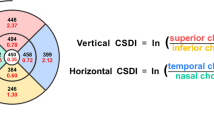
Abstract
Introduction
Image processing of optical coherence tomography scans through binarization techniques represent a non-invasive way to separately asses and measure choroidal components, in vivo. In this review, we systematically search the scientific literature regarding binarization studies published so far.
Methods
A systematic research was conducted at PubMed database, including English literature articles for all of the following terms in various combinations: binarization, choroid/al, enhanced depth spectral domain/swept source optic coherence tomography, and latest publications up to November 2018 were reviewed.
Results
Thirty-seven articles were included and analyzed regarding studied disease, binarization method, studied variables, and outcomes. Most of the studies have focused on the more common retinal pathologies, such as age-related macular degeneration, central serous chorioretinopathy and diabetic retinopathy but binarization techniques have also been applied to the study of choroidal characteristics in ocular inflammatory diseases, corneal dystrophies and in postsurgical follow-up. Advantages and disadvantages of binarization techniques are also discussed.
Conclusion
Binarization of choroidal images seems to represent a promising approach to study choroid subcomponents in an increasingly detailed manner.
Similar content being viewed by others
References
Nickla DL, Wallman J (2010) The multifunctional choroid. Prog Retin Eye Res 29:144–168. https://doi.org/10.1016/j.preteyeres.2009.12.002
Alm A, Bill A (1973) Ocular and optic nerve blood flow at normal and increased intraocular pressures in monkeys (Macaca irus): a study with radioactively labelled microspheres including flow determinations in brain and some other tissues. Exp Eye Res 15:15–29
Kur J, Newman EA, Chan-ling T (2012) Cellular and physiological mechanisms underlying blood flow regulation in the retina and choroid in health and disease. Prog Retin Eye Res 31:377–406. https://doi.org/10.1016/j.preteyeres.2012.04.004
Castro-Correia J (1995) Understanding the choroid. Int Ophthalmol 19:135–147. https://doi.org/10.1007/BF00133730
Imamura Y, Fujiwara T, Margolis RON, Spaide RF (2009) Enhanced depth imaging optical coherence tomography of the choroid in central serous chorioretinopathy. Retina 29:1469–1473
Chung SE, Kang SW, Lee JH, Kim YT (2011) Choroidal thickness in polypoidal choroidal vasculopathy and exudative age-related macular degeneration. Ophthalmology 118:840–845. https://doi.org/10.1016/j.ophtha.2010.09.012
Fujiwara T, Imamura Y, Margolis R et al (2009) Enhanced depth imaging optical coherence tomography of the choroid in highly myopic eyes. Am J Ophthalmol 148:445–450. https://doi.org/10.1016/j.ajo.2009.04.029
Gomi F, Tano Y (2008) Polypoidal choroidal vasculopathy and treatments. Curr Opin Ophthalmol 19:208–212. https://doi.org/10.1097/ICU.0b013e3282fb7c33
Koizumi H, Yamagishi T, Yamazaki T (2011) Subfoveal choroidal thickness in typical age-related macular degeneration and polypoidal choroidal vasculopathy. Graefe’s Arch Clin Exp Ophthalmol 249:1123–1128. https://doi.org/10.1007/s00417-011-1620-1
Grossniklaus HE, Green WR (2004) Choroidal neovascularization. Am J Ophthalmol 137:496–503
Spaide RF, Koizumi H, Pozzoni MC, Pozonni MC (2008) Enhanced depth imaging spectral-domain optical coherence tomography. Am J Ophthalmol 146:496–500. https://doi.org/10.1016/j.ajo.2008.05.032
Spraul CW, Lang GE, Lang GK, Grossniklaus HE (2002) Morphometric changes of the choriocapillaris and the choroidal vasculature in eyes with advanced glaucomatous changes. Vis Res 42:923–932. https://doi.org/10.1016/S0042-6989(02)00022-6
Fryczkowski AW (1994) Anatomical and functional choroidal lobuli. Int Ophthalmol 18:131–141
Hidayat AA, Fine BS (1985) Diabetic choroidopathy: light and electron microscopic observations of seven cases. Ophthalmology 92:512–522. https://doi.org/10.1016/S0161-6420(85)34013-7
Sonoda S, Sakamoto T, Yamashita T et al (2014) Choroidal structure in normal eyes and after photodynamic therapy determined by binarization of optical coherence tomographic images. Invest Ophthalmol Vis Sci 55:3893–3898. https://doi.org/10.1167/iovs.14-14447
Bernsen J (1986) Dynamic thresholding of grey-level images. In: Proceedings of the international conference on pattern recognition, pp 1251–1255
Niblack W (1986) An introduction to digital image processing, vol 34. Prentice-Hall, Englewood Cliffs
Agrawal R, Gupta P, Tan KA et al (2016) Choroidal vascularity index as a measure of vascular status of the choroid: measurements in healthy eyes from a population-based study. Nature 6:1–9. https://doi.org/10.1038/srep21090
Agrawal R, Salman M, Tan KA et al (2016) Choroidal vascularity index (CVI)—a novel optical coherence tomography parameter for monitoring patients with panuveitis? PLoS ONE 11:e0146344
Kuroda Y, Ooto S, Yamashiro K et al (2016) Increased choroidal vascularity in central serous chorioretinopathy quantified using swept-source optical coherence tomography. Am J Ophthalmol 169:199–207. https://doi.org/10.1016/j.ajo.2016.06.043
Wang JC, Laíns I, Providência J et al (2017) Diabetic choroidopathy: choroidal vascular density and volume in diabetic retinopathy with swept-source optical coherence tomography. Am J Ophthalmol 184:75–83. https://doi.org/10.1016/j.ajo.2017.09.030
Zheng F, Gregori G, Schaal KB et al (2018) Choroidal thickness and choroidal vessel density in nonexudative age-related macular degeneration using swept-source optical coherence tomography imaging. Invest Ophthalmol Vis Sci. https://doi.org/10.1167/iovs.16-20161
Fujiwara A, Morizane Y, Hosokawa M et al (2016) Factors affecting choroidal vascular density in normal eyes: quantification using en face swept-source optical coherence tomography. Am J Ophthalmol 170:1–9. https://doi.org/10.1016/j.ajo.2016.07.006
Sonoda S, Sakamoto T, Kakiuchi N et al (2017) Semi-automated software to measure luminal and stromal areas of choroid in optical coherence tomographic images. Jpn J Ophthalmol. https://doi.org/10.1007/s10384-017-0558-1
Vupparaboina KK, Richhariya A, Chhablani J, Jana S (2017) Optical coherence tomography imaging: automated binarization of choroid for stromal-luminal analysis. In: 2016 International conference on signal and information processing, IConSIP 2016
Mahajan NR, Donapati RCR, Channappayya SS et al (2013) An automated algorithm for blood vessel count and area measurement in 2-D choroidal scan images. In: Proceedings of the annual international conference of the IEEE Engineering in Medicine and Biology Society, EMBS, pp 3355–3358
Uppugunduri SR, Rasheed MA, Richhariya A et al (2018) Automated quantification of Haller’ s layer in choroid using swept-source optical coherence tomography. PLoS ONE. https://doi.org/10.1371/journal.pone.0193324
Sonoda S, Sakamoto T, Yamashita T et al (2015) Luminal and stromal areas of choroid determined by binarization method of optical coherence tomographic images. Am J Ophthalmol 159:1123–1131 (e1)
Gupta P, Jing T, Marziliano P et al (2015) Distribution and determinants of choroidal thickness and volume using automated segmentation software in a population-based study. Am J Ophthalmol 159:293–301 (e3)
Sansom LT, Suter CA, McKibbin M (2016) The association between systolic blood pressure, ocular perfusion pressure and subfoveal choroidal thickness in normal individuals. Acta Ophthalmol 94:e157–e158. https://doi.org/10.1111/aos.12794
Wei X, Ting DSW, Ng WY et al (2016) Choroidal vascularity index—a novel optical coherence tomography based parameter in patients with exudative age-related macular degeneration. Retina 37(6):1120–1125
Bakthavatsalam M, Ng DSC, Lai FHP et al (2017) Choroidal structures in polypoidal choroidal vasculopathy, neovascular age-related maculopathy, and healthy eyes determined by binarization of swept source optical coherence tomographic images. Graefe’s Arch Clin Exp Ophthalmol 255:935–943. https://doi.org/10.1007/s00417-017-3591-3
Ruiz-Medrano J, Flores-Moreno I, Peña-García P et al (2014) Macular choroidal thickness profile in a healthy population measured by swept-source optical coherence tomography. Invest Ophthalmol Vis Sci 55:3532–3542. https://doi.org/10.1167/iovs.14-13868
Koh LHL, Agrawal R, Khandelwal N et al (2017) Choroidal vascular changes in age-related macular degeneration. Acta Ophthalmol 95:e597–e601. https://doi.org/10.1111/aos.13399
Ozkaya A, Alagoz C, Garip R et al (2016) The role of indocyanine green angiography imaging in further differential diagnosis of patients with nAMD who are morphologically poor responders to ranibizumab in a real-life setting. Eye 30:958–965. https://doi.org/10.1038/eye.2016.71
Cho M, Barbazetto IA, Freund KB (2009) Refractory neovascular age-related macular degeneration secondary to polypoidal choroidal vasculopathy. Am J Ophthalmol 148:70–78. https://doi.org/10.1016/j.ajo.2009.02.012(e1)
Ma L, Li Z, Liu K et al (2015) Association of genetic variants with polypoidal choroidal vasculopathy: a systematic review and updated meta-analysis. Ophthalmology 122:1854–1865. https://doi.org/10.1016/j.ophtha.2015.05.012
Ming C, Cheung G, Yang E et al (2015) The natural history of polypoidal choroidal vasculopathy: a multi-center series of untreated Asian patients. Graefe’s Arch Clin Exp Ophthalmol. https://doi.org/10.1007/s00417-015-2933-2
Jirarattanasopa P, Ooto S, Nakata I et al (2016) Complement factor H in age-related macular degeneration and polypoidal choroidal vasculopathy. Science. https://doi.org/10.1167/iovs.12-9619
Manjunath V, Goren J, Fujimoto JG, Duker JS (2011) Analysis of choroidal thickness in age-related macular degeneration using spectral-domain optical coherence tomography. Am J Ophthalmol 152:663–668. https://doi.org/10.1016/j.ajo.2011.03.008
Jonas JB, Forster TM, Steinmetz P et al (2014) Choroidal thickness in age-related macular degeneration. Retina 34:1149–1155. https://doi.org/10.1097/iae.0000000000000035
Warrow DJ, Hoang QV, Freund KB (2013) Pachychoroid pigment epitheliopathy. Retina 33:1659–1672. https://doi.org/10.1097/IAE.0b013e3182953df4
Pang CE, Freund KB (2015) Pachychoroid neovasculopathy. Retina 35:1–9. https://doi.org/10.1097/IAE.0000000000000331
Gallego-Pinazo R, Dolz-Marco R, Gómez-Ulla F et al (2014) Pachychoroid diseases of the macula. Med Hypothesis Discov Innov Ophthalmol 3:111–115
Dansingani KK, Balaratnasingam C, Naysan J, Freund KB (2015) En face imaging of pachychoroid spectrum disorders with swept-source optical coherence tomography. Retina 1:499–516. https://doi.org/10.1097/IAE.0000000000000742
Balaratnasingam C, Lee WK, Koizumi H et al (2016) Polypoidal choroidal vasculopathy a distinct disease or manifestation of many? Retina 36:1–8
Daizumoto E, Mitamura Y, Sano H et al (2017) Changes of choroidal structure after intravitreal aflibercept therapy for polypoidal choroidal vasculopathy. Br J Ophthalmol 101:56–61. https://doi.org/10.1136/bjophthalmol-2016-309694
Ng DS, Bakthavatsalam M, Lai FH-P et al (2017) Classification of exudative age-related macular degeneration with pachyvessels on en face swept-source optical coherence tomography. Invest Opthalmol Vis Sci 58:1054. https://doi.org/10.1167/iovs.16-20519
Gupta P, Shu D, Ting WEI et al (2017) Detailed characterization of choroidal morphologic and vascular features in age-related macular degeneration and polypoidal choroidal. Retina 37:2269–2280
Ting DSW, Yanagi Y, Agrawal R et al (2017) Choroidal remodeling in age-related macular degeneration and polypoidal choroidal vasculopathy: a 12-month Prospective study. Sci Rep 7:7868. https://doi.org/10.1038/s41598-017-08276-4
Masuda N, Kojima M, Yamashita M et al (2017) Choroidal structure determined by binarizing optical coherence tomography images in eyes with reticular pseudodrusen. Clin Ophthalmol 11:791–795. https://doi.org/10.2147/OPTH.S135160
Donald J, Gass MMD (1967) Pathogenisis of disciform detachment. Am J Ophthalmol 63:573/1–585/13. https://doi.org/10.1016/0002-9394(67)90026-8
Spaide RF, Campeas L, Haas A et al (1996) Central serous chorioretinopathy in younger and older adults. Ophthalmology 103:2070–2080. https://doi.org/10.1016/S0161-6420(96)30386-2
Gemenetzi M, De Salvo G, Lotery AJ (2010) Central serous chorioretinopathy: an update on pathogenesis and treatment. Eye 24:1743–1756. https://doi.org/10.1038/eye.2010.130
Nicholson B, Noble J, Forooghian F, Meyerle C (2013) MAJOR REVIEW central serous chorioretinopathy: update on pathophysiology and treatment. Surv Ophthalmol 58:103–126. https://doi.org/10.1016/j.survophthal.2012.07.004
Agrawal R, Chhablani J, Tan KA et al (2016) Choroidal vascularity index in central serous chorioretinopathy. Retina 36:1646–1651. https://doi.org/10.1097/IAE.0000000000001040
Sonoda S, Sakamoto T, Kuroiwa N et al (2016) Structural changes of inner and outer choroid in central serous chorioretinopathy determined by optical coherence tomography. PLoS ONE 11:1–16. https://doi.org/10.1371/journal.pone.0157190
Branchini LA, Adhi M, Regatieri CV et al (2013) Analysis of choroidal morphologic features and vasculature in healthy eyes using spectral-domain optical coherence tomography. Ophthalmology 120:1901–1908
Kinoshita T, Mitamura Y, Mori T et al (2016) Changes in choroidal structures in eyes with chronic central serous chorioretinopathy after half-dose photodynamic therapy. PLoS ONE 11:1–15. https://doi.org/10.1371/journal.pone.0163104
Wei WB, Xu L, Jonas JB et al (2012) Subfoveal choroidal thickness: the beijing eye study. Ophthalmology 120:175–180
Ferreira J, Vicente A, Anjos R et al (2015) Choroidal thickness in diabetic patients without retinopathy. Invest Ophthalmol Vis Sci 56:4678
Tavares Ferreira J, Proença R, Alves M et al (2017) Retina and choroid of diabetic patients without observed retinal vascular changes: a Longitudinal Study. Am J Ophthalmol 176:15–25
Esmaeelpour M, Povaz B, Hermann B et al (2018) Three-dimensional 1060-nm OCT: choroidal thickness maps in normal subjects and improved posterior segment visualization in cataract patients. Invest Ophthalmol Vis Sci 51:5260–5266. https://doi.org/10.1167/iovs.10-5196
Querques G, Lattanzio R, Querques L et al (2012) Enhanced depth imaging optical coherence tomography in type 2 diabetes. Invest Ophthalmol Vis Sci 53:6017–6024
Kim JT, Lee DH, Joe SG et al (2013) Changes in choroidal thickness in relation to the severity of retinopathy and macular edema in type 2 diabetic patients. Invest Ophthalmol Vis Sci 54:3378–3384. https://doi.org/10.1167/iovs.12-11503
Lee HK, Lim JW, Shin MC (2013) Comparison of choroidal thickness in patients with diabetes by spectral-domain optical coherence tomography. Korean J Ophthalmol 27:433–439. https://doi.org/10.3341/kjo.2013.27.6.433
Farias LB, Lavinsky D, Schneider WM et al (2014) Choroidal thickness in patients with diabetes and microalbuminuria. Ophthalmology 121:2071–2073. https://doi.org/10.1016/j.ophtha.2014.04.038
Gerendas BS, Waldstein SM, Simader C et al (2014) Three-dimensional automated choroidal volume assessment on standard spectral-domain optical coherence tomography and correlation with the level of diabetic macular edema. Am J Ophthalmol 158:1039–1048. https://doi.org/10.1016/j.ajo.2014.08.001
Unsal E, Eltutar K, Zirtiloğlu S et al (2014) Choroidal thickness in patients with diabetic retinopathy. Clin Ophthalmol 8:637–642. https://doi.org/10.2147/OPTH.S59395
Tan K, Laude A, Yip V et al (2016) Choroidal vascularity index—a novel optical coherence tomography parameter for disease monitoring in diabetes mellitus? Acta Ophthalmol 94:e612–e616
Kim M, Ha MJ, Choi SY, Park Y (2018) Choroidal vascularity index in type-2 diabetes analyzed by swept-source optical coherence tomography. Sci Rep. https://doi.org/10.1038/s41598-017-18511-7
Rao NA, Gupta A, Dustin L et al (2010) Frequency of distinguishing clinical features in Vogt–Koyanagi–Harada disease. Ophthalmology 117:591–599. https://doi.org/10.1016/j.ophtha.2009.08.030(e1)
Read RW, Holland GN, Rao NA et al (2001) Revised diagnostic criteria for Vogt–Koyanagi–Harada disease: report of an international committee on nomenclature. Am J Ophthalmol 131:647–652
Moorthy RS, Inomata H, Rao NA (1995) MAJOR REVIEW: Vogt–Koyanagi–Harada Syndrome. Surv Ophthalmol 39(4):265–292
Maruko I, Iida T, Sugano Y et al (2011) Subfoveal choroidal thickness after treatment of Vogt–Koyanagi–Harada disease. Retina 31:510–517. https://doi.org/10.1097/IAE.0b013e3181eef053
Fong AH, Li KK, Wong D (2011) CHOROIDAL evaluation using enhanced depth imaging spectral-domain optical coherence tomography in Vogt–Koyanagi–Harada disease. Retina 31:502–509. https://doi.org/10.1097/IAE.0b013e3182083beb
Nakai K, Gomi F, Ikuno Y et al (2012) Choroidal observations in Vogt–Koyanagi–Harada disease using high-penetration optical coherence tomography. Graefe’s Arch Clin Exp Ophthalmol 250:1089–1095. https://doi.org/10.1007/s00417-011-1910-7
Agrawal R, Li LKH, Nakhate V et al (2016) Choroidal Vascularity Index in Vogt–Koyanagi–Harada disease: an EDI-OCT derived tool for monitoring disease progression. Transl Vis Sci Technol 5:7. https://doi.org/10.1167/tvst.5.4.7
Jaisankar D, Raman R, Sharma HR et al (2017) Choroidal and retinal anatomical responses following systemic corticosteroid therapy in Vogt–Koyanagi–Harada disease using swept-source optical coherence tomography. Ocul Immunol Inflamm 12:1–9
Liu S, Du L, Zhou Q et al (2017) The Choroidal Vascularity Index decreases and choroidal thickness increases in Vogt–Koyanagi–Harada disease patients during a recurrent anterior uveitis attack. Ocul Immunol Inflamm 3948:1–7
Kawano H, Sonoda S, Yamashita T, Maruko I (2016) Relative changes in luminal and stromal areas of choroid determined by binarization of EDI-OCT images in eyes with Vogt–Koyanagi–Harada disease after treatment. Graefe’s Arch Clin Exp Ophthalmol 254:421–426. https://doi.org/10.1007/s00417-016-3283-4
Onal S, Herbort CP, Akbay AKOC (2018) Quantitative analysis of structural alterations in the choroid of patients with active Behcet uveitis. Retina 38:828–840. https://doi.org/10.1097/IAE.0000000000001587
Agarwal A, Agrawal R, Khandelwal N et al (2017) Choroidal structural changes in tubercular multifocal serpiginoid choroiditis. Ocul Immunol Inflamm 26(6):838–844
Wong TY, Ferreira A, Hughes R et al (2014) Epidemiology and disease burden of pathologic myopia and myopic choroidal neovascularization: an evidence-based systematic review. Am J Ophthalmol 157:9–25. https://doi.org/10.1016/j.ajo.2013.08.010(e12)
Saw SM, Gazzard G, Shin-Yen EC, Chua WH (2005) Myopia and associated pathological complications. Ophthalmic Physiol Opt 25:381–391
Neelam K, Ming C, Cheung G et al (2012) Progress in retinal and eye research choroidal neovascularization in pathological myopia. Prog Retin Eye Res 31:495–525. https://doi.org/10.1016/j.preteyeres.2012.04.001
Flores-Moreno I, Lugo F, Duker JS, Ruiz-Moreno JM (2013) The relationship between axial length and choroidal thickness in eyes with high myopia. Am J Ophthalmol 155:314–319. https://doi.org/10.1016/j.ajo.2012.07.015
Nishida Y, Fujiwara T, Imamura Y et al (2012) Choroidal thickness and visual acuity in highly myopic eyes. Retina 32:1229–1236
Gupta P, Thakku SG, Saw SM et al (2017) Characterization of choroidal morphologic and vascular features in young men with high myopia using spectral-domain optical coherence tomography. Am J Ophthalmol 177:27–33. https://doi.org/10.1016/j.ajo.2017.02.001
Alshareef RA, Khuthaila MK, Goud A et al (2016) Subfoveal choroidal vascularity in myopia: evidence from spectral-domain optical coherence tomography. Ophthalmic Surg Lasers Imaging Retina. https://doi.org/10.3928/23258160-20170301-02
Ng WY, Ting DSW, Agrawal R et al (2016) Choroidal structural changes in myopic choroidal neovascularization after treatment with antivascular endothelial growth factor over 1 year. Invest Opthalmol Vis Sci 57:4933. https://doi.org/10.1167/iovs.16-20191
Ratra D, Tan ROY, Khandelwal N, Agrawal R (2017) Choroidal structural changes and vascularity index in stargardt disease on swept source optical coherence tomography. Retina. https://doi.org/10.1097/IAE.0000000000001879
Birnbach CD, Järveläínen M, Possin DE, Milam AH (1994) Histopathology and immunocytochemistry of the neurosensory retina in fundus flavimaculatus. Ophthalmology 101:1211–1219. https://doi.org/10.1016/S0161-6420(13)31725-4
Lopez PF, Maumenee IH, De Cruz Z, Green WR (1990) Autosomal-dominant fundus flavimaculatus clinicopathologic correlation. Ophthalmology 97:798–809. https://doi.org/10.1016/S0161-6420(90)32508-3
Hirashima T, Miyata M, Ishihara K et al (2017) Choroidal vasculature in bietti crystalline dystrophy with CYP4V2 mutations and in retinitis pigmentosa with EYS mutations. Invest Ophthalmol Vis Sci 58:3871–3878. https://doi.org/10.1167/iovs.17-21515
Agrawal R, Wei X, Goud A et al (2017) Influence of scanning area on choroidal vascularity index measurement using optical coherence tomography. Acta Ophthalmol 95(8):e770–e775
Spaide RF, Ryan EH (2015) Loculation of fluid in the posterior choroid in eyes with central serous chorioretinopathy. Am J Ophthalmol 160:1211–1216. https://doi.org/10.1016/j.ajo.2015.08.018
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Crisostomo, S., Cardigos, J., Fernandes, D.H. et al. Choroidal binarization analysis: clinical application. Int Ophthalmol 39, 2947–2973 (2019). https://doi.org/10.1007/s10792-019-01122-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-019-01122-8