Abstract
Introduction
Exercise-induced inflammation has been shown to be necessary for successful skeletal muscle regeneration post-injury. Accordingly, numerous investigations have demonstrated consequences of COX-inhibitors, anti-inflammatory drugs which prevent prostaglandin formation. In addition to its roles in inflammation, prostaglandin F2α (PGF2α) also mediates vital regenerative processes The majority of research to report consequences of suppressing inflammation has utilized acute injury models in combination with acute COX-inhibitor administration. To address the limited research investigating regular consumption of COX-inhibitors over time in exercising humans, the purpose of this study was to determine effects of a non-selective COX-inhibitor on a PGF2α metabolite and morphological adaptations of the upper body appendicular skeleton during periodized resistance training. Twenty-three (N = 23) recreationally trained college-aged males were randomly assigned to receive placebo (n = 11) or naproxen sodium (n = 12). Treatments were prophylactically administered in double-blind fashion with supervised upper body resistance exercise performed twice per week for 6 weeks. Venous blood was sampled pre- and post-exercise and analyzed for 13, 14-dihydro-15-keto PGF2α using enzyme immunoassay. Factorial mixed-design repeated-measures ANOVAs were utilized to examine relative changes in the plasma PGF2α metabolite and upper body appendicular morphology over the training period.
Results
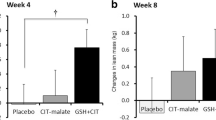
Naproxen sodium significantly reduced the acute PGF2α metabolite response to exercise (p = 0.013); however, this effect diminished over time (p = 0.02), and both treatment groups exhibited significant increases in dominant arm skeletal muscle tissue (p = 0.037).
Conclusion
Despite acute inhibition of the PGF2α metabolite at early time points, naproxen sodium did not hinder positive morphological adaptations of the upper body in response to resistance training.



Similar content being viewed by others
References
Bondesen BA, Mills ST, Kegley KM, Pavlath GK (2004) The COX-2 pathway is essential during early stages of skeletal muscle regeneration. Am J Physiol Cell Physiol 287:C475–C483
Boushel R, Langberg H, Gemmer C, Olesen J, Crameri R, Scheede C, Sander M, Kjaer M (2002) Combined inhibition of nitric oxide and prostaglandins reduces human skeletal muscle blood flow during exercise. J Physiol 543:691–698
Brewer C, Waddell D (2012) The role of prostaglandin F 2α in skeletal muscle regeneration. J Trainol 1:45–52
Brewer C, Bentley JP, Hallam JS, Woodyard C, Waddell D (2013) Use of analgesics for exercise-associated pain: prevalence and predictors of use in recreationally-trained college-aged students. J Strength Cond Res 28:74–81
Burd NA, Dickinson JM, Lemoine JK, Carroll CC, Sullivan BE, Haus JM, Jemiolo B, Trappe SW, Hughes GM, Sanders CE (2010) Effect of a cyclooxygenase-2 inhibitor on postexercise muscle protein synthesis in humans. Am J Physiol Endocrinol Metab 298:E354–E361
Horsley V, Pavlath GK (2003) Prostaglandin F2α stimulates growth of skeletal muscle cells via an NFATC2-dependent pathway. J Cell Biol 161:111–118
Keppel G, Wickens TD (2004) Design and analysis: a researcher’s handbook, 4th edn. Pearson, New Jersey
Kini U, Nandeesh BN (2012) Physiology of bone formation, remodeling, and metabolism. In: Fogelman I, Gnanasegaran G, van der Wall H (eds) Radionuclide and hybrid bone imaging. Springer, Berlin
Krentz JR, Quest B, Farthing JP, Quest DW, Chilibeck PD (2008) The effects of ibuprofen on muscle hypertrophy, strength, and soreness during resistance training. Appl Physiol Nutr Metab 33:470–475
Langberg H, Skovgaard D, Karamouzis M, Bulow J, Kjaer M (1999) Metabolism and inflammatory mediators in the peritendinous space measured by microdialysis during intermittent isometric exercise in humans. J Physiol 515:919–927
Lapointe BM, Frémont P, Côté CH (2002) Adaptation to lengthening contractions is independent of voluntary muscle recruitment but relies on inflammation. Am J Physiol Regul Integr Comp Physiol 282:R323–R329
Lapointe BM, Fremont P, Cote CH (2003) Influence of nonsteroidal anti-inflammatory drug treatment duration and time of onset on recovery from exercise-induced muscle damage in rats. Arch Phys Med Rehabil 84:651–655
Mackey AL, Kjaer M, Dandanell S, Mikkelsen KH, Holm L, Dossing S, Kadi F, Koskinen SO, Jensen CH, Schroder HD, Langberg H (2007) The influence of anti-inflammatory medication on exercise-induced myogenic precursor cell responses in humans. J Appl Physiol 103:425–431
Mendias CL, Tatsumi R, Allen RE (2004) Role of cyclooxygenase-1 and -2 in satellite cell proliferation, differentiation, and fusion. Muscle Nerve 30:497–500
Mikkelsen UR, Helmark IC, Kjaer M, Langberg H (2008) Prostaglandin synthesis can be inhibited locally by infusion of NSAIDS through microdialysis catheters in human skeletal muscle. J Appl Physiol 104:534–537
Mikkelsen UR, Langberg H, Helmark IC, Skovgaard D, Andersen LL, Kjaer M, Mackey AL (2009) Local NSAID infusion inhibits satellite cell proliferation in human skeletal muscle after eccentric exercise. J Appl Physiol 107:1600–1611
Mishra DK, Fridén J, Schmitz MC, Lieber RL (1995) Anti-inflammatory medication after muscle injury. A treatment resulting in short-term improvement but subsequent loss of muscle function. J Bone Joint Surg 77:1510–1519
Novak ML, Billich W, Smith SM, Sukhija KB, McLoughlin TJ, Hornberger TA, Koh TJ (2009) COX-2 inhibitor reduces skeletal muscle hypertrophy in mice. Am J Physiol Regul Integr Comp Physiol 296:R1132–R1139
Otis JS, Burkholder TJ, Pavlath GK (2005) Stretch-induced myoblast proliferation is dependent on the COX2 pathway. Exp Cell Res 310:417–425
Paulsen G, Egner I, Drange M, Langberg H, Benestad H, Fjeld J, Hallen J, Raastad T (2010) A COX-2 inhibitor reduces muscle soreness, but does not influence recovery and adaptation after eccentric exercise. Scand J Med Sci Sports 20:e195–e207
Peterson JM, Trappe TA, Mylona E, White F, Lambert CP, Evans WJ, Pizza FX (2003) Ibuprofen and acetaminophen: effect on muscle inflammation after eccentric exercise. Med Sci Sports Exerc 35:892–896
Phillips SM, Tipton KD, Ferrando AA, Wolfe RR (1999) Resistance training reduces the acute exercise-induced increase in muscle protein turnover. Am J Physiol Endocrinol Metab 276:E118–E124
Raisz LG (1999) Prostaglandins and bone: physiology and pathophysiology. Osteoarthr Cartil 7:419–421
Richards J, Joseph L, Schwartzman K, Kreiger N, Tenenhouse A, Goltzman D (2006) The effect of cyclooxygenase-2 inhibitors on bone mineral density: results from the Canadian Multicentre Osteoporosis Study. Osteoporos Int 17:1410–1419
Rodemann HP, Goldberg AL (1982) Arachidonic acid, prostaglandin E2 and F2 alpha influence rates of protein turnover in skeletal and cardiac muscle. J Biol Chem 257:1632–1638
Shen W, Li Y, Tang Y, Cummins J, Huard J (2005) NS-398, a cyclooxygenase-2-specific inhibitor, delays skeletal muscle healing by decreasing regeneration and promoting fibrosis. Am J Pathol 167:1105–1117
Smith RH, Palmer RM, Reeds P (1983) Protein synthesis in isolated rabbit forelimb muscles. The possible role of metabolites of arachidonic acid in the response to intermittent stretching. Biochem J 214:153–161
Trappe TA, Fluckey JD, White F, Lambert CP, Evans WJ (2001) Skeletal muscle PGF2{alpha}and PGE2 in response to eccentric resistance exercise: influence of ibuprofen and acetaminophen. J Clin Endocrinol Metab 86:5067–5070
Trappe TA, White F, Lambert CP, Cesar D, Hellerstein M, Evans WJ (2002) Effect of ibuprofen and acetaminophen on postexercise muscle protein synthesis. Am J Physiol Endocrinol Metab 282:E551–E556
Trappe TA, Carroll CC, Dickinson JM, Lemoine JK, Haus JM, Sullivan BE, Lee JD, Jemiolo B, Weinheimer EM, Hollon CJ (2011) Influence of acetaminophen and ibuprofen on skeletal muscle adaptations to resistance exercise in older adults 300(3):R655–R662
Vandervoort A, Sale D, Moroz J (1984) Comparison of motor unit activation during unilateral and bilateral leg extension. J Appl Physiol 56:46–51
Vane J (1971) Inhibition of prostaglandin synthesis as a mechanism of action for aspirin-like drugs. Nat New Biol 231:232–235
Vane JR, Botting RM (1996) Mechanism of action of anti-inflammatory drugs. Scand J Rheumatol 25:9–21
Zalin RJ (1987) The role of hormones and prostanoids in the in vitro proliferation and differentiation of human myoblasts. Exp Cell Res 172:265–281
Acknowledgments
The authors would like to acknowledge Lisa Huggins, R.Ph. for assistance with pharmaceutical aspects and placebo design, Andrea Johnson, Ph.D. for DXA scanning, and the participants for full commitment to the study for the required time.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical standard
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
IRB approved informed consent was provided by all participants.
Rights and permissions
About this article
Cite this article
Brewer, C.B., Bentley, J.P., Day, L.B. et al. Resistance exercise and naproxen sodium: effects on a stable PGF2α metabolite and morphological adaptations of the upper body appendicular skeleton. Inflammopharmacol 23, 319–327 (2015). https://doi.org/10.1007/s10787-015-0248-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-015-0248-x