol 131:257–265. https://doi.org/10.1037/com0000075
Mori A, Burghardt GM, Savitzky AH, Roberts KA, Hutchinson DA, Goris RC (2012) Nuchal glands: a novel defensive system in snakes. Chemoecology 22:187–198. https://doi.org/10.1007/s00049-011-0086-2
Mori A, Moriguchi H (1988) Food habits of the snakes in Japan: a critical review. Snake 20:98–113
Opitz SEW, Müller C (2009) Plant chemistry and insect sequestration. Chemoecology 19:117–154. https://doi.org/10.1007/s00049-009-0018-6
Rousseeuw PJ (1987) Silhouettes: a graphical aid to the interpretation and validation of cluster analysis. J Comput Appl Math 20:53–65. https://doi.org/10.1016/0377-0427(87)90125-7
Saporito RA, Donnelly MA, Garraffo HM, Spande TF, Daly JW (2006) Geographic and seasonal variation in alkaloid-based chemical defenses of Dendrobates pumilio from Bocas del Toro. Panama J Chem Ecol 32:795–814. https://doi.org/10.1007/s10886-006-9034-y
Savitzky AH, Mori A, Hutchinson DA, Saporito RA, Burghardt GM, Lillywhite HB, Meinwald J (2012) Sequestered defensive toxins in tetrapod vertebrates: principles, patterns, and prospects for future studies. Chemoecology 22:141–158. https://doi.org/10.1007/s00049-012-0112-z
Speed MH, Ruxton GD, Mappes J, Sherratt T (2012) Why are defensive toxins so variable? An evolutionary perspective. Biol Rev 87:874–884. https://doi.org/10.1111/j.1469-185X.2012.00228.x
Shimada K, Fujii Y, Yamashita E, Niizaki Y, Sato Y, Nambara T (1977) Studies on cardiotonic steroids from the skin of Japanese toad. Chem Pharm Bull 25:714–730. https://doi.org/10.1248/cpb.25.714
Takada W, Sakata T, Shimano S, Enami Y, Mori N (2005) Scheloribatid mites as the source of pumiliotoxins in dendrobatid frogs. J Chem Ecol 31:2403–2415. https://doi.org/10.1007/s10886-005-7109-9
Takeuchi H, Ota H, Oh H, Hikida T (2012) Extensive genetic divergence in the East Asian natricine snake, Rhabdophis tigrinus (Serpentes: Colubridae), with special reference to prominent geographical differentiation of the mitochondrial cytochrome b gene in Japanese populations. Biol J Linn Soc Lond 105:395–408. https://doi.org/10.1111/j.1095-8312.2011.01792.x
Takeuchi H, Savitzky AH, Ding L, de Silva A, Das I, Nguyen TT, Tsai TS, Jono T, Zhu GX, Mahaulpatha D, Tang Y, Mori A (2018) Evolution of nuchal glands, unusual defensive organs of Asian natricine snakes (Serpentes: Colubridae), inferred from a molecular phylogeny. Ecol Evol 8:10219–10232. https://doi.org/10.1002/ece3.4497
Tanaka K, Mori A (2000) Literature survey on predators of snakes in Japan. Curr Herpetol 19:97–111. https://doi.org/10.5358/hsj.19.97
Acknowledgements
We thank K. Eto, S. Fujii, K. Fujishima, M. Fukuda, I. Fukuyama, R. Fukuyama, K. Hamanaka, S. Ichihara, K. Kanda, T. Kodama, J. Marunouchi, T. Matsuki, H. Moriguchi, E. Moriki, K. Nishikawa, K. Niwa, T. Okamoto, M. Tagawa, S. Tanabe, S. Tsukamoto, S. Yoden, and the staff of Shizuoka Prefectural Forest Park for collecting snakes. We thank R. Ujiie and T. Yoshida for their keen insights during discussions.
Funding
This research was supported by grants from Japan–China Joint Research Project (2014–2016) between the Japan Society for the Promotion of Science (JSPS) and National Natural Science Foundation of China (NSFC, 31411140033) and JSPS KAKENHI Grant Numbers JP26440213, JP17H03719, and JP18KK0205. Additional funding was provided by Utah State University.
Author information
Authors and Affiliations
Contributions
Conceptualization: Inoue T, Nakata R, Savitzky AH, Yoshinaga N, Mori A, Mori N.
Data curation: Inoue T, Nakata R, Savitzky AH, Yoshinaga N, Mori A, Mori N.
Formal analysis: Inoue T, Nakata R, Yoshinaga N, Mori N.
Funding acquisition: Mori A.
Investigation: Inoue T, Nakata R, Savitzky AH, Mori A, Mori N.
Methodology: Inoue T, Nakata R, Yoshinaga N, Mori A, Mori N.
Project administration: Inoue T, Nakata R, Mori A, Mori N.
Resources: Inoue T, Mori A.
Supervision: Inoue T, Nakata R, Savitzky AH, Yoshinaga N, Mori A, Mori N.
Validation: Inoue T, Nakata R, Yoshinaga N, Mori A, Mori N.
Visualization: Inoue T, Nakata R, Savitzky AH, Yoshinaga N, Mori A, Mori N.
Writing – original draft: Inoue T, Nakata R.
Writing – review & editing: Inoue T, Nakata R, Savitzky AH, Mori A, Mori N.
Corresponding author
Ethics declarations
Conflicts of Interest/Competing Interests
Not applicable.
Ethics Approval
The present study was carried out in compliance with the guidelines of the Animal Care and Use Committee of Kyoto University.
Consent to Participate
Not applicable.
Consent for Publication
We consent to publication.
Additional information
All authors approved the manuscript to be published and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Inoue, T., Nakata, R., Savitzky, A. et al. New Insights Into Dietary Toxin Metabolism: Diversity in the Ability of the Natricine Snake Rhabdophis tigrinus to Convert Toad-Derived Bufadienolides. J Chem Ecol 47, 915–925 (2021). https://doi.org/10.1007/s10886-021-01287-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-021-01287-6