Abstract
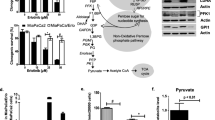
The multidrug resistance (MDR) phenotype, frequently observed during cancer treatment, is often associated with drug efflux pump activity. However, many other factors are also known to be involved. Cancer cells often rely on aerobic glycolysis for energy production; this is known as the “Warburg effect” and is used as a survival mechanism. Associated to this event, a reverse pH gradient across the cell membrane occurs, leading to cytosol alkalinization and extracellular acidification. In the present study, we investigated the role of different mechanisms involved in MDR, such as altered tumor microenvironment and energetic metabolism. The breast cancer cell line MCF-7, used as model, was exposed to two widely used antitumor drugs, paclitaxel (antimitotic agent) and doxorubicin (alkylating agent). Cancer pH regulation was shown to be crucial for malignant characteristics such as cell migration and drug resistance. Our results showed that a lower extracellular pH induced a higher migratory capacity and higher resistance to the studied chemotherapeutical compounds in MCF-7 cells. Besides the influence of the extracellular pH, the role of the tumor metabolism in the MDR phenotype was also investigated. Pre-treatment with different bioenergetic modulators led to cell ATP depletion and altered lactic acid production and glucose consumption, resulting in increased sensitivity to paclitaxel and doxorubicin. Overall, this study supports the potential use of compounds targeting cell metabolism and tumor microenvironment factors such as pH, as co-adjuvants in conventional chemotherapy.
Similar content being viewed by others
References
Belotti D, Rieppi M, Nicoletti MI (1996) Paclitaxel (TAXOL(R)) inhibits motility of paclitaxel-resistant human ovarian carcinoma cells. Clin Cancer Res 2:1725–1730
Brum G et al (2013) N-acetylcysteine potentiates doxorubicin-induced ATM and p53 activation in ovarian cancer cells. Int J Oncol 42:211–219
Cascorbi I (2006) Role of pharmacogenetics of ATP-binding cassette transporters in the pharmacokinetics of drugs. Pharmacol Ther 112:457–473
Chaudhari AA et al (2008) Mitochondrial transmembrane potential and reactive oxygen species generation regulate the enhanced effect of CCCP on TRAIL-induced SNU-638 cell apoptosis. J Vet Med Sci 70:537–542
Choi HJ et al (2008) Hexane/Ethanol of Glycyrrhiza Uralensis Licorice suppresses doxorubicin-induced apoptosis in H9c2 rat cardiac myoblasts. Exp Biol Med 233:1554–1560
Coley HM (2008) Mechanisms and strategies to overcome chemotherapy resistance in metastatic breast cancer. Cancer Treat Rev 34:378–390
Daniel C et al (2013) The role of proton dynamics in the development and maintenance o multidrug resistance in cancer. Biochim Biophys Acta. doi:10.1016/j.bbadis.2013.01.020
Ferguson R, Jackson S, Stanley A, Joyce A, Harnden P, Morrison E, Patel P, Phillips R, Selby P, Banks R (2005) Intrinsic chemotherapy resistance to the tubulin-binding antimitotic agents in renal cell carcinoma. Int J Cancer 115:155–163
Gatenby RA et al (2007) Cellular adaptations to hypoxia and acidosis during somatic evolution of breast cancer. Br J Cancer 97:646–653
Gillies RJ, Gatenby RA (2007) Hypoxia and adaptive landscapes in the evolution of carcinogenesis. Cancer Metastasis Rev 26:311–317
Gillies RJ et al (2004) pH Imaging: a review of Ph measurement methods and applications in cancers. Eng Med Biol Mag 2:57–64
Gillies RJ, Robey I, Gatenby RA (2008) Causes and consequences of increased glucose metabolism of cancers. J Nucl Med 49:24–42
Gottesman MM (2002) Mechanisms of cancer drug resistance. Annu Rev Med 53:615–627
Gottesman MM, Fojo T, Bates SE (2002) Multidrug resistance in cancer: role of ATP-dependent transporters. Nat Rev Cancer 2:48–58
Granchi C, Minutolo F (2012) Anticancer agents that counteract tumor glycolysis. Chem Med Chem 7:1318–1350
Gregory RE, DeLisa AF (1993) Paclitaxel: a new antineoplastic agent for refractory ovarian cancer. Clin Pharm 12:401–415
Hall M, Handley M, Gottesman M (2009) Is resistance useless? Multidrug resistance and collateral sensivity. Trends Pharmacol Sci 30:546–556
Heiden V, Matthew G (2011) Targeting cancer metabolism : a therapeutic window opens. Nat Rev Drug Discov 10:671–684
Hennequin C, Giocanti N, Favaudon V (1996) Interaction of ionizing radiation with paclitaxel (taxol) and docetaxel (taxotere) in Hela and SQ20B cells. Cancer Res 56:1842–1850
Izumi H et al (2003) Cellular pH regulators: potentially promising molecular targets for cancer chemotherapy. Cancer Treat Rev 29:541–549
Larsen A, Escargueil EA, Skladanowski A (2000) Resistance mechanisms associated with altered intracellular distribution of anticancer agents. Pharmacol Ther 85:217–229
Maher JC, Krishan A, Lampidis TJ (2004) Greater cell cycle inhibition and cytotoxicity induced by 2-deoxy-D-glucose in tumor cells treated under hypoxic vs aerobic conditions. Cancer Chemother Pharmacol 53:116–22
Moreno-Sánchez R et al (2007) Energy metabolism in tumor cells. FEBS J 274:1393–1418
Moutasim KA, Nystrom ML, Thomas GJ (2011) Cell migration and invasion assays. Methods Mol Biol 731:333–43
Neri D, Supuran CT (2011) Interfering with pH regulation in tumours as a therapeutic strategy. Discovery Nat Rev Drug Discov 10:767–777
Papandreou I, Goliasova T, Denko NC (2011) Anticancer drugs that target metabolism: is dichloroacetate the new paradigm? Int J Cancer 128:1001–1008
Queirós O et al (2012) Butyrate activates the monocarboxylate transporter MCT4 expression in breast cancer cells and enhances the antitumor activity of 3-bromopyruvate. J Bioenerg Biomembr 44:141–153
Raghunand N, Gillies RJ (2000) pH and drug resistance in tumors. Drug Resist Updat 3:39–47
Robey IF, Martin NK (2011) Bicarbonate and dicholoroacetate : evaluating pH altering therapies in a mouse model for metastatic breast cancer. BMC Cancer 11:1–10
Rohwer N, Cramer T (2011) Hypoxia-mediated drug resistance: novel insights on the functional interaction of HIFs and cell death pathways. Drug Resist Updat 14:191–201
Stockwin LH et al (2010) Sodium dichloroacetate selectively targets cells with defects in the mitochondrial ETC. Int J Cancer 127:2510–2519
Swietach P, Vaughan-Jones RD, Harris AL (2007) Regulation of tumor pH and the role of carbonic anhydrase 9. Cancer Metastasis Rev 26:299–310
Thews O et al (2006) Impact of extracellular acidity on the activity of P-glycoprotein and the cytotoxicity of chemotherapeutic drugs. Neoplasia 8:143–152
Tong J et al (2011) Synergistic antitumor effect of dichloroacetate in combination with 5-Fluorouracil in colorectal cancer. J Biomed Biotechnol 2011:1–7
Warburg O (1956) On the origin of cancer cells. Science 123:309–314
Wartenberg M et al (2010) Glycolytic pyruvate regulates P-Glycoprotein expression in multicellular tumor spheroids via modulation of the intracellular redox state. J Cell Biochem 109:434–446
Webb BA et al (2011) Dysregulated pH: a perfect storm for cancer progression. Nat Rev Cancer 11:671–677
Weljie AM, Jirik FR (2011) Hypoxia-induced metabolic shifts in cancer: moving beyond the Warburg effect. Int J Biochem Cell Biol 43:981–989
Williams AC, Collard TJ, Paraskeva C (1999) An acidic environment leads to p53 dependent induction of apoptosis in human adenoma and carcinoma cell lines: implications for clonal selection during colorectal carcinogenesis. Oncogene 18:3199–3204
Yuan J, Narayanan L, Rockwell S (2000) Diminished DNA repair and elevated mutagenesis in mammalian cells exposed to hypoxia and low pH. Cancer Res 60:4372–4376
Zong ZP et al (2004) Phorbol myristate induces apoptosis of taxol-resistant sarcoma cells in vitro. Eur J Pharmacol 489(1–2):3–11
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 566 KB)
Rights and permissions
About this article
Cite this article
Tavares-Valente, D., Baltazar, F., Moreira, R. et al. Cancer cell bioenergetics and pH regulation influence breast cancer cell resistance to paclitaxel and doxorubicin. J Bioenerg Biomembr 45, 467–475 (2013). https://doi.org/10.1007/s10863-013-9519-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-013-9519-7