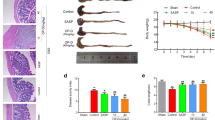
Abstract
To investigate the beneficial effects of oridonin, a diterpenoid compound isolated from Rabdosia rubescens, on the inflammatory response in TNBS-induced post-inflammatory irritable bowel syndrome (PI-IBS) model and the underlying mechanism. Using the PI-IBS rat model and Caco-2 cell lines, we found that intestinal barrier function reflected by lactulose/mannitol (L/M) ratio and tight junction protein level was significantly ameliorated by oridonin. We also demonstrated that oridonin abrogated inflammation through inhibiting the phosphorylation of NF-κBp65 as well as its downstream gene (iNOS, COX-2, IL-1β, and IL-6) level. Molecular docking studies confirmed the good binding activity between oridonin and PXR. In Caco-2 cell lines, oridonin markedly inhibited LPS-induced NF-κB activation in a PXR-dependent manner. Meanwhile, PXR and its target genes CYP3A4 and P-gp were induced by oridonin, which was associated with the decreased expression of NF-κB and the recovery of intestinal barrier. This study indicated that the therapeutic effect of oridonin on experimental PI-IBS through repairing intestinal barrier function may be closely associated with the regulatory role of PXR/NF-κB signaling pathway. Oridonin may serve as a PXR ligand for the development of drugs in the therapy for PI-IBS.








Similar content being viewed by others
Abbreviations
- PI-IBS:
-
post-inflammatory irritable bowel syndrome
- IBD:
-
inflammation bowel diseases
- L/M:
-
lactulose/mannitol; NF-κB: Nuclear factor-κB
- PXR:
-
pregnane X receptor
- TNBS:
-
2,4,6-trinitrobenzenesulfonic acid
- RFP:
-
rifampicin
- KCZ:
-
ketoconazole
- AWR:
-
abdominal withdrawal reflex
- DAI:
-
disease activity index
- LPS:
-
lipopolysaccharide;
- HPLC-MS/MS:
-
high-performance liquid chromatography electrospray ionization tandem mass spectrometry;
- LBD:
-
ligand binding domain
- TJ:
-
tight junction
- TLR4:
-
toll-like receptor 4
- MPO:
-
myeloperoxidase
References
Wang, H., J. Gu, X. Hou, J. Chen, N. Yang, Y. Liu, G. Wang, M. du, H. Qiu, Y. Luo, Z. Jiang, and L. Feng. 2017. Anti-inflammatory effect of miltirone on inflammatory bowel disease via TLR4/NF-kappaB/IQGAP2 signaling pathway. Biomedicine & Pharmacotherapy 85: 531–540. https://doi.org/10.1016/j.biopha.2016.11.061.
Du, L., Y. Long, J.J. Kim, B. Chen, Y. Zhu, and N. Dai. 2019. Protease Activated Receptor-2 Induces Immune Activation and Visceral Hypersensitivity in Post-infectious Irritable Bowel Syndrome Mice. Digestive Diseases and Sciences 64 (3): 729–739. https://doi.org/10.1007/s10620-018-5367-y.
Zhang, S., W. Xu, H. Wang, M. Cao, M. Li, J. Zhao, Y. Hu, Y. Wang, S. Li, Y. Xie, G. Chen, R. Liu, Y. Cheng, Z. Xu, K. Zou, S. Gong, and L. Geng. 2019. Inhibition of CREB-mediated ZO-1 and activation of NF-kappaB-induced IL-6 by colonic epithelial MCT4 destroys intestinal barrier function. Cell Proliferation 52 (6): e12673. https://doi.org/10.1111/cpr.12673.
Robertson, M.D., C. Pedersen, P.J. Hinton, Mendis Asjr, P.D. Cani, and B.A. Griffin. 2018. Elevated high density lipoprotein cholesterol and low grade systemic inflammation is associated with increased gut permeability in normoglycemic men. Nutrition, Metabolism, and Cardiovascular Diseases 28 (12): 1296–1303. https://doi.org/10.1016/j.numecd.2018.07.006.
Fukui, H. 2016. Increased Intestinal Permeability and Decreased Barrier Function: Does It Really Influence the Risk of Inflammation? Inflammatory Intestinal Diseases 1 (3): 135–145. https://doi.org/10.1159/000447252.
Lan, C., X.N. Sun, X.C. Zhou, B. Yang, B.L. Huang, T.Z. Deng, Z.T. He, and X.Y. Han. 2016. Preinduced intestinal HSP70 improves visceral hypersensitivity and abnormal intestinal motility in PI-IBS mouse model. Asian Pacific Journal of Tropical Medicine 9 (3): 302–305. https://doi.org/10.1016/j.apjtm.2016.01.022.
Tao, J.H., J.A. Duan, W. Zhang, S. Jiang, J.M. Guo, and D.D. Wei. 2018. Polysaccharides From Chrysanthemum morifolium Ramat Ameliorate Colitis Rats via Regulation of the Metabolic Profiling and NF-kappa B/TLR4 and IL-6/JAK2/STAT3 Signaling Pathways. Frontiers in Pharmacology 9: 746. https://doi.org/10.3389/fphar.2018.00746.
Jiang, F., M. Liu, H. Wang, G. Shi, B. Chen, T. Chen, X. Yuan, P. Zhu, J. Zhou, Q. Wang, and Y. Chen. 2020. Wu Mei Wan attenuates CAC by regulating gut microbiota and the NF-kB/IL6-STAT3 signaling pathway. Biomedicine & Pharmacotherapy 125: 109982. https://doi.org/10.1016/j.biopha.2020.109982.
Chen, S., J. Ye, X. Chen, J. Shi, W. Wu, W. Lin, W. Lin, Y. Li, H. Fu, and S. Li. 2018. Valproic acid attenuates traumatic spinal cord injury-induced inflammation via STAT1 and NF-kappaB pathway dependent of HDAC3. Journal of Neuroinflammation 15 (1): 150. https://doi.org/10.1186/s12974-018-1193-6.
Li, F., D. Liang, Z. Yang, T. Wang, W. Wang, X. Song, M. Guo, E. Zhou, D. Li, Y. Cao, and N. Zhang. 2013. Astragalin suppresses inflammatory responses via down-regulation of NF-kappaB signaling pathway in lipopolysaccharide-induced mastitis in a murine model. International Immunopharmacology 17 (2): 478–482. https://doi.org/10.1016/j.intimp.2013.07.010.
Deuring, J.J., M. Li, W. Cao, S. Chen, W. Wang, C. de Haar, C.J. van der Woude, and M. Peppelenbosch. 2019. Pregnane X receptor activation constrains mucosal NF-kappaB activity in active inflammatory bowel disease. PLoS One 14 (10): e0221924. https://doi.org/10.1371/journal.pone.0221924.
Wahli, W. 2008. A gut feeling of the PXR, PPAR and NF-kappaB connection. Journal of Internal Medicine 263 (6): 613–619. https://doi.org/10.1111/j.1365-2796.2008.01951.x.
Xu, C., M. Huang, and H. Bi. 2016. PXR- and CAR-mediated herbal effect on human diseases. Biochimica et Biophysica Acta 1859 (9): 1121–1129. https://doi.org/10.1016/j.bbagrm.2016.02.009.
Xu, L., L. Li, C.Y. Zhang, H. Schluesener, and Z.Y. Zhang. 2019. Natural Diterpenoid Oridonin Ameliorates Experimental Autoimmune Neuritis by Promoting Anti-inflammatory Macrophages Through Blocking Notch Pathway. Frontiers in Neuroscience 13: 272. https://doi.org/10.3389/fnins.2019.00272.
Yang, H., H. Lv, H. Li, X. Ci, and L. Peng. 2019. Oridonin protects LPS-induced acute lung injury by modulating Nrf2-mediated oxidative stress and Nrf2-independent NLRP3 and NF-kappaB pathways. Cell Communication and Signaling: CCS 17 (1): 62. https://doi.org/10.1186/s12964-019-0366-y.
Zang, K.H., Y.Y. Shao, X. Zuo, Z. Rao, and H.Y. Qin. 2016. Oridonin alleviates visceral hyperalgesia in a rat model of postinflammatory irritable bowel syndrome: Role of colonic enterochromaffin cell and serotonin availability. Journal of Medicinal Food 19 (6): 586–592. https://doi.org/10.1089/jmf.2015.3595.
Qin, Hong-Yan. 2012. Key factors in developing the trinitrobenzene sulfonic acid-induced post-inflammatory irritable bowel syndrome model in rats. World Journal of Gastroenterology 18 (20): 2481–2492. https://doi.org/10.3748/wjg.v18.i20.2481.
Hou, Q., Y. Huang, Z. Zhu, L. Liao, X. Chen, Q. Han, and F. Liu. 2019. Tong-Xie-Yao-Fang improves intestinal permeability in diarrhoea-predominant irritable bowel syndrome rats by inhibiting the NF-kappaB and notch signalling pathways. BMC Complementary and Alternative Medicine 19 (1): 337. https://doi.org/10.1186/s12906-019-2749-4.
Sanchez de Medina, F., I. Romero-Calvo, C. Mascaraque, and O. Martinez-Augustin. 2014. Intestinal inflammation and mucosal barrier function. Inflammatory Bowel Diseases 20 (12): 2394–2404. https://doi.org/10.1097/MIB.0000000000000204.
Zhang, Wenjing, Qilai Huang, and Zi-Chun Hua. 2010. Oridonin: A promising anticancer drug from China. Frontiers in Biology 5 (6): 540–545. https://doi.org/10.1007/s11515-010-0610-8.
Xu, X.J., Y.L. Zhang, L. Liu, L. Pan, and S.K. Yao. 2017. Increased expression of nerve growth factor correlates with visceral hypersensitivity and impaired gut barrier function in diarrhoea-predominant irritable bowel syndrome: A preliminary explorative study. Alimentary Pharmacology & Therapeutics 45 (1): 100–114. https://doi.org/10.1111/apt.13848.
Wang, Q., L. Ning, Y. Niu, H. Liu, and X. Yao. 2015. Molecular mechanism of the inhibition and remodeling of human islet amyloid polypeptide (hIAPP(1-37)) oligomer by resveratrol from molecular dynamics simulation. The Journal of Physical Chemistry. B 119 (1): 15–24. https://doi.org/10.1021/jp507529f.
Yang, M., W. Jia, D. Wang, F. Han, W. Niu, H. Zhang, D.Q. Shih, and X. Zhang. 2019. Effects and mechanism of constitutive TL1A expression on intestinal mucosal barrier in DSS-induced colitis. Digestive Diseases and Sciences 64 (7): 1844–1856. https://doi.org/10.1007/s10620-019-05580-z.
Ning, L., X. Lou, F. Zhang, and G. Xu. 2019. Nuclear receptors in the pathogenesis and management of inflammatory bowel disease. Mediators of Inflammation 2019: 2624941–2624913. https://doi.org/10.1155/2019/2624941.
Gabbia, D., L. Pozzo, G. Zigiotto, M. Roverso, D. Sacchi, A. Dalla Pozza, M. Carrara, S. Bogialli, A. Floreani, M. Guido, and S. de Martin. 2018. Dexamethasone counteracts hepatic inflammation and oxidative stress in cholestatic rats via CAR activation. PLoS One 13 (9): e0204336. https://doi.org/10.1371/journal.pone.0204336.
Guo, M.Z., M. Meng, C.C. Feng, X. Wang, and C.L. Wang. 2019. A novel polysaccharide obtained from Craterellus cornucopioides enhances immunomodulatory activity in immunosuppressive mice models via regulation of the TLR4-NF-kappaB pathway. Food & Function 10 (8): 4792–4801. https://doi.org/10.1039/c9fo00201d.
Zhang, J., Z. Zhao, H. Bai, M. Wang, L. Jiao, W. Peng, T. Wu, T. Liu, H. Chen, X. Song, L. Wu, X. Hu, Q. Wu, J. Zhou, J. Song, M. Lyv, and B. Ying. 2019. Genetic polymorphisms in PXR and NF-kappaB1 influence susceptibility to anti-tuberculosis drug-induced liver injury. PLoS One 14 (9): e0222033. https://doi.org/10.1371/journal.pone.0222033.
Funding
We thank the Youth fund of the second affiliated hospital of Shanxi medical university (grant: 201802-1 and 201902-2) and Shanxi Basic Applied Research Program (201901D111389).
Author information
Authors and Affiliations
Contributions
Y.S designed the study. R.H. and J.G conceived the experimental protocol. X.F., Z.C., G.D., and D.X. performed the experiments. Y.G. interpreted the results and wrote the manuscript. J.L. reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no conflict of interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
About this article
Cite this article
Shao, Yy., Guo, Y., Feng, Xj. et al. Oridonin Attenuates TNBS-induced Post-inflammatory Irritable Bowel Syndrome via PXR/NF-κB Signaling. Inflammation 44, 645–658 (2021). https://doi.org/10.1007/s10753-020-01364-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-020-01364-0