Abstract
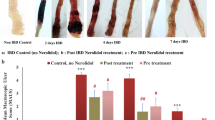
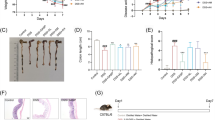
Jasonia glutinosa (L.) DC., known as rock tea (RT), is traditionally used in Spain as a digestive due to its beneficial properties in bowel disorders. The pharmacological nature of these properties has not been established yet. The aim of this work was to evaluate the therapeutic utility of RT in experimental colitis and to identify chemical constituents with anti-inflammatory and/or anti-oxidative properties. RT extract was prepared with ethanol in a Soxhlet apparatus and analysed by HPLC–DAD. Superoxide radical scavenging properties, xanthine oxidase and lipoxygenase (5-LOX) inhibitory activity, and capability to lower nitric oxide (NO) and tumor necrosis factor α (TNF-α) levels were measured in cell-free and cell-based assays. In the 2.5%-dextran-sodium sulphate (DSS) injury-repair model of ulcerative colitis (UC), mice were daily treated with sulfasalazine (SSZ, as reference drug, 100 mg/kg bw), RT (5, 25 and 50 mg/kg bw, p.o.), or vehicle over 20 days. Colitis was scored daily. Colon samples were examined macroscopically and histopathologically. Protein levels of myeloperoxidase (MPO), interleukins 6, and 10 (IL-6, IL-10), inducible NO synthase (iNOS), and cyclooxygenase-2 (COX-2) were studied as markers of oxidative stress and inflammatory activity. The integrity of the apical epithelial layer was assessed by immunofluorescence staining of zonula ocludens-1 (ZO-1). Finally, intestinal contractility was also evaluated by isometric myography. Fifteen phenolic compounds and three pigments were identified and quantified, of which caffeoylquinic acids, and the flavonoid, quercetin-3-O-galactoside, were the most abundant. RT extract significantly scavenged superoxide radicals, inhibited 5-LOX activity, and lowered NO and TNF-α levels. DSS-treated mice receiving RT scored clinically lower than controls during the first 3 days of DSS treatment and during the recovery period. SSZ was less effective than RT. Anatomical and histological examination of colon samples revealed that RT significantly prevented colon shortening, increased colon thickness, and lowered the macroscopic damage score. RT also significantly prevented the increase of MPO activity, IL-6 levels, iNOS and COX-2 expression, the loss of ZO-1 apical expression, and normalized contractility disturbances. In conclusion, daily administration of RT showed therapeutic properties in the DSS-model of UC. The benefits of RT can likely be attributed to its anti-inflammatory and antioxidant phenolic and flavonoid constituents.







Similar content being viewed by others
Abbreviations
- ACh:
-
Acetylcholine
- AUC:
-
Area under the curve
- COX-2:
-
Cyclooxygenase-2
- CV:
-
Coefficient of variation
- DAI:
-
Disease activity index,
- DMSO:
-
Dimethyl sulfoxide
- DSS:
-
Dextran-sodium sulphate
- HE:
-
Haematoxylin–eosin
- HPLC–DAD:
-
High-pressure liquid chromatography coupled to a diode-array detector
- iNOS:
-
Inducible nitric oxide synthase
- IBD:
-
Inflammatory bowel disease
- IL-6:
-
Interleukin 6
- IL-10:
-
Interleukin 10
- LOD:
-
Limit of detection
- LOQ:
-
Limit of quantification
- 5-LOX:
-
5-Lipoxygenase
- LPS:
-
Lipopolysaccharide
- MPO:
-
Myeloperoxidase
- NADH/PMS:
-
Β-Nicotinamide adenine dinucleotide/phenazine methosulfate
- NBT:
-
Nitrotetrazolium blue chloride
- NO:
-
Nitric oxide
- \({\text{O}}_{2}^{ \cdot - }\) :
-
Superoxide radical
- RT:
-
Rock tea
- SSZ:
-
Sulfasalazine
- TNF-α:
-
Tumor necrosis factor-alpha
- UC:
-
Ulcerative colitis
- X/XO:
-
Xanthine/xanthine oxidase
- ZO-1:
-
Zonula occludens-1
References
Akerreta S, Cavero RY, López V, Calvo MI (2007) Analyzing factors that influence the folk use and phytonomy of 18 medicinal plants in Navarra. J Ethnobiol Ethnomed 3:16. https://doi.org/10.1186/1746-4269-3-16
Alarcόn R, Pardo-de-Santayana M, Priestley C, Morales R, Heinrich M (2015) Medicinal and local food plants in the south of Alava (Basque Country, Spain). J Ethnopharmacol 176:207–224. https://doi.org/10.1016/j.jep.2015.10.022
Amaro H et al (2015) Effect of solvent system on extractability of lipidic components of Scenedesmus obliquus (M2-1) and Gloeothece sp. on antioxidant scavenging capacity thereof. Mar Drugs 13(10):6453–6471. https://doi.org/10.3390/md13106453
Barbosa M et al (2017) Phlorotannin extracts from Fucales: marine polyphenols as bioregulators engaged in inflammation-related mediators and enzymes. Algal Res 28:1–8. https://doi.org/10.1016/J.ALGAL.2017.09.009
Baumgart DC, Carding SR (2007) Inflammatory bowel disease: cause and immunobiology. Lancet 369(9573):1627–1640. https://doi.org/10.1016/S0140-6736(07)60750-8
Baumgart DC, Sandborn WJ (2007) Inflammatory bowel disease: clinical aspects and established and evolving therapies. Lancet 369(9573):1641–1657. https://doi.org/10.1016/S0140-6736(07)60751-X
Bermejo BP, Abad MJ, Díaz AM, Villaescusa L, González MA, Silván AM (2002) Sesquiterpenes from Jasonia glutinosa: in vitro anti-inflammatory activity. Biol Pharm Bull 25(1):1–4
Brückner M, Westphal S, Domschke W, Kucharzik T, Lügering A (2012) Green tea polyphenol epigallocatechin-3-gallate shows therapeutic antioxidative effects in a murine model of colitis. J. Crohn’s Colitis 6(2):226–235. https://doi.org/10.1016/j.crohns.2011.08.012
Camuesco D et al (2004) The intestinal anti-inflammatory effect of quercitrin is associated with an inhibition in iNOS expression. Br J Pharmacol 143(7):908–918. https://doi.org/10.1038/sj.bjp.0705941
Capaldo CT, Nusrat A (2009) Cytokine regulation of tight junctions. Biochim Biophys Acta 1788(4):864–871. https://doi.org/10.1016/j.bbamem.2008.08.027
Castro M et al (2016) Spasmolytic effect of Jasonia glutinosa on rodent intestine. Rev esp enferm dig 108:785–789. https://doi.org/10.17235/reed.2016.4327/2016
Chaudhuri L et al (2000) Prokinetic effect of black tea on gastrointestinal motility. Life Sci 66(9):847–854
Comalada M et al (2005) In vivo quercitrin anti-inflammatory effect involves release of quercetin, which inhibits inflammation through down-regulation of the NF-κB pathway. Eur J Immunol 35(2):584–592. https://doi.org/10.1002/eji.200425778
Dai Y et al (2017) Jianpi Qingchang decoction regulates intestinal motility of dextran sulfate sodium-induced colitis through reducing autophagy of interstitial cells of Cajal. World J Gastroenterol 23(26):4724–4734. https://doi.org/10.3748/wjg.v23.i26.4724
Danese S, Fiocchi C (2011) Ulcerative colitis. N Engl J Med 365(18):1713–1725. https://doi.org/10.1056/NEJMra1102942
Desai TR, Leeper NJ, Hynes KL, Gewertz BL (2002) Interleukin-6 causes endothelial barrier dysfunction via the protein kinase C pathway. J Surg Res 104(2):118–123. https://doi.org/10.1006/jsre.2002.6415
Fagundes DS, Grasa L, Arruebo MP, Plaza MA, Murillo MD (2007) Ca2+-activated K+ channels involved in duodenal dismotility induced by ethanol. Alcohol Alcohol 42(4):291–295. https://doi.org/10.1093/alcalc/agm037
Głąbska D, Guzek D, Zakrzewska P, Włodarek D, Lech G (2016) Lycopene, Lutein and Zeaxanthin may reduce faecal blood, mucus and pus but not abdominal pain in individuals with ulcerative colitis. Nutrients 8(10):613. https://doi.org/10.3390/nu8100613
Głąbska D, Guzek D, Zakrzewska P, Lech G (2019) Intake of lutein and zeaxanthin as a possible factor influencing gastrointestinal symptoms in caucasian individuals with ulcerative colitis in remission phase. J Clin Med 8(1):77. https://doi.org/10.3390/jcm8010077
Hussain T, Tan B, Yin Y, Blachier F, Tossou MCB, Rahu N (2016) Oxidative stress and inflammation: what polyphenols can do for us? Oxid Med Cell Longev 2016:7432797. https://doi.org/10.1155/2016/7432797
Kang S et al (2017) Aronia berry extract ameliorates the severity of dextran sodium sulfate-induced ulcerative colitis in mice. J Med Food 20(7):667–675. https://doi.org/10.1089/jmf.2016.3822
Kaulmann A, Bohn T (2014) Carotenoids, inflammation, and oxidative stress—implications of cellular signaling pathways and relation to chronic disease prevention. Nutr. Res 34(11):907–929. https://doi.org/10.1016/j.nutres.2014.07.010
Kim K et al (2014) Sasa quelpaertensis leaf extract suppresses dextran sulfate sodium–induced colitis in mice by inhibiting the proinflammatory mediators and mitogen-activated protein kinase phosphorylation. Nutr Res 34(10):894–905. https://doi.org/10.1016/j.nutres.2014.09.002
Lee B et al (2010) Anti-ulcerogenic effect and HPLC analysis of the caffeoylquinic acid-rich extract from Ligularia stenocephala. Biol Pharm Bull 33(3):493–497
Liang N, Kitts D (2015) Role of chlorogenic acids in controlling oxidative and inflammatory stress conditions. Nutrients 8(1):16. https://doi.org/10.3390/nu8010016
Liu S et al (2014) Lotus Leaf (Nelumbo nucifera) and its active constituents prevent rnflammatory Responses in macrophages via JNK/NF-κB signaling pathway. Am J Chin Med 42(4):869–889. https://doi.org/10.1142/S0192415X14500554
López V, Akerreta S, Casanova E, García-Mina J, Cavero R, Calvo M (2008) Screening of spanish medicinal plants for antioxidant and antifungal activities. Pharm. Biol 46(9):602–609. https://doi.org/10.1080/13880200802179634
Low D, Nguyen D, Mizoguchi E (2013) Animal models of ulcerative colitis and their application in drug research. Drug Des. Devel. Ther 7:1341. https://doi.org/10.2147/DDDT.S40107
Lykov AP, Poveshchenko OV, Bondarenko NA, Surovtseva MA, Kim II, Bgatova NP (2018) Therapeutic potential of biomedical cell product in DSS-induced inflammation in the small intestine of C57Bl/6 J mice. Bull Exp Biol Med 165(4):576–580. https://doi.org/10.1007/s10517-018-4216-5
Ma T et al (2004) TNF-α-induced increase in intestinal epithelial tight junction permeability requires NF-κB activation. Am. J. Physiol. Liver Physiol 286(3):G367–G376. https://doi.org/10.1152/ajpgi.00173.2003
Mankertz J, Schulzke JD (2007) Altered permeability in inflammatory bowel disease: pathophysiology and clinical implications. Curr. Opin. Gastroenterol 23(4):379–383. https://doi.org/10.1097/MOG.0b013e32816aa392
Molodecky N et al (2012) Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology 142(1):46–54.e42, quiz e30. https://doi.org/10.1053/j.gastro.2011.10.001
Ortega-Vidal J, Ruiz-Riaguas A, Fernández-de Córdova ML, Ortega-Barrales P, Llorent-Martínez EJ (2019) Phenolic profile and antioxidant activity of Jasonia glutinosa herbal tea. Influence of simulated gastrointestinal in vitro digestion. Food Chem 287:258–264. https://doi.org/10.1016/j.foodchem.2019.02.101
Oz H, Chen T, de Villiers W (2013) Green tea polyphenols and sulfasalazine have parallel anti-inflammatory properties in colitis models. Front. Immunol 4:132. https://doi.org/10.3389/fimmu.2013.00132
Pardo Santayana M, Morales R (2004) Consideraciones sobre el género Jasonia (Compositae, Inuleae). Sistemática y usos. Acta Bot, Malacit
Park M, Ji G, Sung M (2012) Dietary kaempferol suppresses inflammation of dextran sulfate sodium-induced colitis in mice. Dig Dis Sci 57(2):355–363. https://doi.org/10.1007/s10620-011-1883-8
Poritz L, Garver K, Green C, Fitzpatrick L, Ruggiero F, Koltun W (2007) Loss of the tight junction protein ZO-1 in dextran sulfate sodium induced colitis. J Surg Res 140(1):12–19. https://doi.org/10.1016/j.jss.2006.07.050
Rådmark O, Werz O, Steinhilber D, Samuelsson B (2015) 5-Lipoxygenase, a key enzyme for leukotriene biosynthesis in health and disease. Biochim Biophys Acta 1851(4):331–339. https://doi.org/10.1016/j.bbalip.2014.08.012
Rodríguez-Chávez J, Coballase-Urrutia E, Nieto-Camacho A, Delgado-Lamas G (2015) Antioxidant capacity of “Mexican Arnica” heterotheca inuloides cass natural products and some derivatives: their anti-inflammatory evaluation and effect on C. elegans life span. Oxid Med Cell Longev 2015:843237. https://doi.org/10.1155/2015/843237
Rubio B, Villaescusa L, Diaz A, Fernandez L, Martin T (1995) Flavonol Glycosides from Scolymus hispanicus and Jasonia glutinosa. Planta Med 61(6):583. https://doi.org/10.1055/s-2006-959386
Schneider I, Bucar F (2005) Lipoxygenase inhibitors from natural plant sources. Part 2: medicinal plants with inhibitory activity on arachidonate 12-lipoxygenase, 15-lipoxygenase and leukotriene receptor antagonists. Phyther Res 19(4):263–272. https://doi.org/10.1002/ptr.1604
Silva FA, Rodrigues BL, de Ayrizono M, Leal RF (2016) The immunological basis of inflammatory bowel disease. Gastroenterol Res Pract 2016:2097274. https://doi.org/10.1155/2016/2097274
Singh K, Jaggi AS, Singh N (2009) Exploring the ameliorative potential of Punica granatum in dextran sulfate sodium induced ulcerative colitis in mice. Phyther. Res 23(11):1565–1574. https://doi.org/10.1002/ptr.2822
Slattery ML, Benson J, Curtin K, Ma K-N, Schaeffer D, Potter JD (2000) Carotenoids and colon cancer. Am J Clin Nutr 71(2):575–582. https://doi.org/10.1093/ajcn/71.2.575
Trivedi PP, Jena GB (2015) Mechanistic insight into beta-carotene-mediated protection against ulcerative colitis-associated local and systemic damage in mice. Eur J Nutr 54(4):639–652. https://doi.org/10.1007/s00394-014-0745-5
Valentão P, Fernandes E, Carvalho F, Andrade PB, Seabra RM, Bastos ML (2001) Antioxidant activity of Centaurium erythraea infusion evidenced by its superoxide radical scavenging and xanthine oxidase inhibitory activity. J Agric Food Chem 49(7):3476–3479
Valero MS, Fagundes DS, Grasa L, Arruebo MP, Plaza MÁ, Murillo MD (2011) Contractile effect of tachykinins on rabbit small intestine. Acta Pharmacol Sin 32(4):487–494. https://doi.org/10.1038/aps.2010.227
Valero M, Berzosa C, Langa E, Gómez-Rincón C, López V (2013) Jasonia glutinosa D.C: botanical, phytochemical and pharmacological aspects. Boletín Latinoam. y del Caribe Plantas Med. y Aromáticas 12(6):543–557
Valero MS et al (2015) Rock Tea extract (Jasonia glutinosa) relaxes rat aortic smooth muscle by inhibition of L-type Ca2+ channels. J. Physiol. Biochem 71(4):785–793. https://doi.org/10.1007/s13105-015-0442-8
Villaescusa L, Díaz A, Bartolomé C (1995) Methoxylated flavonoids from Jasonia glutinosa, D.C., and their relation with others species of Jasonia. Pharmazie 50(9):639–640
Villaescusa L, Diaz A, Martin T, Gasquet M, Delmas F, Balansard G (1996) Preliminary screening of antiprotozoal activity of Jasonia glutinosa aerial parts. Int. J. Pharmacogn 34(4):303–304. https://doi.org/10.1076/phbi.34.4.303.13231
Villaescusa-Castillo L et al (2000) Antiprotozoal activity of sesquiterpenes from Jasonia glutinosa. Pharm. Biol 38(3):176–180. https://doi.org/10.1076/1388-0209(200007)3831-SFT176
Whittem CG, Williams AD, Williams CS (2010) Murine colitis modeling using dextran sulfate sodium (DSS). J. Vis. Exp 35:1652. https://doi.org/10.3791/1652
Won K, Suzuki T, Hori M, Ozaki H (2006) Motility disorder in experimentally obstructed intestine: relationship between muscularis inflammation and disruption of the ICC network. Neurogastroenterol Motil 18(1):53–61. https://doi.org/10.1111/j.1365-2982.2005.00718.x
Xiao H et al (2013) Inhibitory effect of the gallotannin corilagin on dextran sulfate sodium-induced murine ulcerative colitis. J Nat Prod 76(11):2120–2125. https://doi.org/10.1021/np4006772
Yazbeck R, Howarth GS, Butler RN, Geier MS, Abbott CA (2011) Biochemical and histological changes in the small intestine of mice with dextran sulfate sodium colitis. J Cell Physiol 226(12):3219–3224. https://doi.org/10.1002/jcp.22682
Zhang Z et al (2017) Chlorogenic acid ameliorates experimental colitis by promoting growth of Akkermansia in mice. Nutrients 9(7). https://doi.org/10.3390/nu9070677
Zheng K et al (2017) Traditional Chinese medicine combination therapy for patients with steroid-dependent ulcerative colitis: study protocol for a randomized controlled trial. Trials 18(1):8. https://doi.org/10.1186/s13063-016-1763-9
Acknowledgements
This work was supported by the government of Aragón, Spain (E-02 and B61), Universidad de Zaragoza (ref. JIUZ-2018-BIO-09 and BIO-2015-02), National Funds (FCT/MEC, Fundação para a Ciência e a Tecnologia/Ministério da Educação e Ciência) through project UID/QUI/50006/2013, co-financed by European Union (FEDER under the Partnership Agreement PT2020), from Norte Portugal Regional Operational Programme (NORTE 2020), under the PORTUGAL 2020 Partnership Agreement, through the European Regional Development Fund (ERDF) (project NORTE-01-0145-FEDER-000024), and from Programa de Cooperación Interreg V-A España – Portugal (POCTEP) 2014-2020 (project 0377_IBERPHENOL_6_E) and the European Social Fund “Construyendo Europa desde Aragón”. The authors also thank the Scientific Services of the Center for Biomedical Investigation (CIBA) for technical assistance. Jose Manuel López Cano is acknowledged for carrying out in vitro experiments with macrophages and Dr. Elisa Langa (USJ) for extraction procedures with the plant material.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval and consent to participate
This study was performed in accordance with the European Union Directive 2010/63 EU concerning experimental animal protection. Experimental protocols were approved by the Ethics Committee of the University of Zaragoza (PI66/14, Spain).
Consent for publication
All persons gave their informed consent prior to their inclusion in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Valero, M.S., González, M., Ramón-Gimenez, M. et al. Jasonia glutinosa (L.) DC., a traditional herbal medicine, reduces inflammation, oxidative stress and protects the intestinal barrier in a murine model of colitis. Inflammopharmacol 28, 1717–1734 (2020). https://doi.org/10.1007/s10787-019-00626-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-019-00626-0