Abstract
Background
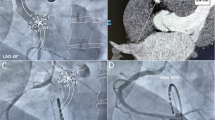
Despite luminal esophageal temperature (LET) monitoring, esophageal injury remains a risk which impacts decision making during atrial fibrillation (AF) ablation. We sought to compare procedural characteristics including radiofrequency (RF) power, duration, and LET, among ablation procedures with and without image segmentation for esophageal visualization (EV).
Methods
The retrospective cohort included 73 patients (mean age 65.2 ± 8.6 years, 36% female, 55% paroxysmal AF) who underwent pre-procedural cardiac magnetic resonance or computed tomography and LET monitoring. Of all patients, 35 were historical patients that underwent standard AF ablation without EV, and 38 were contemporary patients, 28 of whom underwent AF ablation with EV and 10 that underwent AF ablation without EV.
Results
Total RF time was similar between the groups. The distribution of ablation power delivery was skewed toward higher power in the contemporary patients. However, among patients in the contemporary group, the proportion of > 35 Watts lesions was lower with EV (P < 0.001). There was no difference between the max or mean LET. The standard deviation of LET change within patient during posterior wall ablation was lower in those with esophageal visualization compared to historical controls, but no change was seen compared to a smaller group of contemporary controls. No long-term clinical esophageal injury was observed.
Conclusions
In a retrospective analysis, EV was successfully performed in 28 patients. EV impacted RF power delivery decisions but was unassociated with RF time, changes in LET, or long-term safety.

Similar content being viewed by others
References
Nattel S. New ideas about atrial fibrillation 50 years on. Nature. 2002;415:219–26.
Tzou WS, Marchlinski FE, Zado ES, et al. Long-term outcome after successful catheter ablation of atrial fibrillation. Circ Arrhythm Electrophysiol. 2010;3:237–42.
Katz-Agranov N, Nevah Rubin MI. Severe esophageal injury after radiofrequency ablation - a deadly complication. World J Gastroenterol. 2017;23:3374–8.
Di Biase L, Saenz LC, Burkhardt DJ, et al. Esophageal capsule endoscopy after radiofrequency catheter ablation for atrial fibrillation: documented higher risk of luminal esophageal damage with general anesthesia as compared with conscious sedation. Circ Arrhythm Electrophysiol. 2009;2:108–12.
Rillig A, Meyerfeldt U, Birkemeyer R, et al. Oesophageal temperature monitoring and incidence of oesophageal lesions after pulmonary vein isolation using a remote robotic navigation system. Europace. 2010;12:655–61.
Singh SM, d’Avila A, Doshi SK, et al. Esophageal injury and temperature monitoring during atrial fibrillation ablation. Circ Arrhythm Electrophysiol. 2008;1:162–8.
Lemola K, Sneider M, Desjardins B, et al. Computed tomographic analysis of the anatomy of the left atrium and the esophagus: implications for left atrial catheter ablation. Circulation. 2004;110:3655–60.
Barbhaiya CR, Kumar S, Guo Y, et al. Global survey of esophageal injury in atrial fibrillation ablation: characteristics and outcomes of esophageal perforation and fistula. JACC Clin Electrophysiol. 2016;2:143–50.
Garg L, Garg J, Gupta N, et al. Gastrointestinal complications associated with catheter ablation for atrial fibrillation. Int J Cardiol. 2016;224:424–30.
Leite LR, Santos SN, Maia H, et al. Luminal esophageal temperature monitoring with a deflectable esophageal temperature probe and intracardiac echocardiography may reduce esophageal injury during atrial fibrillation ablation procedures: results of a pilot study. Circ Arrhythm Electrophysiol. 2011;4:149–56.
Bunch TJ, May HT, Crandall BG, et al. Intracardiac ultrasound for esophageal anatomic assessment and localization during left atrial ablation for atrial fibrillation. J Cardiovasc Electrophysiol. 2013;24:33–9.
Koruth JS, Reddy VY, Miller MA, et al. Mechanical esophageal displacement during catheter ablation for atrial fibrillation. J Cardiovasc Electrophysiol. 2012;23:147–54.
Tsuchiya T, Ashikaga K, Nakagawa S, Hayashida K, Kugimiya H. Atrial fibrillation ablation with esophageal cooling with a cooled water-irrigated intraesophageal balloon: a pilot study. J Cardiovasc Electrophysiol. 2007;18:145–50.
Nakahara S, Ramirez RJ, Buch E, et al. Intrapericardial balloon placement for prevention of collateral injury during catheter ablation of the left atrium in a porcine model. Heart Rhythm. 2010;7:81–7.
Deneke T, Bunz K, Bastian A, et al. Utility of esophageal temperature monitoring during pulmonary vein isolation for atrial fibrillation using duty-cycled phased radiofrequency ablation. J Cardiovasc Electrophysiol. 2011;22:255–61.
Koranne K, Basu-Ray I, Parikh V, et al. Esophageal temperature monitoring during radiofrequency ablation of atrial fibrillation: a meta-analysis. J Atr Fibrillation. 2016;9:1452.
Perzanowski C, Teplitsky L, Hranitzky PM, Bahnson TD. Real-time monitoring of luminal esophageal temperature during left atrial radiofrequency catheter ablation for atrial fibrillation: observations about esophageal heating during ablation at the pulmonary vein ostia and posterior left atrium. J Cardiovasc Electrophysiol. 2006;17:166–70.
Redfearn DP, Trim GM, Skanes AC, et al. Esophageal temperature monitoring during radiofrequency ablation of atrial fibrillation. J Cardiovasc Electrophysiol. 2005;16:589–93.
Ye Y, Chen SQ, Lu YF, et al. PV isolation guided by esophageal visualization with a tailored ablation strategy for the avoidance of esophageal thermal injury: a randomized trial. J Interv Card Electrophysiol. 2020;58:219–27.
Reddy VY, Grimaldi M, De Potter T, et al. Pulmonary vein isolation with very high power, short duration, temperature-controlled lesions: the QDOT-FAST trial. JACC Clin Electrophysiol. 2019;5:778–86.
Khoshknab M, Kuo L, Zghaib T, et al. Esophageal luminal temperature rise during atrial fibrillation ablation is associated with lower radiofrequency electrode distance and baseline impedance. J Cardiovasc Electrophysiol. 2021;32:1857–64.
Pappone C, Oral H, Santinelli V, et al. Atrio-esophageal fistula as a complication of percutaneous transcatheter ablation of atrial fibrillation. Circulation. 2004;109:2724–6.
Schmidt M, Nolker G, Marschang H, et al. Incidence of oesophageal wall injury post-pulmonary vein antrum isolation for treatment of patients with atrial fibrillation. Europace. 2008;10:205–9.
Funding
This work was supported by the Mark Marchlinski EP Research and Education Fund and indirectly by NIH R01HL142893 awarded to Dr. Nazarian.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The source cohort was approved by the University of Pennsylvania institutional review committee, and all patients gave informed consent for the use of clinical, imaging, and procedural data for medical research before the procedure.
Conflict of interest
Dr. Nazarian is a consultant for CardioSolv and Circle CVI; and principal investigator for research funding from Biosense Webster, ImriCor, Siemens, and ADAS software. The University of Pennsylvania Conflict of Interest Committee manages all commercial arrangements. The other authors report no conflicts of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khoshknab, M., Zghaib, T., Xu, L. et al. Esophageal image segmentation for guidance of posterior wall lesions during atrial fibrillation ablation. J Interv Card Electrophysiol 65, 543–550 (2022). https://doi.org/10.1007/s10840-022-01307-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-022-01307-9