Abstract
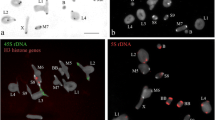
Satellite DNAs (satDNAs) are highly repetitive sequences that occur in virtually all eukaryotic genomes and can undergo rapid copy number and nucleotide sequence variation among relatives. After chromosomal mapping of the satDNA JcSAT1, it was found a large accumulation at subtelomeres of Jatropha curcas (subgenus Curcas), but an absence of these monomers in J. integerrima (subgenus Jatropha). This fact suggests a dynamic scenario for this satellite repeat in Jatropha genomes. Here, we used a multitasking approach (sequence analysis, DNA blotting and chromosomal mapping) to investigate the molecular organization and chromosomal abundance and distribution of JcSAT1 in a broader group of species from the subgenus Jatropha (J. gossypiifolia, J. mollissima, J. podagrica, and J. multifida) in addition to J. curcas, with the aiming of understanding the evolution of this satDNA. Based on the analysis of BAC clone sequences of J. curcas, a large array (~ 30 kb) of 80 homogeneous monomers of JcSAT1 was identified in BAC 23J11. The monomer size was conserved (~ 358 bp) and contained a telomeric motif at the 5’ end. PCR amplification coupled with a Southern blot revealed the presence of JcSAT1-like sequences in all species examined. However, a large set of genome copies was identified only in J. curcas, where a ladder-like pattern with multimers of different sizes was observed. In situ hybridization of BAC 23J11 confirmed the subtelomeric pattern for J. curcas, but showed no signals on chromosomes of species from the subgenus Jatropha. Our data indicate that JcSAT1 is a highly homogeneous satDNA that originated from a region near the telomeres and spread throughout the chromosomal subtermini, possibly due to frequent ectopic recombination between these regions. The abundance of JcSAT1 in the genome of J. curcas suggests that an amplification event occurred either at the base of the subgenus Curcas or at least in this species, although the repeat is shared by all species of the genus studied so far.




Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
References
Carvalho CR, Saraiva LS (1993) An air drying technique for maize chromosomes without enzymatic maceration. Biotech Histochem 68:142–145. https://doi.org/10.3109/10520299309104684
Chen NWM, Thareau V, Ribeiro T, Magdelenat G, Ashfield T, Innes RW, Pedrosa-Harand A, Geffroy V (2018) Common bean subtelomeres are hot spots of recombination and favor resistance gene evolution. Front Plant Sci 9:1185. https://doi.org/10.3389/fpls.2018.01185
Crooks GE, Hon G, Chandonia JM, Brenner SE (2004) WebLogo: A sequence logo generator. Genome Res 14:1188–1190. https://doi.org/10.1101/gr.849004
de Souza RC, Marques DA, Filho MMC, Oliveira ARS, Siqueira WJ, Benko-Iseppon AM, Brasileiro-Vidal AC (2019) Genome composition and pollen viability of Jatropha (Euphorbiaceae) interspecific hybrids by genomic in situ hybridization (GISH). Genet Mol Biology 42(4):e20190112. https://doi.org/10.1590/1678-4685-GMB-2019-0112
Dehgan B (1984) Phylogenetic significance of interspecific hybridization in Jatropha (Euphorbiaceae). Systematic Botany (1984), 9(4): pp. 467–478
Ferreira ME, Grattapaglia D (1995) Introdução ao ao uso de marcadores moleculares em analise genetica. 3 ed. Brasilia: EMBRAPA-CENARGEN, 1998. p 220
Fukuhara S, Muakrong N, Kikuchi S, Tanya P, Sassa H, Koba T, Srinives P (2016) Cytological characterization of an interspecific hybrid in Jatropha and its progeny reveals preferential uniparental chromosome transmission and interspecific translocation. Breeding Science 66: 838–844 (2016)https://doi.org/10.1270/jsbbs.16069
Garrido-Ramos MA (2015) Satellite DNA in plants: more than just rubbish. Cytogenet Genome Res 146:153–170. https://doi.org/10.1159/000437008
Ishii T, Juranić M, Maheshwari S, Bustamante FO, Vogt M, Gamboa-Salinas R et al (2020) Unequal contribution of two paralogous CENH3 variants in cowpea centromere function. Commun Biology 3:775. https://doi.org/10.1038/s42003-020-01507-x
Iwata A, Tek AL, Richard MMS, Abernathy B, Fônseca A, Schmutz J et al (2013) Identification and characterization of functional centromeres of the common bean. Plant J 76:47–60. https://doi.org/10.1111/tpj.12269
Kazama Y, Sugiyama R, Suto Y, Uchida W, Kawano S (2006) The clustering of four subfamilies of satellite DNA at individual chromosome ends in Silene latifolia. Genome 49(5):520–530. https://doi.org/10.1139/g05-130
Kikuchi S, Tsujimoto H, Sassa H, Koba T (2010) JcSat1, a novel subtelomeric repeat of Jatropha curcas L. and its use in karyotyping. Chromosome Sci 1311–16. https://doi.org/10.11352/scr.13.11
Leclear NM (2019) Evolution of Jatropha: Phylogenetics, Biogeography, and Phylogeography. Dissertation, University of Texas at Austin
Lima LG, Svartman M, Kuhn GCS (2017) Dissecting the satellite DNA landscape in three cactophilic Drosophila sequenced genomes. G3 7:2831–2843. https://doi.org/10.1534/g3.117.042093
Linardopoulou EV, Williams EM, Fan Y, Friedman C, Young JM, Trask BJ (2005) Human subtelomeres are hot spots of interchromosomal recombination and segmental duplication. Nature 473(7055):94–100. https://doi.org/10.1038/nature04029
Lower SS, McGurk MP, Clark AG, Barbash DA (2018) Satellite DNA evolution: old ideas, new approaches. Curr Opin Genet Dev 49:70–78. https://doi.org/10.1016/j.gde.2018.03.003
Marinho ACTA, Vasconcelos S, Vasconcelos EV, Marques DA, Benko-Iseppon AM, Brasileiro-Vidal AC (2018) Karyotype and genome size comparative analyses among six species of the oilseed-bearing genus Jatropha (Euphorbiaceae). Genet Mol Biology 41(2):442–449. https://doi.org/10.1590/1678-4685-GMB-2017-0120
Mefford HC, Trask BJ (2002) The complex structure and dynamic evolution of human subtelomeres. Nat Rev 3:91–102
Mlinarec J, Skuhala A, Jurković A, Malenica N, McCann J, Weiss-Schneeweiss H, Bohanec B, Besendorfer V (2019) The repetitive DNA composition in the natural pesticide producer Tanacetum cinerariifolium: interindividual variation of subtelomeric tandem repeats. Front Plant Sci 10:613. https://doi.org/10.3389/fpls.2019.00613
Muakrong N, Kikuchi S, Tanya P, Srinives P (2018a) Conservation and variation of 35S ribosomal DNA among five Jatropha species revealed by Fluorescence. Situ Hybrid Cytologia 83(1):57–61. https://doi.org/10.1508/cytologia.83.57
Muakrong N, Kikuchi S, Fukuhara S, Tanya P, Srinives P (2018b) Two Jatropha karyotypes constructed from meiotic pachytene chromosomes: pericentric distribution of heterochromatin and variation in repetitive DNAs. PLoS ONE 13(12):e0208549. https://doi.org/10.1371/journal.pone.0208549
Navajas-Pérez R, Schwarzacher T, Rejón MR, Garrido-Ramos MA (2009) Characterization of RUSI, a telomere-associated satellite DNA, in the genus Rumex (Polygonaceae). Cytogenet Genome Res 124:81–89. https://doi.org/10.1159/000200091
Novák P, Neumann P, Macas J (2020) Global analysis of repetitive DNA from unassembled sequence reads using RepeatExplorer2. Nat Protoc 15(11):3745–3776. https://doi.org/10.1038/s41596-020-0400-y
Pedrosa A, Sandal N, Stougaard J, Schweizer D, Bachmair A (2002) Chromosomal map of the model legume Lotus japonicus. Genetics 161:1661–1672
Pullaiah T, Bahadur B (2013) Economic and Medicinal Importance of Jatrophas. In: Bahadur B, Sujatha M, Carels M (eds) Jatropha, challenges for a new energy crop, volume 2: genetic improvement and biotechnology, 1st edn. Springer Science + Business Media, New York, pp 187–217
Reddy MP, Sudheer DVN, Mastan SG, Rahman H, Carels N, Bahadur B (2013) Karyology and genomics of Jatropha: current status and future prospects. In: Bahadur B, Sujatha M, Carels M (eds) Jatropha, challenges for a new energy crop. Genetic Improvement and Biotechnology, vol 2, 1st edn. Springer Science + Business Media, New York, pp 301–320
Ribeiro T, Santos KGB, Fonsêca A, Pedrosa-Harand A (2011) Isolation and characterization of a new repetitive DNA family recently amplified in the mesoamerican gene pool of the common bean (Phaseolus vulgaris L., Fabaceae). Genetica 139:1135–1142. https://doi.org/10.1007/s10709-011-9615-8
Ribeiro T, dos Santos KGB, Richard MMS, Sévignac M, Thareau V, Geffroy V, Pedrosa-Harand A (2017) Evolutionary dynamics of satellite DNA repeats from Phaseolus beans. Protoplasma 254:791–801. https://doi.org/10.1007/s00709-016-0993-8
Sato S, Hirakawa H, Isobe S, Fukai E, Watanabe A, Kato M et al (2011) Sequence analysis of the genome of an oil-bearing Tree, Jatropha curcas L. DNA Res 18(1):65–76. https://doi.org/10.1093/dnares/dsq030
Šatović-Vukšić E, Plohl M (2023) Satellite DNAs - from localized to highly dispersed genome components. Genes 14(3):742. https://doi.org/10.3390/genes14030742
Sonnhammer ELL, Durbin R (1995) A dot-matrix program with dynamic threshold control suited for genomic DNA and protein sequence analysis. Gene 167(1–2):GC1–G10. https://doi.org/10.1016/0378-1119(95)00714-8
Thakur J, Packiaraj J, Henikoff S (2021) Sequence, chromatin and evolution of Satellite DNA. Int J Mol Sci 22(9):4309. https://doi.org/10.3390/ijms22094309
Torres GA, Gong Z, Iovene M, Hirsch CD, Buell CR et al (2011) Organization and evolution of subtelomeric satellite repeats in the potato genome. Genes Genomes Genet 1(2):85–92. https://doi.org/10.1534/g3.111.000125
Vasconcelos S, Souza AA, Gusmão CLS, Milani M, Benko-Iseppon AM, Brasileiro-Vidal AC (2010) Heterochromatin and rDNA 5S and 45S sites as reliable cytogenetic markers for castor bean (Ricinus communis, Euphorbiaceae). Micron 41:746–753. https://doi.org/10.1016/j.micron.2010.06.002
Wanzenböck E-M, Schöfer C, Schweizer D, Bachmair A (1997) Ribosomal transcription units integrated via T-DNA transformation associate with the nucleolus and do not require upstream repeat sequences for activity in Arabidopsis thaliana. Plant J 11:1007–1016
Acknowledgements
We thank Andrea Pedrosa-Harand from the Laboratory of Plant Cytogenetics and Evolution (Department of Botany, UFPE, Recife-PE, Brazil) for providing the chemicals and devices used in our PCR and Southern blot experiments.
Funding
This study was partially funded by the Brazilian National Council for Scientific and Technological Development (CNPq; PDJ 150912/2017-0) and the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES) - Finance Code 001.
Author information
Authors and Affiliations
Contributions
The study was conceived by A.C.B.V., E.V. and T.R. D.A.M provided the plant material. T.R. conducted bioinformatics, PCR and Southern blot experiments, while E.V. and S.S. assisted with BAC DNA isolation. FISH assays were performed by E.V. and J.R.M.F. A.C.B.V., D.A.M. and S.S. contributed to the discussion. T.R. wrote the paper with final approval from all authors.
Corresponding authors
Ethics declarations
Ethical approval
Not applicable.
Competing interests
Not applicable
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ribeiro, T., Vasconcelos, E., de Mendonça Filho, J.R. et al. Differential amplification of the subtelomeric satellite DNA JcSAT1 in the genus Jatropha L. (Euphorbiaceae). Genetica 152, 43–49 (2024). https://doi.org/10.1007/s10709-024-00204-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10709-024-00204-5