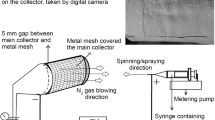
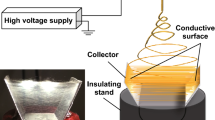
Abstract
In recent years, the engineering of biomimetic cellular microenvironments has emerged as a top priority for regenerative medicine, being the in vitro recreation of the arcade-like cartilaginous tissue one of the most critical challenges due to the notorious absence of cost- and time-efficient microfabrication techniques capable of building 3D fibrous scaffolds with precise anisotropic properties. Taking this into account, we suggest a feasible and accurate methodology that uses a sequential adaptation of an electrospinning-electrospraying set up to construct a hierarchical system comprising both polycaprolactone (PCL) fibres and polyethylene glycol sacrificial microparticles. After porogen leaching, the bi-layered PCL scaffold was capable of presenting not only a depth-dependent fibre orientation similar to natural cartilage, but also mechanical features and porosity proficient to encourage an enhanced cell response. In fact, cell viability studies confirmed the biocompatibility of the scaffold and its ability to guarantee suitable cell adhesion, proliferation and migration throughout the 3D anisotropic fibrous network during 21 days of culture. Additionally, likewise the hierarchical relationship between chondrocytes and their extracellular matrix, the reported PCL scaffold was able to induce depth-dependent cell-material interactions responsible for promoting a spatial modulation of the morphology, alignment and density of the cells in vitro.





Similar content being viewed by others
References
Rose JC, De Laporte L. Hierarchical design of tissue regenerative constructs. Adv Health Mater. 2018;7:1701067. https://doi.org/10.1002/adhm.201701067.
Benetti EM, Gunnewiek MK, van Blitterswijk CA, Julius Vancso G, Moroni L. Mimicking natural cell environments: design, fabrication and application of bio-chemical gradients on polymeric biomaterial substrates. J Mater Chem B. 2016;4:4244–57. https://doi.org/10.1039/c6tb00947f.
Malda J, Groll J, van Weeren PR. Rethinking articular cartilage regeneration based on a 250-year-old statement. Nat Rev Rheumatol. 2019;15:571–2. https://doi.org/10.1038/s41584-019-0278-7.
Camarero-Espinosa S, Rothen-Rutishauser B, Foster EJ, Weder C. Articular cartilage: from formation to tissue engineering. Biomater Sci. 2016;4:734–67. https://doi.org/10.1039/c6bm00068a.
Armiento AR, Stoddart MJ, Alini M, Eglin D. Biomaterials for articular cartilage tissue engineering: learning from biology. Acta Biomater. 2018;65:1–20. https://doi.org/10.1016/j.actbio.2017.11.021.
Onofrillo C, Duchi S, O’Connell CD, Blanchard R, O’Connor AJ, Scott M, et al. Biofabrication of human articular cartilage: a path towards the development of a clinical treatment. Biofabrication. 2018;10:045006. https://doi.org/10.1088/1758-5090/aad8d9.
Kwon H, Paschos NK, Hu JC, Athanasiou K. Articular cartilage tissue engineering: the role of signaling molecules. Cell Mol Life Sci. 2016;73:1173–94. https://doi.org/10.1007/s00018-015-2115-8.
Li K, Zhang C, Qiu L, Gao L, Zhang X. Advances in application of mechanical stimuli in bioreactors for cartilage tissue engineering. Tissue Eng Part B Rev. 2017;23:399–411. https://doi.org/10.1089/ten.teb.2016.0427.
Pedde RD, Mirani B, Navaei A, Styan T, Wong S, Mehrali M, et al. Emerging Biofabrication Strategies for Engineering Complex Tissue Constructs. Adv Mater. 2017;29:1606061. https://doi.org/10.1002/adma.201606061.
Li F, Truong VX, Fisch P, Levinson C, Glattauer V, Zenobi-Wong M, et al. Cartilage tissue formation through assembly of microgels containing mesenchymal stem cells. Acta Biomater. 2018;77:48–62. https://doi.org/10.1016/j.actbio.2018.07.015.
Yang X, Lu Z, Wu H, Li W, Zheng L, Zhao J. Collagen-alginate as bioink for three-dimensional (3D) cell printing based cartilage tissue engineering. Mater Sci Eng C. 2018;83:195–201. https://doi.org/10.1016/j.msec.2017.09.002.
Zhang Y, Ye L, Cui J, Yang B, Sun H, Li J, et al. A biomimetic poly(vinyl alcohol)–carrageenan composite scaffold with oriented microarchitecture. ACS Biomater Sci Eng. 2016;2:544–57. https://doi.org/10.1021/acsbiomaterials.5b00535.
Camarero-Espinosa S, Rothen-Rutishauser B, Weder C, Foster EJ. Directed cell growth in multi-zonal scaffolds for cartilage tissue engineering. Biomaterials. 2016;74:42–52. https://doi.org/10.1016/j.biomaterials.2015.09.033.
Stocco TD, Bassous NJ, Zhao S, Granato AEC, Webster TJ, Lobo AO. Nanofibrous scaffolds for biomedical applications. Nanoscale. 2018;10:12228–55. https://doi.org/10.1039/c8nr02002g.
Fiorani A, Gualandi C, Panseri S, Montesi M, Marcacci M, Focarete ML, et al. Comparative performance of collagen nanofibers electrospun from different solvents and stabilized by different crosslinkers. J Mater Sci Mater Med. 2014;25:2313–21. https://doi.org/10.1007/s10856-014-5196-2.
Lakra R, Kiran MS, Korrapati PS. Electrospun gelatin–polyethylenimine blend nanofibrous scaffold for biomedical applications. J Mater Sci Mater Med. 2019;30:129. https://doi.org/10.1007/s10856-019-6336-5.
Chen H, Malheiro AdBFB, van Blitterswijk C, Mota C, Wieringa PA, Moroni L. Direct writing electrospinning of scaffolds with multidimensional fiber architecture for hierarchical tissue engineering. ACS Appl Mater Interfaces. 2017;9:38187–200. https://doi.org/10.1021/acsami.7b07151.
Chen W, Xu Y, Liu Y, Wang Z, Li Y, Jiang G, et al. Three-dimensional printed electrospun fiber-based scaffold for cartilage regeneration. Mater Des. 2019;179:107886. https://doi.org/10.1016/j.matdes.2019.107886.
Bas O, De-Juan-Pardo EM, Meinert C, D’Angella D, Baldwin JG, Bray LJ, et al. Biofabricated soft network composites for cartilage tissue engineering. Biofabrication. 2017;9:025014. https://doi.org/10.1088/1758-5090/aa6b15.
Reboredo JW, Weigel T, Steinert A, Rackwitz L, Rudert M, Walles H. Investigation of migration and differentiation of human mesenchymal stem cells on five-layered collagenous electrospun scaffold mimicking native cartilage structure. Adv Health Mater. 2016;5:2191–8. https://doi.org/10.1002/adhm.201600134.
Walser J, Stok KS, Caversaccio MD, Ferguson SJ. Direct electrospinning of 3D auricle-shaped scaffolds for tissue engineering applications. Biofabrication. 2016;8:025007. https://doi.org/10.1088/1758-5090/8/2/025007.
Gao S, Chen M, Wang P, Li Y, Yuan Z, Guo W, et al. An electrospun fiber reinforced scaffold promotes total meniscus regeneration in rabbit meniscectomy model. Acta Biomater. 2018;73:127–40. https://doi.org/10.1016/j.actbio.2018.04.012.
Chen W, Chen S, Morsi Y, El-Hamshary H, El-Newhy M, Fan C, et al. Superabsorbent 3D scaffold based on electrospun nanofibers for cartilage tissue engineering. ACS Appl Mater Interfaces. 2016;8:24415–25. https://doi.org/10.1021/acsami.6b06825.
Sophia Fox AJ, Bedi A, Rodeo SA. The basic science of articular cartilage: structure, composition, and function. Sports Health. 2009;1:461–8. https://doi.org/10.1177/1941738109350438.
Barroca N, Marote A, Vieira SI, Almeida A, Fernandes MHV, Vilarinho PM, et al. Electrically polarized PLLA nanofibers as neural tissue engineering scaffolds with improved neuritogenesis. Colloids Surf B. 2018;167:93–103. https://doi.org/10.1016/j.colsurfb.2018.03.050.
Xiao J, Zhang Y, Wang J, Yu W, Wang W, Ma X. Monitoring of cell viability and proliferation in hydrogel-encapsulated system by resazurin assay. Appl Biochem Biotechnol. 2010;162:1996–2007. https://doi.org/10.1007/s12010-010-8975-3.
Stylianou A, Gkretsi V, Stylianopoulos T. Transforming growth factor-β modulates pancreatic cancer associated fibroblasts cell shape, stiffness and invasion. BBA-GEN Subjects. 2018;1862:1537–46. https://doi.org/10.1016/j.bbagen.2018.02.009.
Woodruff MA, Hutmacher DW. The return of a forgotten polymer—polycaprolactone in the 21st century. Prog Polym Sci. 2010;35:1217–56. https://doi.org/10.1016/j.progpolymsci.2010.04.002.
Wiggenhauser PS, Schwarz S, Koerber L, Hoffmann TK, Rotter N. Addition of decellularized extracellular matrix of porcine nasal cartilage improves cartilage regenerative capacities of PCL-based scaffolds in vitro. J Mater Sci Mater Med. 2019;30:121. https://doi.org/10.1007/s10856-019-6323-x.
Shitole AA, Raut PW, Sharma N, Giram P, Khandwekar AP, Garnaik B. Electrospun polycaprolactone/hydroxyapatite/ZnO nanofibers as potential biomaterials for bone tissue regeneration. J Mater Sci Mater Med. 2019;30:51. https://doi.org/10.1007/s10856-019-6255-5.
Wu J, Hong Y. Enhancing cell infiltration of electrospun fibrous scaffolds in tissue regeneration. Bioact Mater. 2016;1:56–64. https://doi.org/10.1016/j.bioactmat.2016.07.001.
Lavielle N, Hébraud A, Schlatter G, Thöny-Meyer L, Rossi RM, Popa A-M. Simultaneous electrospinning and electrospraying: a straightforward approach for fabricating hierarchically structured composite membranes. ACS Appl Mater Interfaces. 2013;5(20):10090–7. https://doi.org/10.1021/am402676m.
Bean AC, Tuan RS. Fiber diameter and seeding density influence chondrogenic differentiation of mesenchymal stem cells seeded on electrospun poly(ε-caprolactone) scaffolds. Biomed Mater. 2015;10:015018. https://doi.org/10.1088/1748-6041/10/1/015018.
Whited BM, Whitney JR, Hofmann MC, Xu Y, Rylander MN. Pre-osteoblast infiltration and differentiation in highly porous apatite-coated PLLA electrospun scaffolds. Biomaterials. 2011;32:2294–304. https://doi.org/10.1016/j.biomaterials.2010.12.003.
Wright LD, Andric T, Freeman JW. Utilizing NaCl to increase the porosity of electrospun materials. Mater Sci Eng C. 2011;31:30–6. https://doi.org/10.1016/j.msec.2010.02.001.
Wang J, Valmikinathan CM, Liu W, Laurencin CT, Yu X. Spiral-structured, nanofibrous, 3D scaffolds for bone tissue engineering. J Biomed Mater Res A. 2010;93A:753–62. https://doi.org/10.1002/jbm.a.32591.
Manoukian OS, Aravamudhan A, Lee P, Arul MR, Yu X, Rudraiah S, et al. Spiral layer-by-layer micro-nanostructured scaffolds for bone tissue engineering. ACS Biomater Sci Eng. 2018;4:2181–92. https://doi.org/10.1021/acsbiomaterials.8b00393.
Costantini M, Idaszek J, Szöke K, Jaroszewicz J, Dentini M, Barbetta A, et al. 3D bioprinting of BM-MSCs-loaded ECM biomimetic hydrogels for in vitro neocartilage formation. Biofabrication. 2016;8:035002. https://doi.org/10.1088/1758-5090/8/3/035002.
Naseri N, Poirier J-M, Girandon L, Fröhlich M, Oksman K, Mathew AP. 3-Dimensional porous nanocomposite scaffolds based on cellulose nanofibers for cartilage tissue engineering: tailoring of porosity and mechanical performance. RSC Adv. 2016;6:5999–6007. https://doi.org/10.1039/c5ra27246g.
McCullen SD, Autefage H, Callanan A, Gentleman E, Stevens MM. Anisotropic fibrous scaffolds for articular cartilage regeneration. Tissue Eng Part A. 2012;18:2073–83. https://doi.org/10.1089/ten.TEA.2011.0606.
Wang J, Shah A, Yu X. The influence of fiber thickness, wall thickness and gap distance on the spiral nanofibrous scaffolds for bone tissue engineering. Mater Sci Eng C. 2011;31:50–6. https://doi.org/10.1016/j.msec.2009.09.005.
Jiang H, Zuo Y, Zou Q, Wang H, Du J, Li Y, et al. Biomimetic spiral-cylindrical scaffold based on hybrid chitosan/cellulose/nano-hydroxyapatite membrane for bone regeneration. ACS Appl Mater Interfaces. 2013;5:12036–44. https://doi.org/10.1021/am4038432.
Morales TI. Chondrocyte moves: clever strategies? Osteoarthr Cartil. 2007;15:861–71. https://doi.org/10.1016/j.joca.2007.02.022.
Cai S, Xu H, Jiang Q, Yang Y. Novel 3D electrospun scaffolds with fibers oriented randomly and evenly in three dimensions to closely mimic the unique architectures of extracellular matrices in soft tissues: fabrication and mechanism study. Langmuir. 2013;29:2311–8. https://doi.org/10.1021/la304414j.
Youn I, Choi JB, Cao L, Setton LA, Guilak F. Zonal variations in the three-dimensional morphology of the chondron measured in situ using confocal microscopy. Osteoarthr Cartil. 2006;14:889–97. https://doi.org/10.1016/j.joca.2006.02.017.
Xu T, Miszuk JM, Zhao Y, Sun H, Fong H. Electrospun polycaprolactone 3D nanofibrous scaffold with interconnected and hierarchically structured pores for bone tissue engineering. Adv Health Mater. 2015;4:2238–46. https://doi.org/10.1002/adhm.201500345.
Miszuk JM, Xu T, Yao Q, Fang F, Childs JD, Hong Z, et al. Functionalization of PCL-3D electrospun nanofibrous scaffolds for improved BMP2-induced bone formation. Appl Mater Today. 2018;10:194–202. https://doi.org/10.1016/j.apmt.2017.12.004.
Acknowledgements
This work was supported by the Portuguese funding of Program COMPETE-FEDER, Programa Operacional Competitividade e Internacionalização through the projects POCI-01-0145-FEDER-016574, POCI-01-0145-FEDER-028424 and CENTRO-01-0145-FEDER-022083. Also by Fundação para a Ciência e Tecnologia I.P. (FCT, IP) through the projects PTDC/EME-SIS/28424/2017, PTDC/EMSTEC/3263/2014, IF/00917/2013/CP1162/CT0016 and UID/EMS/00481/2013. The authors thank to FCT for the PhD grants SFRH/BD/130287/2017 and SFRH/BD/133129/2017 and for the Investigator Program IF/00917/2013. The authors are also grateful for the generosity and valuable input of Gonçalo Ramalho, Susana Cristina, Nathalie Barroca and Nuno Almeida.
Authors contributions
AFG: conceptualisation, investigation, writing—original draft. AS: conceptualisation, investigation, writing—review and editing. ALP: validation. AC: writing—review and editing, supervision. PAAPM: writing—review and editing, supervision.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Girão, A.F., Semitela, Â., Pereira, A.L. et al. Microfabrication of a biomimetic arcade-like electrospun scaffold for cartilage tissue engineering applications. J Mater Sci: Mater Med 31, 69 (2020). https://doi.org/10.1007/s10856-020-06407-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10856-020-06407-4