Abstract
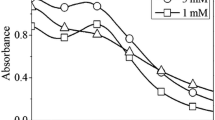
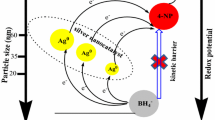
Silver nanoparticles (AgNPs) with two different morphologies were synthesized using aqueous extract of Cnidium officinale rhizomes under different reaction conditions and applied to the facile aqueous phase reduction of 4-nitroaniline (4-NA) as catalysts. The synthesized AgNPs are characterized by UV–Visible spectroscopy, Fourier transform infrared (FT-IR) spectroscopy, X-ray diffraction (XRD) method and high resolution transmission electron microscopy (HR-TEM) with energy dispersive X-ray spectroscopy (EDS) analysis. HR-TEM showed that the synthesized AgNPs had two different morphological forms; truncated triangular prism shapes with a mean size of ~35 nm and spherical shapes with a mean size of ~9 nm. The HR-TEM and XRD results are consistent with the standard values. The catalytic activities of both AgNPs were compared on the reduction of 4-NA in water by NaBH4 as a reducing agent. The reduction of 4-NA was monitored using UV–Visible spectroscopy. The ~9 nm (spherical) sized AgNPs showed superior catalytic activity than the larger AgNPs. The calculated rate constant (k) for the catalytic reduction of 4-nitroanilie by spherical AgNPs was 11.16 × 10−3 s−1.













Similar content being viewed by others
References
J. L. Elechiguerra, J. Reyes-Gasga, and M. J. Yacaman (2006). J. Mater. Chem. 16, 3906.
K. L. Kelly, E. Coronado, L. L. Zhao, and G. C. Schatz (2003). J. Phys. Chem. B 107, 668.
M. Chen, Y. Cai, Z. Yan, and D. W. Goodman (2006). J. Am. Chem. Soc. 128, 6341.
C. Medina, M. J. Santos-Martinez, A. Radomski, O. I. Corrigan, and M. W. Radomski (2007). Br. J. Pharmacol. 150, 552.
A. Moores and F. Goettmann (2006). New J. Chem. 30, 1121.
T. K. Sau, A. L. Rogach, F. Jackel, T. A. Klar, and J. Feldmann (2010). Adv. Mater. 22, 1805.
K. G. Stamplecoskie and J. C. Scaiano (2010). J. Am. Chem. Soc. 132, 1825.
Z. Sadowski, I. H. Maliszewska, B. Grochowalska, I. Polowczyk, and T. Kozlecki (2008). Mater. Sci-Poland 26, 419.
S. Yallappa and J. Manjanna (2014). J. Clust. Sci 25, 1449.
S. J. Hoseini, M. Darroudi, R. K. Oskuee, L. Gholami, and A. K. Zak (2015). Adv. Powder Technol 26, 991.
A. Miri, M. Sarani, M. R. Bazaz, and M. Darroudi (2015). Spectrochim. Acta A 141, 287.
M. Venkatesham, D. Ayodhya, A. Madhusudhan, A. S. Kumari, G. Veerabhadram, and K. G. Mangatayaru (2014). J. Clust. Sci. 25, 409.
S. Iravani (2011). Green Chem. 13, 2638.
S. A. Umoren, I. B. Obot, and Z. M. Gasem (2014). J. Mater. Environ. Sci. 5, 907.
M. Noruzi, D. Zare, and D. Davoodi (2012). Spectrochim. Acta A 94, 84.
J. B. Jeong, J. H. Park, H. K. Lee, S. Y. Ju, S. C. Hong, J. R. Lee, G. Y. Chung, J. H. Lim, and H. J. Jeong (2009). Food Chem. Toxicol. 47, 525.
M. Ramalingam and P. Yong-Ki (2010). Pharmacogn. Mag. 6, 323.
K. Bae, Y. Choi, S. Kim, and Y. Kim (2011). Molecules 16, 8833.
T. Tsukamoto, Y. Ishikawa, and M. Miyazawa (2005). J. Agric. Food Chem. 53, 5549.
J. Kwon and Y. Ahn (2002). J. Agric. Food Chem. 50, 4479.
R. V. Jagadeesh, G. Wienhofer, F. A. Westerhaus, A. Surkus, M. Pohl, H. Junge, K. Junge, and M. Beller (2011). Chem. Commun. 47, 10972.
K. Li, Z. Zheng, J. Feng, J. Zhang, X. Luo, G. Zhao, and X. Huang (2009). J. Hazard. Mater. 166, 1180.
S. Eustis and M. A. El-Sayed (2006). Chem. Soc. Rev. 35, 209.
M. Umadevi, M. R. Bindhu, and V. Sathe (2013). J. Mater. Sci. Technol. 29, 317.
T. J. I. Edison and M. G. Sethuraman (2013). Spectrochim. Acta A 104, 262.
A. Parveen and S. Rao (2015). J. Clust. Sci. 26, 1295.
S. L. Smitha, K. M. Nissamudeen, D. Philip, and K. G. Gopchandran (2008). Spectrochim. Acta A 71, 186.
T. J. I. Edison and M. G. Sethuraman (2012). Process Biochem. 47, 1351.
S. K. Mehta, S. Chaudhary, and M. Gradzielski (2010). J. Colloid Interface Sci. 343, 447.
A. J. Kora, S. R. Beedu, and A. Jayaraman (2012). Org. Med. Chem. Lett. 2, 1.
N. Basavegowda, K. B. S. Magar, K. Mishra, and Y. R. Lee (2014). New J. Chem. 38, 5415.
S. Sing, P. Patel, S. Jaiswal, A. A. Prabhune, C. V. Ramana, and B. L. V. Prasad (2009). New J. Chem. 33, 646.
N. Singh and P. K. Khanna (2007). Mater. Chem. Phys. 104, 367.
S. Li, Y. Shen, A. Xie, X. Yu, L. Qiu, L. Zhang, and Q. Zhang (2007). Green Chem. 9, 852.
S. Sadhasivam, P. Shanmugam, and K. Yun (2010). Colloids Surf. B 81, 358.
I. A. Wani, A. Ganguly, J. Ahmed, and T. Ahmad (2011). Mater. Lett. 65, 520.
B. Tang, L. Sun, J. Li, M. Zhang, and X. Wang (2015). Chem. Eng. J. 260, 99.
T. Tan, C. Tian, Z. Ren, J. Yang, Y. Chen, L. Sun, Z. Li, A. Wu, J. Yin, and H. Fu (2013). Phys. Chem. Chem. Phys. 15, 21034.
S. Kundu, K. Wang, and H. Liang (2009). J. Phys. Chem. C 113, 5157.
A. Gangula, R. Podila, M. Ramakrishna, L. Karanam, C. Janardhana, and A. M. Rao (2011). Langmuir 27, 15268.
A. Hernández-Gordillo and V. R. González (2015). Chem. Eng. J. 261, 53.
S. Wunder, F. Polzer, Y. Lu, Y. Mei, and M. Ballauff (2010). J. Phys. Chem. C 114, 8814.
V. Reddy, R. S. Torati, S. Oh, and C. Kim (2013). Ind. Eng. Chem. Res. 52, 556.
Acknowledgments
This study was supported by Priority Research Centers Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2014R1A6A1031189).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Edison, T.N.J.I., Baral, E.R., Lee, Y.R. et al. Biogenic Synthesis of Silver Nanoparticles Using Cnidium officinale Extract and Their Catalytic Reduction of 4-Nitroaniline. J Clust Sci 27, 285–298 (2016). https://doi.org/10.1007/s10876-015-0929-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-015-0929-z