Abstract
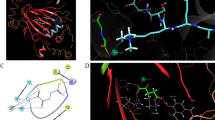
In this work, we analyze the structure–activity relationships (SAR) of epigenetic inhibitors (lysine mimetics) against lysine methyltransferase (G9a or EHMT2) using a combined activity landscape, molecular docking and molecular dynamics approach. The study was based on a set of 251 G9a inhibitors with reported experimental activity. The activity landscape analysis rapidly led to the identification of activity cliffs, scaffolds hops and other active an inactive molecules with distinct SAR. Structure-based analysis of activity cliffs, scaffold hops and other selected active and inactive G9a inhibitors by means of docking followed by molecular dynamics simulations led to the identification of interactions with key residues involved in activity against G9a, for instance with ASP 1083, LEU 1086, ASP 1088, TYR 1154 and PHE 1158. The outcome of this work is expected to further advance the development of G9a inhibitors.







Similar content being viewed by others
References
Miranda-Gonçalves V, Lameirinhas A, Henrique R, Jerónimo C (2018) Metabolism and epigenetic interplay in cancer: regulation and putative therapeutic targets. Front Genet 9:427. https://doi.org/10.3389/fgene.2018.00427
Rabal O, Castellar A, Oyarzabal J (2018) Novel pharmacological maps of protein lysine methyltransferases: key for target deorphanization. J Cheminform 10:32. https://doi.org/10.1186/s13321-018-0288-5
Rathert P, Dhayalan A, Murakami M et al (2008) Protein lysine methyltransferase G9a acts on non-histone targets. Nat Chem Biol 4:344–346. https://doi.org/10.1038/nchembio.88
Huang J, Dorsey J, Chuikov S et al (2010) G9a and Glp methylate lysine 373 in the tumor suppressor p53. J Biol Chem 285:9636–9641. https://doi.org/10.1074/jbc.M109.062588
Casciello F, Windloch K, Gannon F, Lee JS (2015) Functional role of g9a histone methyltransferase in cancer. Front Immunol 6:487. https://doi.org/10.3389/fimmu.2015.00487
Chen M-W, Hua K-T, Kao H-J et al (2010) H3K9 histone methyltransferase G9a promotes lung cancer invasion and metastasis by silencing the cell adhesion molecule Ep-CAM. Cancer Res 70:7830–7840. https://doi.org/10.1158/0008-5472.CAN-10-0833
Hua K-T, Wang M-Y, Chen M-W et al (2014) The H3K9 methyltransferase G9a is a marker of aggressive ovarian cancer that promotes peritoneal metastasis. Mol Cancer 13:189. https://doi.org/10.1186/1476-4598-13-189
Bárcena-Varela M, Caruso S, Llerena S et al (2019) Dual targeting of histone methyltransferase G9a and DNA-methyltransferase 1 for the treatment of experimental hepatocellular carcinoma. Hepatology 69:587–603. https://doi.org/10.1002/hep.30168
Segovia C, San José-Enériz E, Munera-Maravilla E et al (2019) Inhibition of a G9a/DNMT network triggers immune-mediated bladder cancer regression. Nat Med 25:1073–1081. https://doi.org/10.1038/s41591-019-0499-y
Imai K, Togami H, Okamoto T (2010) Involvement of histone H3 lysine 9 (H3K9) methyltransferase G9a in the maintenance of HIV-1 latency and its reactivation by BIX01294. J Biol Chem 285:16538–16545. https://doi.org/10.1074/jbc.M110.103531
Antignano F, Burrows K, Hughes MR et al (2014) Methyltransferase G9A regulates T cell differentiation during murine intestinal inflammation. J Clin Invest 124:1945–1955. https://doi.org/10.1172/JCI69592
Benevento M, van de Molengraft M, van Westen R et al (2015) The role of chromatin repressive marks in cognition and disease: A focus on the repressive complex GLP/G9a. Neurobiol Learn Mem 124:88–96. https://doi.org/10.1016/j.nlm.2015.06.013
Epsztejn-Litman S, Feldman N, Abu-Remaileh M et al (2008) De novo DNA methylation promoted by G9a prevents reprogramming of embryonically silenced genes. Nat Struct Mol Biol 15:1176–1183. https://doi.org/10.1038/nsmb.1476
Fukuda M, Sakaue-Sawano A, Shimura C et al (2019) G9a-dependent histone methylation can be induced in G1 phase of cell cycle. Sci Rep 9:956. https://doi.org/10.1038/s41598-018-37507-5
Shi Y, Desponts C, Do JT et al (2008) Induction of pluripotent stem cells from mouse embryonic fibroblasts by Oct4 and Klf4 with small-molecule compounds. Cell Stem Cell 3:568–574. https://doi.org/10.1016/j.stem.2008.10.004
Charles MRC, Dhayalan A, Hsieh H-P, Coumar MS (2019) Insights for the design of protein lysine methyltransferase G9a inhibitors. Future Med Chem 11:993–1014. https://doi.org/10.4155/fmc-2018-0396
Zang L, Kondengaden SM, Zhang Q et al (2017) Structure based design, synthesis and activity studies of small hybrid molecules as HDAC and G9a dual inhibitors. Oncotarget 8:63187–63207. https://doi.org/10.18632/oncotarget.18730
San José-Enériz E, Agirre X, Rabal O et al (2017) Discovery of first-in-class reversible dual small molecule inhibitors against G9a and DNMTs in hematological malignancies. Nat Commun 8:15424. https://doi.org/10.1038/ncomms15424
Rabal O, San José-Enériz E, Agirre X et al (2018) Discovery of reversible DNA methyltransferase and lysine methyltransferase G9a inhibitors with antitumoral in vivo efficacy. J Med Chem 61:6518–6545. https://doi.org/10.1021/acs.jmedchem.7b01926
Rodriguez-Madoz JR, San Jose-Eneriz E, Rabal O et al (2017) Reversible dual inhibitor against G9a and DNMT1 improves human iPSC derivation enhancing MET and facilitating transcription factor engagement to the genome. PLoS ONE 12:e0190275. https://doi.org/10.1371/journal.pone.0190275
Rabal O, Sánchez-Arias JA, San José-Enériz E et al (2018) Detailed exploration around 4-aminoquinolines chemical space to navigate the lysine methyltransferase G9a and DNA methyltransferase biological spaces. J Med Chem 61:6546–6573. https://doi.org/10.1021/acs.jmedchem.7b01925
López-López E, Prieto-Martínez FD, Medina-Franco JL (2018) Activity landscape and molecular modeling to explore the SAR of dual epigenetic inhibitors: A focus on G9a and DNMT1. Molecules. https://doi.org/10.3390/molecules23123282
Yu M, LI Z, Q L (2017) HISTONE METHYLTRANSFERASE INHIBITORS. 95.
Yu M, LI Z (2018) HISTONE METHYLTRANSFERASE INHIBITORS. 206.
Gaulton A, Hersey A, Nowotka M et al (2017) The ChEMBL database in 2017. Nucleic Acids Res 45:D945–D954. https://doi.org/10.1093/nar/gkw1074
Yongye AB, Byler K, Santos R et al (2011) Consensus models of activity landscapes with multiple chemical, conformer, and property representations. J Chem Inf Model 51:1259–1270. https://doi.org/10.1021/ci200081k
González-Medina M, Méndez-Lucio O, Medina-Franco JL (2017) Activity landscape plotter: a web-based application for the analysis of structure-activity relationships. J Chem Inf Model 57:397–402. https://doi.org/10.1021/acs.jcim.6b00776
López-López E, Naveja JJ, Medina-Franco JL (2019) DataWarrior: an evaluation of the open-source drug discovery tool. Expert Opin Drug Discov 14:335–341. https://doi.org/10.1080/17460441.2019.1581170
Trott O, Olson AJ (2010) AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J Comput Chem 31:455–461. https://doi.org/10.1002/jcc.21334
Medina-Franco JL (2012) Scanning structure-activity relationships with structure-activity similarity and related maps: from consensus activity cliffs to selectivity switches. J Chem Inf Model 52:2485–2493. https://doi.org/10.1021/ci300362x
Medina-Franco JL, Naveja JJ, López-López E (2019) Reaching for the bright StARs in chemical space. Drug Discov Today. https://doi.org/10.1016/j.drudis.2019.09.013
Bajorath J (2017) Computational scaffold hopping: cornerstone for the future of drug design? Future Med Chem 9:629–631. https://doi.org/10.4155/fmc-2017-0043
Maggiora GM (2006) On outliers and activity cliffs–why QSAR often disappoints. J Chem Inf Model 46:1535. https://doi.org/10.1021/ci060117s
Guha R, Van Drie JH (2008) Structure–activity landscape index: identifying and quantifying activity cliffs. J Chem Inf Model 48:646–658. https://doi.org/10.1021/ci7004093
Bemis GW, Murcko MA (1996) The properties of known drugs. 1. Molecular frameworks. J Med Chem 39:2887–2893. https://doi.org/10.1021/jm9602928
Naveja JJ, Norinder U, Mucs D et al (2018) Chemical space, diversity and activity landscape analysis of estrogen receptor binders. RSC Adv 8:38229–38237. https://doi.org/10.1039/C8RA07604A
Vedadi M, Barsyte-Lovejoy D, Liu F et al (2011) A chemical probe selectively inhibits G9a and GLP methyltransferase activity in cells. Nat Chem Biol 7:566–574. https://doi.org/10.1038/nchembio.599
Krieger E, Joo K, Lee J et al (2009) Improving physical realism, stereochemistry, and side-chain accuracy in homology modeling: four approaches that performed well in CASP8. Proteins 77(Suppl 9):114–122. https://doi.org/10.1002/prot.22570
Chemical Computing Group Inc (2019) Molecular operating environment (MOE). Chemical Computing Group Inc., Montreal
Cho AE, Guallar V, Berne BJ, Friesner R (2005) Importance of accurate charges in molecular docking: quantum mechanical/molecular mechanical (QM/MM) approach. J Comput Chem 26:915–931. https://doi.org/10.1002/jcc.20222
J. Bowers K, E. Chow D, Xu H, et al. (2006) Scalable algorithms for molecular dynamics simulations on commodity clusters.
Rea S, Eisenhaber F, O’Carroll D et al (2000) Regulation of chromatin structure by site-specific histone H3 methyltransferases. Nature 406:593–599. https://doi.org/10.1038/35020506
Shi C, Rafal PW, Fanwang M et al (2019) The dynamic conformational landscape of the protein methyltransferase SETD8. eLIFE 8:e45403. https://doi.org/10.7554/eLife.45403
Matthieu S (2016) Chemical inhibition of protein methyltransferases. Cell Chem Biol 9:1067–1076. https://doi.org/10.1016/j.chembiol.2016.07.014
Po H, Shenglong W, Yingkai Z (2008) How do SET-domain protein lysine methyltransferases achieve the methylation state specificity? Revisited by ab initio QM/MM molecular dynamics simulations. J Am Chem Soc 12:3806–3813. https://doi.org/10.1021/ja075896n
Shi C, Kanishk K, Chamara S et al (2019) Substrate-differentiated transition states of SET7/9-catalyzed lysine methylation. J Am Chem Soc 20:8064–8067. https://doi.org/10.1021/jacs.9b02553
Medina-Franco JL (2016) Epi-Informatics. https://doi.org/10.1016/C2014-0-03789-6
Kulkarni RA, Montgomery DC, Meier JL (2019) Epigenetic regulation by endogenous metabolite pharmacology. Curr Opin Chem Biol 51:30–39. https://doi.org/10.1016/j.cbpa.2019.02.002
Naveja JJ, Medina-Franco JL (2018) Insights from pharmacological similarity of epigenetic targets in epipolypharmacology. Drug Discov Today 23:141–150. https://doi.org/10.1016/j.drudis.2017.10.006
Prieto-Martínez FD, López-López E, Eurídice Juárez-Mercado K, Medina-Franco JL (2019) Computational drug design methods—current and future perspectives. In: Roy K (ed) silico drug design. Elsevier, Amsterdam, pp 19–44. https://doi.org/10.1016/B978-0-12-816125-8.00002-X
Norinder U, Naveja JJ, López-López E et al (2019) Conformal prediction of HDAC inhibitors. SAR QSAR Environ Res 30:265–277. https://doi.org/10.1080/1062936X.2019.1591503
Funding
This research was funded by the School of Chemistry of the Universidad Nacional Autónoma de México (UNAM), the Programa de Apoyo a Proyectos de Investigación e Innovación Tecnológica (PAPIIT) grant number IA203718, UNAM and the Consejo Nacional de Ciencia y Tecnología (CONACyT) grant number 282785. We also thank the program Nuevas Alternativas para el Tratamiento de Enfermedades Infecciosas NUATEI-UNAM for funding. We thank the Foundation for Applied Medical Research, University of Navarra (Pamplona, Spain), as well as Fundación Fuentes Dutor, for financial support.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by EL-L and OR. The first draft of the manuscript was written by EL-L and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
López-López, E., Rabal, O., Oyarzabal, J. et al. Towards the understanding of the activity of G9a inhibitors: an activity landscape and molecular modeling approach. J Comput Aided Mol Des 34, 659–669 (2020). https://doi.org/10.1007/s10822-020-00298-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10822-020-00298-x