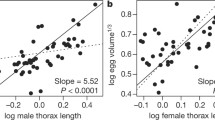
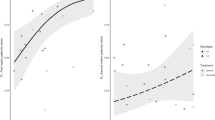
Abstract
As evidence mounts that male genitalia can affect relative fertilisation success, the role that sexual selection has played in the rapid and divergent evolution of genitalia is becoming increasingly recognized. Unfortunately, the limited functional understanding of these complex structures and their interactions with the female reproductive tract often limit interpretation regarding their evolution. Here, we address this issue using the earwig Euborellia brunneri, where both the male intromittent organ and the female spermatheca are highly exaggerated in length yet structurally simple. In a double mating design, we use the sterile male technique to study how sperm precedence patterns are affected by male genital length, male age, and the size of the male sperm storage organ, the seminal vesicle. Relative fertilisation success exhibited considerable variation around modest last-male paternity. Only an interaction between first and second male genital length affected paternity, where males gained reduced paternity when preceded by rivals with longer genitalia. Longer genitalia confer defensive benefits in sperm competition by apparently depositing ejaculate deeper in the tubular spermatheca, safe from removal by rivals. Paternity similarly depended on an interaction between the ages of both males, likely mediated by sperm traits as testes size decreased with age. Seminal vesicle size showed positive allometry but did not affect paternity; instead, greater seminal vesicle size in last males expedited oviposition. The exaggerated yet relatively simple genitalia of E. brunneri facilitate an unusually clear example of post-copulatory selection on phenotypic variation in multiple reproductive traits.



Similar content being viewed by others
References
Andrade C, Vieira R, Ananina G, Klaczko L (2009) Evolution of the male genitalia: morphological variation of the aedeagi in a natural population of Drosophila mediopunctata. Genetica 135:13–23
Andrés JA, Cordero Rivera A (2000) Copulation duration and fertilization success in a damselfly: an example of cryptic female choice? Anim Behav 59:695–703
Arnqvist G (2006) Sensory exploitation and sexual conflict. Philos Trans R Soc Lond B Biol Sci 361:375–386
Arnqvist G, Danielsson I (1999) Copulatory behavior, genital morphology, and male fertilization success in water striders. Evolution 53:147–156
Arnqvist G, Thornhill R (1998) Evolution of animal genitalia: patterns of phenotypic and genotypic variation and condition dependence of genital and non-genital morphology in water strider (Heteroptera: Gerridae: Insecta). Genet Res 71:193–212
Beck CW, Powell LA (2000) Evolution of female mate choice based on male age: are older males better mates? Evol Ecol Res 2:107–118
Bertin A, Fairbairn DJ (2007) The form of sexual selection on male genitalia cannot be inferred from within-population variance and allometry—a case study in Aquarius remigis. Evolution 61:825–837
Brooks R, Kemp DJ (2001) Can older males deliver the good genes? Trends Ecol Evol 16:308–313
Chapman T, Liddle LF, Kalb JM, Wolfner MF, Partridge L (1995) Cost of mating in Drosophila melanogaster females is mediated by male accessory gland products. Nature 373:241–244
Cook PA, Harvey IF, Parker GA (1997) Predicting variation in sperm precedence. Philos Trans R Soc Lond B Biol Sci 352:771–780
Cordero C, Eberhard WG (2005) Interaction between sexually antagonistic selection and mate choice in the evolution of female responses to male traits. Evol Ecol 19:111–122
Cordoba-Aguilar A (2002) Sensory trap as the mechanism of sexual selection in a damselfly genitalic trait (Insecta: calopterygidae). Am Nat 160:594–601
Eberhard WG (1985) Sexual selection and animal genitalia. Harvard University Press, Cambridge
Eberhard WG (1996) Female control: sexual selection by cryptic female choice. Princeton University Press, Princeton
Eberhard WG (2001) Species-specific genitalic copulatory courtship in sepsid flies (Diptera, Sepsidae, Microsepsis) and theories of genitalic evolution. Evolution 55:93–102
Eberhard WG (2004) Rapid divergent evolution of sexual morphology: comparative tests of antagonistic coevolution and traditional female choice. Evolution 58:1947–1970
Eberhard WG (2009a) Evolution of genitalia: theories, evidence, and new directions. Genetica 138:5–18
Eberhard WG (2009b) Static allometry and animal genitalia. Evolution 63:48–66
Edvardsson M, Canal D (2006) The effects of copulation duration in the bruchid beetle Callosobruchus maculatus. Behav Ecol 17:430–434
Edvardsson M, Tregenza T (2005) Why do male Callosobruchus maculatus harm their mates? Behav Ecol 16:788–793
Fisher RA (1930) The genetical theory of natural selection. Clarendon Press, Oxford
García-González F, Simmons LW (2005) Sperm viability matters in insect sperm competition. Curr Biol 15:271–275
Hosken DJ, Stockley P (2004) Sexual selection and genital evolution. Trends Ecol Evol 19:87–93
Hosken DJ, Garner TWJ, Tregenza T, Wedell N, Ward PI (2003) Superior sperm competitors sire higher-quality young. Proc R Soc Biol Sci Ser B 270:1933–1938
House CM, Simmons LW (2003) Genital morphology and fertilization success in the dung beetle Onthophagus taurus: an example of sexually selected male genitalia. Proc R Soc Lond B Biol Sci 270:447–455
Jamet C, Caussanel C (1995) Données biologiques, fonctionnement des appareils génitaux, comportements sexuels et maternels chez Euborellia annulipes (Lucas) (Dermaptère, Carcinophoridae). Bull Soc Entomol Fr 100:37–58
Jones TM, Balmford A, Quinnell RJ (2000) Adaptive female choice for middle-aged mates in a lekking sandfly. Proc R Soc Lond B Biol Sci 267:681–686
Kamimura Y (2000) Possible removal of rival sperm by the elongated genitalia of the earwig, Euborellia plebeja. Zool Sci 17:667–672
Kamimura Y (2005) Last-male paternity of Euborellia plebeja, an earwig with elongated genitalia and sperm-removal behavior. J Ethol 23:35–41
Kamimura Y, Iwase R (in press) Evolutionary genetics of genital size and lateral asymmetry in the earwig Euborellia plebeja (Dermaptera: anisolabididae). Biol J Linn Soc
Keller L, Reeve HK (1995) Why do females mate with multiple males? The sexually selected sperm hypothesis. Adv Study Behav 24:291–315
Kidd SA, Eskenazi B, Wyrobek AJ (2001) Effects of male age on semen quality and fertility: a review of the literature. Fertil Steril 75:237–248
Kocsis E, Trus BL, Steer CJ, Bisher ME, Steven AC (1991) Image averaging of flexible fibrous macromolecules : the clathrin triskelion has an elastic proximal segment. J Struct Biol 107:6–14
Kodric-Brown A, Sibly RM, Brown JH (2006) The allometry of ornaments and weapons. Proc Natl Acad Sci 103:8733–8738
Kokko H, Brooks R, Jennions MD, Morley J (2003) The evolution of mate choice and mating biases. Proc R Soc Lond B Biol Sci 270:653–664
Kuntner MA, Coddington JA, Schneider JM (2009) Intersexual arms race? Genital coevolution in nephilid spiders (Araneae, Nephilidae). Evolution 63:1451–1463
Lemos WP, Ramalho FS, Zanuncio JC (2003) Age-dependent fecundity and life-fertility tables for Euborellia annulipes (Lucas) (Dermaptera: Anisolabididae) a cotton boll weevil predator in laboratory studies with an artificial diet. Environ Entomol 32:592–601
Markow TA (1988) Drosophila males provide a material contribution to offspring sired by other males. Funct Ecol 2:77–79
Morrow EH, Arnqvist G, Pitnick S (2003) Adaptation versus pleiotropy: why do males harm their mates? Behav Ecol 14:802–806
Parker GA (1970) Sperm competition and its evolutionary consequences in insects. Biol Rev 45:525–567
Pizzari T, Dean R, Pacey A, Moore H, Bonsall MB (2008) The evolutionary ecology of pre- and post-meiotic sperm senescence. Trends Ecol Evol 23:131–140
Radesäter T, Halldórsdóttir H (1993) Two male types of the common earwig: male-male competition and mating success. Ethology 95:89–96
Radwan J (2003) Male age, germline mutations and the benefits of polyandry. Ecol Lett 6:581–586
Rankin SM, TeBrugge VA, Murray JA, Schuler AM, Tobe SS (2009) Effects of selected neuropeptides, mating status and castration on male reproductive tract movements and immunolocalization of neuropeptides in earwigs. Comp Biochem Physiol Part A Mol Integr Physiol 152:83–90
Schäfer M, Uhl G (2002) Determinants of paternity success in the spider Pholcus phalangioides (Pholcidae: Araneae): the role of male and female mating behaviour. Behav Ecol Sociobiol 51:368–377
Shapiro AM, Porter AH (1989) The lock-and-key hypothesis: evolutionary and biosystematic interpretation of insect genitalia. Annu Rev Entomol 34:231–245
Simmons LW (2001) Sperm competition and its evolutionary consequences in the insects. Princeton University Press, Princeton
Siva-Jothy MT (2000) The young sperm gambit. Ecol Lett 3:172–174
Stockley P (1997) Sexual conflict resulting from adaptations to sperm competition. Trends Ecol Evol 12:154–159
Stutt AD, Siva-Jothy MT (2001) Traumatic insemination and sexual conflict in the bed bug Cimex lectularius. Proc Natl Acad Sci 98:5683–5687
Takami Y, Sota T (2007) Rapid diversification of male genitalia and mating strategies in Ohomopterus ground beetles. J Evol Biol 20:1385–1395
van Lieshout E (in press) Male genital length and mating status differentially affect mating behaviour in an earwig. Behav Ecol Sociobiol
Walker WF (1980) Sperm utilization strategies in nonsocial insects. Am Nat 115:780–799
Wenninger EJ, Averill AL (2006) Influence of body and genital morphology on relative male fertilization success in oriental beetle. Behav Ecol 17:656–663
Werner M, Simmons LW (2008) The evolution of male genitalia: functional integration of genital sclerites in the dung beetle Onthophagus taurus. Biol J Linn Soc 93:257–266
Yamane T, Miyatake T, Kimura Y (2008) Female mating receptivity after injection of male-derived extracts in Callosobruchus maculatus. J Insect Physiol 54:1522–1527
Zuk M, Johnson K, Thornhill R, Ligon JD (1990) Mechanisms of female choice in red jungle fowl. Evolution 44:477–485
Acknowledgments
We thank the Holsworth Wildlife Research Endowment for the funding provided. We also thank David Hosken and the anonymous reviewers for their helpful comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
van Lieshout, E., Elgar, M.A. Longer exaggerated male genitalia confer defensive sperm-competitive benefits in an earwig. Evol Ecol 25, 351–362 (2011). https://doi.org/10.1007/s10682-010-9422-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-010-9422-1