Abstract
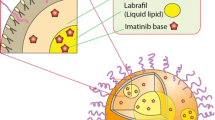
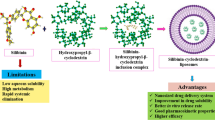
In this study, preparation and evaluation of liposomes, intended for intravenous administration, encapsulating synthetic MMP inhibitor (Ro 28-2653) – cyclodextrin complexes were realized. An increase in Ro solubility, via formation of binary (Ro/HPβCD) or ternary (Ro/HPβCD/L-lysine) complexes, permitted a similar increase in encapsulation efficiency of liposomes (Table 1). Moreover, Ro release kinetics depend on the encapsulation efficiency.

Similar content being viewed by others
References
Grams, F., et al: Pyrimidine-2,4,6-Triones: a new effective and selective class of matrix metalloproteinase inhibitors. Biol. Chem. 382, 1277–1285 (2001)
Lein, M., et al: The new synthetic matrix metalloproteinase inhibitor (Roche 28-2653) reduces tumor growth and prolongs survival in a prostate cancer standard rat model. Oncogene 21, 2089–2096 (2002)
Maquoi, E., et al: Anti-invasive, antitumoral, and antiangiogenic efficacy of a pyrimidine-2,4,6-trione derivative, an orally active and selective matrix metalloproteinases inhibitor. Clin. Cancer Res. 10, 4038–4047 (2004)
Bertholet, P., et al: The effect of cyclodextrins on the aqueous solubility of a new MMP inhibitor: phase solubility, 1H-NMR spectroscopy and molecular modeling studies, preparation and stability study of nebulizable solutions. J. Pharm. Pharm. Sci. 8, 164–175 (2005)
Acknowledgements
This work was supported by the Ministry of the Walloon Region (Belgium). The authors are grateful to F. Sideri for her aid during the experimental work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Piette, M., Castagne, D., Delattre, L. et al. Preparation and evaluation of liposomes encapsulating synthetic MMP inhibitor (Ro 28-2653)—cyclodextrin complexes. J Incl Phenom Macrocycl Chem 57, 101–103 (2007). https://doi.org/10.1007/s10847-006-9214-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-006-9214-y