Abstract
Purpose
One of the most common causes of infertility in adult women is polycystic ovary syndrome (PCOS) which has been identified with symptoms such as chronic hyperandrogenism, anovulation, and polycystic ovaries. Adiponectin modulates steroidogenesis and the expression of ovulation-related genes. Herein, we assessed the effect of AdipoRon (adiponectin agonist) in the PCOS model mice.
Methods
The PCOS model was induced with letrozole in the adult female mice and the animals received intraperitoneal injection of AdipoRon (5 mg/kg) for 10 days. Expression of CYP11A, CYP17A, and CYP19A genes, StAR protein, and histomorphology of the ovary were evaluated using real-time RT-PCR, western blotting, and histochemistry methods, respectively.
Results
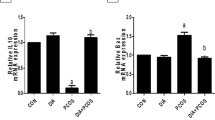
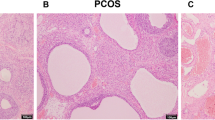
Although administration of letrozole caused an increase in the expression of CYP11A, CYP17A, and StAR and a decrease in the CYP19A1 expression, injection of AdipoRon reversed these changes. Moreover, AdipoRon treatment resulted in an improvement of folliculogenesis and a reduction of cysts compared to the letrozole-treated mice.
Conclusion
It is likely that AdipoRon has protective effects on the PCOS through modulation of cytochrome P450-related genes and steroidogenesis but needs further study to be sure.







Similar content being viewed by others
Data availability
The data presented in this study are available on request from the corresponding author.
References
Franks S. Polycystic ovary syndrome. N Engl J Med. 1995;333(13):853–61.
Himelein MJ, Thatcher SS. Polycystic ovary syndrome and mental health: a review. Obstet Gynecol Surv. 2006;61(11):723–32.
Goodarzi MO, et al. Polycystic ovary syndrome: etiology, pathogenesis and diagnosis. Nat Rev Endocrinol. 2011;7(4):219–31.
Rodriguez Paris V, Bertoldo MJ. The mechanism of androgen actions in PCOS etiology. Med Sci. 2019;7(9):89.
Diamanti-Kandarakis E, Dunaif A. Insulin resistance and the polycystic ovary syndrome revisited: an update on mechanisms and implications. Endocr Rev. 2012;33(6):981–1030.
Dewailly D, et al. Role of anti-Müllerian hormone in the pathogenesis of polycystic ovary syndrome. Front Endocrinol. 2020;11:641.
Zeng X, et al. Polycystic ovarian syndrome: correlation between hyperandrogenism, insulin resistance and obesity. Clin Chim Acta. 2020;502:214–21.
Rosenfield RL, Ehrmann DA. The pathogenesis of polycystic ovary syndrome (PCOS): the hypothesis of PCOS as functional ovarian hyperandrogenism revisited. Endocr Rev. 2016;37(5):467–520.
Wawrzkiewicz-Jałowiecka A, et al. In search of new therapeutics—molecular aspects of the PCOS pathophysiology: genetics, hormones, metabolism and beyond. Int J Mol Sci. 2020;21(19):7054.
Aghaie F, et al. The effects of exercise on expression of CYP19 and StAR mRNA in steroid-induced polycystic ovaries of female rats. Int J Fertil Steril. 2018;11(4):298.
Esfahani M, Shabab N, Saidijam M. AdipoRon may be benefit for atherosclerosis prevention Iran. J Basic Med Sci. 2017;20(2):107.
Okada-Iwabu M, et al. A small-molecule AdipoR agonist for type 2 diabetes and short life in obesity. Nature. 2013;503(7477):493–9.
Galluzzo A, Amato MC, Giordano C. Insulin resistance and polycystic ovary syndrome. Nutr Metab Cardiovasc Dis. 2008;18(7):511–8.
Maliqueo M, et al. Continuous administration of a P450 aromatase inhibitor induces polycystic ovary syndrome with a metabolic and endocrine phenotype in female rats at adult age. Endocrinology. 2013;154(1):434–45.
Kafali H, et al. Letrozole-induced polycystic ovaries in the rat: a new model for cystic ovarian disease. Arch Med Res. 2004;35(2):103–8.
Monsef F, et al. Effects of adipose-derived stromal vascular fraction on asherman syndrome model. Acta Histochem. 2020;122(5):151556.
GohariTaban S, et al. Abnormal expressions of ADAMTS-1, ADAMTS-9 and progesterone receptors are associated with lower oocyte maturation in women with polycystic ovary syndrome. Arch Gynecol Obstet. 2019;299(1):277–86.
Azouz AA, et al. Modulation of steroidogenesis by Actaea racemosa and vitamin C combination, in letrozole induced polycystic ovarian syndrome rat model: promising activity without the risk of hepatic adverse effect. Chin Med. 2021;16(1):1–17.
Abtahi-Eivari SH, et al. The effect of Galega officinalis on hormonal and metabolic profile in a rat model of polycystic ovary syndrome. Int J Women's Health Reprod Sci. 2018;6(3):276–82.
Brennan K, Huang A, Azziz R. Dehydroepiandrosterone sulfate and insulin resistance in patients with polycystic ovary syndrome. Fertil Steril. 2009;91(5):1848–52.
Goodarzi MO, Carmina E, Azziz R. DHEA, DHEAS and PCOS. J Steroid Biochem Mol Biol. 2015;145:213–25.
Guo C, et al. Transcriptional regulation of human CYP11A1 in gonads and adrenals. J Biomed Sci. 2007;14(4):509–15.
Franks S, et al. The genetic basis of polycystic ovary syndrome. Hum Reprod. 1997;12(12):2641–8.
Qin K-N, Rosenfield RL. Role of cytochrome P450c17 in polycystic ovary syndrome. Mol Cell Endocrinol. 1998;145(1-2):111–21.
Shen W, et al. Common polymorphisms in the CYP1A1 and CYP11A1 genes and polycystic ovary syndrome risk: a meta-analysis and meta-regression. Arch Gynecol Obstet. 2014;289(1):107–18.
Xu X, et al. The single nucleotide polymorphism rs743572 of CYP17A1 shows significant association with polycystic ovary syndrome: a meta-analysis. Reprod Biomed Online. 2021;43(5):941–51.
Munawar Lone N, et al. Association of the CYP17 and CYP19 gene polymorphisms in women with polycystic ovary syndrome from Punjab, Pakistan. Gynecol Endocrinol. 2021;37(5):456–61.
Marshall JC, Eagleson CA. Neuroendocrine aspects of polycystic ovary syndrome. Endocrinol Metab Clin N Am. 1999;28(2):295–324.
Wickenheisser JK, et al. Differential activity of the cytochrome P450 17α-hydroxylase and steroidogenic acute regulatory protein gene promoters in normal and polycystic ovary syndrome theca cells. J Clin Endocrinol Metabol. 2000;85(6):2304–11.
Wu S, et al. Obesity-induced infertility and hyperandrogenism are corrected by deletion of the insulin receptor in the ovarian theca cell. Diabetes. 2014;63(4):1270–82.
Jakimiuk AJ, et al. Luteinizing hormone receptor, steroidogenesis acute regulatory protein, and steroidogenic enzyme messenger ribonucleic acids are overexpressed in thecal and granulosa cells from polycystic ovaries. J Clin Endocrinol Metabol. 2001;86(3):1318–23.
Kosova G, Urbanek M. Genetics of the polycystic ovary syndrome. Mol Cell Endocrinol. 2013;373(1-2):29–38.
Panghiyangani R, et al. CYP19A1 gene expression in patients with polycystic ovarian syndrome. J Hum Reprod Sci. 2020;13(2):100.
Erickson GF. Normal regulation of ovarian androgen production. in Seminars in reproductive endocrinology. Copyright© 1993 by Thieme Medical Publishers, Inc; 1993.
Guet P, et al. Aromatase activity of human granulosa cells in vitro: effects of gonadotrophins and follicular fluid. Hum Reprod. 1999;14(5):1182–9.
Kirilovas D, et al. Granulosa cell aromatase enzyme activity: effects of follicular fluid from patients with polycystic ovary syndrome, using aromatase conversion and [11C] vorozole-binding assays. Gynecol Endocrinol. 2006;22(12):685–91.
Hogg K, et al. Enhanced thecal androgen production is prenatally programmed in an ovine model of polycystic ovary syndrome. Endocrinology. 2012;153(1):450–61.
Sanders SL, Stouffer RL. Localization of steroidogenic enzymes in macaque luteal tissue during the menstrual cycle and simulated early pregnancy: immunohistochemical evidence supporting the two-cell model for estrogen production in the primate corpus luteum. Biol Reprod. 1997;56(5):1077–87.
Kahsar-Miller MD, et al. Steroidogenic acute regulatory protein (StAR) in the ovaries of healthy women and those with polycystic ovary syndrome. Am J Obstet Gynecol. 2001;185(6):1381–7.
Waterman MR. A rising StAR: an essential role in cholesterol transport. Science. 1995;267(5205):1780–2.
Smolinska N, et al. Effect of adiponectin on the steroidogenic acute regulatory protein, P450 side chain cleavage enzyme and 3β-hydroxysteroid dehydrogenase gene expression, progesterone and androstenedione production by the porcine uterus during early pregnancy. J Physiol Pharmacol. 2016;67:443–56.
Comim FV, et al. Disorders of follicle development and steroidogenesis in ovaries of androgenised foetal sheep. J Endocrinol. 2015;225(1):39–46.
Li P, et al. Expression of adiponectin receptors in mouse adrenal glands and the adrenocortical Y-1 cell line: adiponectin regulates steroidogenesis. Biochem Biophys Res Commun. 2009;390(4):1208–13.
Comim FV, et al. Effects of adiponectin including reduction of androstenedione secretion and ovarian oxidative stress parameters in vivo. PLoS One. 2016;11(5):e0154453.
Sepilian V, Nagamani M. Adiponectin levels in women with polycystic ovary syndrome and severe insulin resistance. J Soc Gynecol Investig. 2005;12(2):129–34.
Funding
This study was supported by the Hamadan University of Medical Sciences (9906254309).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hazrati, P., Ramezani, M., Ahmadimoghaddam, D. et al. The effects of AdipoRon on cytochrome P450-related gene expression, acute steroidogenic regulatory protein, and structure of ovary in polycystic ovary syndrome model. J Assist Reprod Genet 40, 2453–2461 (2023). https://doi.org/10.1007/s10815-023-02900-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-023-02900-1