Abstract
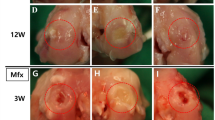
We have developed crosslinked salmon-derived atelocollagen (SC) sponge, which has a denaturation temperature of 47°C. Sixty-four knees of 32 mature rabbits were randomly divided into 4 groups after creating an osteochondral defect in the femoral trochlea. Defects in Groups I, II, and III were filled with the crosslinked SC sponge, the crosslinked porcine collagen (PC) sponge, and the non-crosslinked PC sponge, respectively. In Group IV, defects were left untreated as the control. At 12 weeks after implantation, the histological score showed that Group I was significantly greater than Groups III (P = 0.0196) and IV (P = 0.0021). In addition, gene expression of type-2 collagen, aggrecan, and SOX9 was the greatest in Group I at 12 weeks. The fundamental in vivo properties of the crosslinked SC sponge showed that this is a promising biomaterial, specifically as a scaffold for cartilage tissue engineering.






Similar content being viewed by others
References
McPherson JM, Sawamura S, Armstrong R. An examination of the biologic response to injectable, glutaraldehyde crosslinked collagen implants. J Biomed Mater Res. 1986;20:93–107.
Swatschek D, Schatton W, Kellermann J, Muller WE, Kreuter J. Marine sponge collagen: isolation, characterization and effects on the skin parameters surface-pH, moisture and sebum. Eur J Pharm Biopharm. 2002;53:107–13.
Burjanadze TV. New analysis of the phylogenetic change of collagen thermostability. Biopolymers. 2000;53:523–8.
Yunoki S, Nagai N, Suzuki T, Munekata M. Novel biomaterial from reinforced salmon collagen gel prepared by fibril formation and cross-linking. J Biosci Bioeng. 2004;98:40–7.
Nagai N, Yunoki S, Suzuki T, Sakata M, Tajima K, Mumekata M. Application of crosslinked salmon atelocollagen to the scaffold of human periodontal ligament cells. J Biosci Bioeng. 2004;97:389–94.
Nagai N, Mori K, Saoh Y, Takahashi N, Yunoki S, Tajima K, Munekata M. In vitro growth and differentiated activities of human periodontal ligament fibroblasts cultured on salmon collagen gel. J Biomed Mater Res A. 2007;82:395–402.
Nagai N, Mori K, Munekata M. Biological properties ofcrosslinked salmon collagen fibrillar gel as a scaffold for human umbilical vein endothelial cells. J Biomater Appl. 2008;23:275–87.
Iwasa J, Ochi M, Uchio Y, Katsube K, Adachi N, Kawasaki K. Effects of cell density on proliferation and matrix synthesis of chondrocytes embedded in atelocollagen gel. Artif Organs. 2003;27:249–55.
Nehrer S, Breinan HA, Ramappa A, Shortkroff S, Young G, Minas T, Sledge CB, Yannas IV, Spector M. Canine chondrocytes seeded in type I and type II collagen implants investigated in vitro. J Biomed Mater Res. 1997;38:95–104.
Jiang CC, Chiang H, Liao CJ, Lin YJ, Kuo TF, Shieh CS, Huang YY, Tuan RS. Repair of porcine articular cartilage defect with a biphasic osteochondral composite. Repair of porcine articular cartilage defect with a biphasic osteochondral composite. J Orthop Res. 2007;25:1277–90.
Dorotka R, Windberger U, Macfelda K, Bindreiter U, Toma C, Nehrer S. Repair of articular cartilage defects treated by microfracture and a three-dimensional collagen matrix. Biomaterials. 2005;26:3617–29.
Yunoki S, Suzuki T, Takai M. Stabilization of low denaturation temperature collagen from fish by physical cross-linking methods. J Biosci Bioeng. 2003;96:575–7.
Wayne JS, Mcdoell CL, Shields KJ, Tuan RS. In vivo response of polylactic acid-alginate scaffolds and bone marrow-derived cells for cartilage tissue engineering. Tissue Eng. 2005;11:953–63.
Nishikawa A, Taira T, Yoshizato K. In vitro maturation of collagen fibrils modulates spreading, DNA synthesis, and collagenolysis of epidermal cells and fibroblasts. Exp Cell Res. 1987;171:164–77.
Kuzuya M, Satake S, Ai S, Asai T, Kanda S, Ramos MA, Miura H, Ueda M, Iguchi A. Inhibition of angiogenesis on glycated collagen lattices. Diabetologia. 1998;41:491–9.
Semler EJ, Ranucci CS, Moghe PV. Mechanochemical manipulation of hepatocyte aggregation can selectively induce or repress liver-specific function. Biotechnol Bioeng. 2000;69:359–69.
Acknowledgments
We thank Yuko Taniguro for assistance with the animal experiments, and Dr. Yoshie Tanabe for data collection. This work was financially supported by grants from the Ministry of Education, Culture, Sports, Science and Technology, Japan, and from Takeda Science Foundation, Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kawaguchi, Y., Kondo, E., Kitamura, N. et al. In vivo effects of isolated implantation of salmon-derived crosslinked atelocollagen sponge into an osteochondral defect. J Mater Sci: Mater Med 22, 397–404 (2011). https://doi.org/10.1007/s10856-010-4215-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10856-010-4215-1