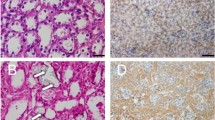
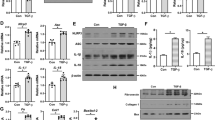
Abstract
Uric acid (UA) has been associated with renal fibrosis and progression of chronic kidney disease. However, the underlying mechanisms of this process have still not been identified. Here, we studied the role of the innate imunity receptor NLRP3/ASC in UA induced epithelial-mesenchymal transition (EMT) in kidney. Wistar rats were fed with oxonic acid 2% and UA 2% (OXA + U), OXA + U plus allopurinol (ALL) or regular chow (C) for 7 weeks. We analyzed the presence of EMT markers, the expression of NLRP3, ASC, Caspase-1 and Smad 2/3 molecules and the mitochondrial morphological and functional characteristics. High UA induced renal fibrosis, mild chronic inflammation, as well as morphological and biochemical evidence of EMT. High UA also increased the expression of NLRP3/ASC with activation of both inflammasome related caspase-1 and inflammasome unrelated Smad 2/3 pathways. Ultrastructural co-localization of NLRP3 and Smad 2/3 indicated physical interaction between the two molecules. No morphological or functional changes were found between mitochondria exposed to high UA. In conclusion, kidney epithelial NLRP3/ASC expression was increased in high UA state in rats and both inflammasome related caspase-1 and non-inflammasome related P-Smad 2/3 pathways were associated with the observed EMT, inflammation and fibrosis induced by UA in the kidney.




Similar content being viewed by others
References
Brand MD, Nicholls DG (2011) Assessing mitochondrial dysfunction in cells. Biochem J 435:297–312. doi:10.1042/BJ20110162
Cassina A, Radi R (1996) Differential inhibitory action of nitric oxide and peroxynitrite on mitochondrial electron transport. Arch Biochem Biophys 328:309–316. doi:10.1006/abbi.1996.0178
Cristobal-Garcia M, Garcia-Arroyo FE, Tapia E et al (2015) Renal oxidative stress induced by long-term hyperuricemia alters mitochondrial function and maintains systemic hypertension. Oxid Med Cell Longev 2015:535686. doi:10.1155/2015/535686
Fischer JC, Ruitenbeek W, Berden JA et al (1985) Differential investigation of the capacity of succinate oxidation in human skeletal muscle. Clin Chim Acta 153:23–36
George J, Struthers AD (2009) Role of urate, xanthine oxidase and the effects of allopurinol in vascular oxidative stress. Vasc Health Risk Manag 5:265–272. doi:10.2147/VHRM.S4265
Granata S, Masola V, Zoratti E et al (2015) NLRP3 inflammasome activation in dialyzed chronic kidney disease patients. PLoS One 10:1–16. doi:10.1371/journal.pone.0122272
He X, Liu Y, Usa K, et al (2014) Ultrastructure of mitochondria and the endoplasmic reticulum in renal tubules of Dahl salt-sensitive rats. Am J Physiol Renal Physiol 306:F1190–F1197. doi:10.1152/ajprenal.00073.2014
Hruska KA, Guo G, Wozniak M et al (2000) Osteogenic protein-1 prevents renal fibrogenesis associated with ureteral obstruction. Am J Physiol Renal Physiol 279:F130–F143
Kalluri R, Weinberg RA (2009) The basics of epithelial-mesenchymal transition. J Clin Invest 119:1420–1428. doi:10.1172/JCI39104
Khosla UM, Zharikov S, Finch JL et al (2005) Hyperuricemia induces endothelial dysfunction. Kidney Int 67:1739–1742. doi:10.1111/j.1523-1755.2005.00273.x
Kim SM, Lee SH, Kim YG et al (2015) Hyperuricemia-induced NLRP3 activation of macrophages contributes to the progression of diabetic nephropathy. Am J Physiol Renal Physiol 308:F993–F1003. doi:10.1152/ajprenal.00637.2014
Kriz W, Kaissling B, Le Hir M (2011) Epithelial-mesenchymal transition (EMT) in kidney fibrosis: fact or fantasy? J Clin Invest 121:468–474. doi:10.1172/JCI44595
Lech M, Avila-Ferrufino A, Skuginna V et al (2010) Quantitative expression of RIG-like helicase, NOD-like receptor and inflammasome-related mRNAs in humans and mice. Int Immunol 22:717–728. doi:10.1093/intimm/dxq058
Leemans JC, Kors L, Anders HJ et al (2014) Pattern recognition receptors and the inflammasome in kidney disease. Nat Rev Nephrol 10:398–414. doi:10.1038/nrneph.2014.91
Li L, Yang C, Zhao Y et al (2014) Is hyperuricemia an independent risk factor for new-onset chronic kidney disease? A systematic review and meta-analysis based on observational cohort studies. BMC Nephrol 15:122. doi:10.1186/1471-2369-15-122
Liu D, Wen Y, Tang TT et al (2015) Megalin/Cubulin-Lysosome-mediated albumin reabsorption is involved in the tubular cell activation of NLRP3 inflammasome and tubulointerstitial inflammation. J Biol Chem 290:18018–18028. doi:10.1074/jbc.M115.662064
Lorenz G, Darisipudi MN, Anders HJ (2014) Canonical and non-canonical effects of the NLRP3 inflammasome in kidney inflammation and fibrosis. Nephrology, dialysis, transplantation: official publication of the European Dialysis and Transplant Association—European Renal Association. Nephrol Dial Transplant 29:41–48. doi:10.1093/ndt/gft332
Martinon F, Petrilli V, Mayor A et al (2006) Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 440:237–241. doi:10.1038/nature04516
Mazzali M, Hughes J, Kim YG et al (2001) Elevated uric acid increases blood pressure in the rat by a novel crystal-independent mechanism. Hypertension 38:1101–1106. doi:10.1161/hy1101.092839
Mills KT, Xu Y, Zhang W et al (2015) A systematic analysis of worldwide population-based data on the global burden of chronic kidney disease in 2010. Kidney Int 88:950–957. doi:10.1038/ki.2015.230
Moriyama T, Itabashi M, Takei T et al (2015) High uric acid level is a risk factor for progression of IgA nephropathy with chronic kidney disease stage G3a. J Nephrol 28:451–456. doi:10.1007/s40620-014-0154-0
In: Guide for the care and use of laboratory animals (2011) Committee for the Update of the Guide care and use of laboratory animals. National Academy of Sciences. Chapter 2nd, 3rd, and 8th edn. Washington, DC
Petiti JP, Sosa Ldel V, Sabatino ME et al (2015) Involvement of MEK/ERK1/2 and PI3K/Akt pathways in the refractory behavior of GH3B6 pituitary tumor cells to the inhibitory effect of TGFbeta1. Endocrinology 156:534–547. doi:10.1210/en.2014-1070
Rodenbach KE, Schneider MF, Furth SL et al (2015) Hyperuricemia and progression of CKD in children and adolescents: the chronic kidney disease in children (CKiD) cohort study. Am J Kidney Dis 66:984–992. doi:10.1053/j.ajkd.2015.06.015
Rustin P, Chretien D, Bourgeron T et al (1994) Biochemical and molecular investigations in respiratory chain deficiencies. Clin Chim Acta 228:35–51
Ryu ES, Kim MJ, Shin HS, et al (2013) Uric acid-induced phenotypic transition of renal tubular cells as a novel mechanism of chronic kidney disease. Am J Physiol Renal Physiol 304:F471–F480. doi:10.1152/ajprenal.00560.2012
Sanchez-Lozada LG, Tapia E, Santamaria J et al (2005) Mild hyperuricemia induces vasoconstriction and maintains glomerular hypertension in normal and remnant kidney rats. Kidney Int 67:237–247. doi:10.1111/j.1523-1755.2005.00074.x
Sanchez-Lozada LG, Lanaspa MA, Cristobal-Garcia M et al (2012) Uric acid-induced endothelial dysfunction is associated with mitochondrial alterations and decreased intracellular ATP concentrations. Nephron Exp Nephrol 121:e71–e78. doi:10.1159/000345509
Schapira AH, Cooper JM, Dexter D et al (1990) Mitochondrial complex I deficiency in Parkinson’s disease. J Neurochem 54:823–827
Strutz F, Okada H, Lo CW et al (1995) Identification and characterization of a fibroblast marker: FSP1. J Cell Biol 130:393–405
Sutterwala FS, Haasken S, Cassel SL (2014) Mechanism of NLRP3 inflammasome activation. Ann N Y Acad Sci 1319:82–95. doi:10.1111/nyas.12458
Talaat KM, el-Sheikh AR (2007) The effect of mild hyperuricemia on urinary transforming growth factor beta and the progression of chronic kidney disease. Am J Nephrol 27:435–440. doi:10.1159/000105142
Usui F, Shirasuna K, Kimura H et al (2015) Inflammasome activation by mitochondrial oxidative stress in macrophages leads to the development of angiotensin II-induced aortic aneurysm. Arterioscler Thromb Vasc Biol 35:127–136. doi:10.1161/ATVBAHA.114.303763
Vilaysane A, Chun J, Seamone ME et al (2010) The NLRP3 inflammasome promotes renal inflammation and contributes to CKD. Am J Soc Nephrol 21:1732–1744. doi:10.1681/ASN.2010020143
Wang W, Wang X, Chun J et al (2013) Inflammasome-independent NLRP3 augments TGF-beta signaling in kidney epithelium. J Immunol 190:1239–1249. doi:10.4049/jimmunol.1201959
Wang W, Zhou PH, Xu CG et al (2015) Baicalein attenuates renal fibrosis by inhibiting inflammation via down-regulating NF-κB and MAPK signal pathways. J Mol Histol 46:283–290. doi:10.1007/s10735-015-9621-8
Zeisberg M, Neilson EG (2009) Biomarkers for epithelial-mesenchymal transitions. J Clin Invest 119:1429–1437. doi:10.1172/JCI36183
Zhou R, Yazdi AS, Menu P et al (2011) A role for mitochondria in NLRP3 inflammasome activation. Nature 469:221–225. doi:10.1038/nature09663
Zhuang Y, Yasinta M, Hu C et al (2015) Mitochondrial dysfunction confers albumin-induced NLRP3 inflammasome activation and renal tubular injury. Am J Physiol Renal Physiol 308:F857–F866. doi:10.1152/ajprenal.00203.2014
Acknowledgements
The authors thanks to Dr. Carolina Leimgruber, Mrs. Lucia Artino, Mrs. Maria Elena Pereyra, and Miss. María Soledad Santa Cruz for their technical support. We would also like to thank native speaker Dr. Paul Hobson for editing the English language of the manuscript.
Funding
This work was funded by the grants from Secretaria de Ciencia y Tecnologia from National University of Cordoba (Secyt, UNC) and Fondo para la Investigacion Cientifica y Tecnología (FonCyT), de la Agencia Nacional Cientifica y Tecnología.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that they have conflict of interest related to this work and approved the final version of the manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Romero, C.A., Remor, A., Latini, A. et al. Uric acid activates NRLP3 inflammasome in an in-vivo model of epithelial to mesenchymal transition in the kidney. J Mol Hist 48, 209–218 (2017). https://doi.org/10.1007/s10735-017-9720-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-017-9720-9