Abstract
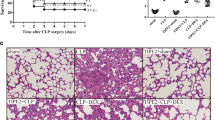
We have found earlier that Tubastatin A (TubA), a selective inhibitor of histone deacetylase 6 (HDAC6), improves survival in a mouse model of lethal cecal ligation and puncture (CLP)-induced sepsis. However, the underlying mechanisms have not been fully established. This study sought to test the hypothesis that TubA could affect both lung and splenic functions. C57BL/6J mice were subjected to CLP, and randomized to receive either TubA (70 mg/kg) dissolved in dimethyl sulfoxide (DMSO), or DMSO alone, 1 h following CLP. Sham animals acted as control. Twenty-four hours later, lung tissue was harvested for pathological examination, and splenic tissue was harvested for bacterial colonization. In a parallel study, the spleen was collected 48 h following CLP, and single cell suspension was prepared. Splenocytes then underwent flow cytometry to analyze the immune cell population. RAW264.7 macrophages were treated with lipopolysaccharide (LPS) with or without the presence of TubA (10 μM) at 37 °C for 3 h to assess the effect on macrophage phagocytosis. We found that acute lung injury secondary to lethal sepsis was attenuated by TubA. Treatment with TubA restored the percentage of B lymphocytes, and significantly increased percentages of innate immune cells and macrophages compared to the vehicle-treated CLP group. Moreover, TubA significantly decreased the bacterial load in the spleen, and improved the phagocytic ability of RAW264.7 murine macrophages in vitro. Such findings may help to explain the beneficial effects of TubA treatment in a model of lethal sepsis, as previously reported.




Similar content being viewed by others
References
Hicks, P., D.J. Cooper, and Australian, New Zealand Intensive Care Society B, Clinical Trials Group Executive C. 2008. The Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock: 2008. Critical Care and Resuscitation 10 (1): 8.
Singer, M., C.S. Deutschman, C.W. Seymour, M. Shankar-Hari, D. Annane, M. Bauer, R. Bellomo, G.R. Bernard, J.D. Chiche, C.M. Coopersmith, R.S. Hotchkiss, M.M. Levy, J.C. Marshall, G.S. Martin, S.M. Opal, G.D. Rubenfeld, T. van der Poll, J.L. Vincent, and D.C. Angus. 2016. The third international consensus definitions for sepsis and septic shock (sepsis-3). JAMA : the journal of the American Medical Association. 315 (8): 801–810.
Shankar-Hari, M., G.S. Phillips, M.L. Levy, C.W. Seymour, V.X. Liu, C.S. Deutschman, D.C. Angus, G.D. Rubenfeld, M. Singer, and for the Sepsis Definitions Task Force. 2016. Developing a new definition and assessing new clinical criteria for septic shock: for the third international consensus definitions for sepsis and septic shock (sepsis-3). JAMA : the journal of the American Medical Association. 315 (8): 775–787.
Daviaud, F., D. Grimaldi, A. Dechartres, J. Charpentier, G. Geri, N. Marin, J.D. Chiche, A. Cariou, J.P. Mira, and F. Pène. 2015. Timing and causes of death in septic shock. Annals of Intensive Care 5 (1): 16.
Varisco, B.M. 2011. The pharmacology of acute lung injury in sepsis. Advances in Pharmacological Sciences 2011: 254619.
Luan, Y.Y., Y.M. Yao, X.Z. Xiao, and Z.Y. Sheng. 2015. Insights into the apoptotic death of immune cells in sepsis. Journal of interferon & cytokine research : the official journal of the International Society for Interferon and Cytokine Research. 35 (1): 17–22.
Mortaz, E., M.R. Masjedi, P.J. Barnes, and I.M. Adcock. 2011. Epigenetics and chromatin remodeling play a role in lung disease. Tanaffos. 10 (4): 7–16.
Zhang, X., Z. Yuan, Y. Zhang, S. Yong, A. Salas-Burgos, J. Koomen, N. Olashaw, J.T. Parsons, X.J. Yang, S.R. Dent, T.P. Yao, W.S. Lane, and E. Seto. 2007. HDAC6 modulates cell motility by altering the acetylation level of cortactin. Molecular Cell 27 (2): 197–213.
Hubbert, C., A. Guardiola, R. Shao, Y. Kawaguchi, A. Ito, A. Nixon, M. Yoshida, X.F. Wang, and T.P. Yao. 2002. HDAC6 is a microtubule-associated deacetylase. Nature 417 (6887): 455–458.
Ai, J., Y. Wang, J.A. Dar, J. Liu, L. Liu, J.B. Nelson, and Z. Wang. 2009. HDAC6 regulates androgen receptor hypersensitivity and nuclear localization via modulating Hsp90 acetylation in castration-resistant prostate cancer. Molecular Endocrinology 23 (12): 1963–1972.
Hackanson, B., L. Rimmele, M. Benkisser, M. Abdelkarim, M. Fliegauf, M. Jung, et al. 2012. HDAC6 as a target for antileukemic drugs in acute myeloid leukemia. Leukemia research. 36 (8): 1055–1062.
Hideshima, T., J. Qi, R.M. Paranal, W. Tang, E. Greenberg, N. West, M.E. Colling, G. Estiu, R. Mazitschek, J.A. Perry, H. Ohguchi, F. Cottini, N. Mimura, G. Görgün, Y.T. Tai, P.G. Richardson, R.D. Carrasco, O. Wiest, S.L. Schreiber, K.C. Anderson, and J.E. Bradner. 2016. Discovery of selective small-molecule HDAC6 inhibitor for overcoming proteasome inhibitor resistance in multiple myeloma. Proceedings of the National Academy of Sciences of the United States of America 113 (46): 13162–13167.
Medler, T.R., J.M. Craig, A.A. Fiorillo, Y.B. Feeney, J.C. Harrell, and C.V. Clevenger. 2016. HDAC6 deacetylates HMGN2 to regulate Stat5a activity and breast cancer growth. Molecular Cancer Research 14 (10): 994–1008.
Lemon, D.D., T.R. Horn, M.A. Cavasin, M.Y. Jeong, K.W. Haubold, C.S. Long, D.C. Irwin, S.A. McCune, E. Chung, L.A. Leinwand, and T.A. McKinsey. 2011. Cardiac HDAC6 catalytic activity is induced in response to chronic hypertension. Journal of Molecular and Cellular Cardiology 51 (1): 41–50.
Rivieccio, M.A., C. Brochier, D.E. Willis, B.A. Walker, M.A. D’Annibale, K. McLaughlin, A. Siddiq, A.P. Kozikowski, S.R. Jaffrey, J.L. Twiss, R.R. Ratan, and B. Langley. 2009. HDAC6 is a target for protection and regeneration following injury in the nervous system. Proceedings of the National Academy of Sciences of the United States of America 106 (46): 19599–19604.
Li, Y., T. Zhao, B. Liu, I. Halaweish, R. Mazitschek, X. Duan, and H.B. Alam. 2015. Inhibition of histone deacetylase 6 improves long-term survival in a lethal septic model. The journal of trauma and acute care surgery. 78 (2): 378–385.
Zhao T, Li Y, Liu B, Pan B, Cheng X, Georgoff P, et al. Inhibition of histone deacetylase 6 restores innate immune cells in the bone marrow in a lethal septic model. The journal of trauma and acute care surgery. 2016;80(1):34-40; discussion -1.
Zhao, T., B. Pan, H.B. Alam, B. Liu, R.T. Bronson, Q. Deng, E. Wu, and Y. Li. 2016. Protective effect of Cl-amidine against CLP-induced lethal septic shock in mice. Scientific Reports 6: 36696.
Liu, Z., Y. Li, W. Chong, D.K. Deperalta, X. Duan, B. Liu, I. Halaweish, P. Zhou, and H.B. Alam. 2014. Creating a prosurvival phenotype through a histone deacetylase inhibitor in a lethal two-hit model. Shock 41 (2): 104–108.
Kim, K., Y. Li, G. Jin, W. Chong, B. Liu, J. Lu, K. Lee, M. deMoya, G.C. Velmahos, and H.B. Alam. 2012. Effect of valproic acid on acute lung injury in a rodent model of intestinal ischemia reperfusion. Resuscitation 83 (2): 243–248.
Thangavel, J., S. Samanta, S. Rajasingh, B. Barani, Y.T. Xuan, B. Dawn, and J. Rajasingh. 2015. Epigenetic modifiers reduce inflammation and modulate macrophage phenotype during endotoxemia-induced acute lung injury. Journal of Cell Science 128 (16): 3094–3105.
Yu, J., Z. Ma, S. Shetty, M. Ma, and J. Fu. 2016. Selective HDAC6 inhibition prevents TNF-alpha-induced lung endothelial cell barrier disruption and endotoxin-induced pulmonary edema. American journal of physiology Lung cellular and molecular physiology. 311 (1): L39–L47.
Shukla, P., G.M. Rao, G. Pandey, S. Sharma, N. Mittapelly, R. Shegokar, and P.R. Mishra. 2014. Therapeutic interventions in sepsis: current and anticipated pharmacological agents. British Journal of Pharmacology 171 (22): 5011–5031.
Boomer, J.S., To K, K.C. Chang, O. Takasu, D.F. Osborne, A.H. Walton, et al. 2011. Immunosuppression in patients who die of sepsis and multiple organ failure. JAMA : the journal of the American Medical Association. 306 (23): 2594–2605.
Ariffin JK, das Gupta K, Kapetanovic R, Iyer A, Reid RC, Fairlie DP, et al. Histone deacetylase inhibitors promote mitochondrial reactive oxygen species production and bacterial clearance by human macrophages. Antimicrobial Agents and Chemotherapy 2015;60(3):1521–1529.
Borges da Silva H, Fonseca R, Pereira RM, Cassado Ados A, Alvarez JM, D’Imperio Lima MR. Splenic macrophage subsets and their function during blood-borne infections. Frontiers in immunology. 2015;6:480.
Roger T LJ, Le Roy D, Goy G, Mombelli M, Koessler T, Ding XC, Chanson AL, Reymond MK, Miconnet I, Schrenzel J, François P, Calandra T. Histone deacetylase inhibitors impair innate immune responses to Toll-like receptor agonists and to infection.pdf. Blood 2011;117:1205–1217.
Chen, Y., R.L. Pan, X.L. Zhang, J.Z. Shao, L.X. Xiang, X.J. Dong, and G.R. Zhang. 2009. Induction of hepatic differentiation of mouse bone marrow stromal stem cells by the histone deacetylase inhibitor VPA. Journal of Cellular and Molecular Medicine 13 (8B): 2582–2592.
Ali, D., H. Alshammari, R. Vishnubalaji, E.P. Chalisserry, R. Hamam, M. Alfayez, et al. 2016. CUDC-907 promotes bone marrow adipocytic differentiation through inhibition of histone deacetylase and regulation of cell cycle. Stem Cells and Development.
Yan, B., S. Xie, Z. Liu, J. Ran, Y. Li, J. Wang, Y. Yang, J. Zhou, D. Li, and M. Liu. 2014. HDAC6 deacetylase activity is critical for lipopolysaccharide-induced activation of macrophages. PLoS One 9 (10): e110718.
Funding
This work was funded by a grant from NIH RO1 GM084127 to H.B.A. Data was presented at the 12th Annual Academic Surgical Congress, Clinical Congress Las Vegas, NV (February 2017).
Author information
Authors and Affiliations
Contributions
Q.D., T.Z., Y.L., and H.B.A. designed this study, for which H.B.A. secured funding. Q.D. and T.Z. performed the experiments and collected and analyzed data. B.L. and B.P provided experimental support. N.L. performed statistical analysis. E.C. presented the data at the 12th Annual Academic Surgical Congress. Q.D., T.Z., I.S.D., A.M.W., U.F.B., and Y.L. wrote the manuscript, which was critically reviewed and revised by Y.L. and H.B.A. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
The protocol for this study was approved by the University of Michigan Institutional Animal Care and Use Committee. All experiments complied with animal welfare and research regulations.
Conflict of Interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Deng, Q., Zhao, T., Pan, B. et al. Protective Effect of Tubastatin A in CLP-Induced Lethal Sepsis. Inflammation 41, 2101–2109 (2018). https://doi.org/10.1007/s10753-018-0853-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-018-0853-0