Abstract
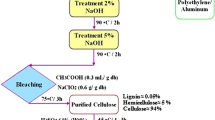
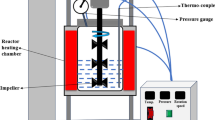
Water Hyacinth Leaf Protein Concentrate/Graphene Oxide (WHLPC/GO) hydrogel was synthesized for the removal of Cr(VI) from wastewater. About 90% of the prepared hydrogel constitutes WHLPC. The prepared material was characterized by FT-IR and XRD. The process variables such as pH, contact time, adsorbent dosage, initial Cr(VI) concentration, and temperature were optimized using a batch mode experiment. Kinetic studies were also conducted and it was observed that the chemosorptive pseudo-second-order best described the adsorption system with a correlation coefficient (R2) of 0.984. The highest adsorption capacity of 322.00 mg/g was achieved at pH 1.0, and equilibrium was achieved within 420 min. Various isotherm models were analyzed using non-linear fitting. It was found that the Sips model provides the best fit, indicating heterogeneous and uniform active site surface adsorption of Cr(VI) on the WHLPC/GO. The reuse efficiency of the synthesized material was also found to be greater than 84% for five consecutive cycles. Thermodynamic studies were conducted and results revealed that the adsorption was spontaneous and endothermic.







Similar content being viewed by others
Data availability
Raw data files are available from the corresponding author upon reasonable request.
References
Allam, F., Elnouby, M., El-Khatib, K. M., El-Badan, D. E., & Sabry, S. A. (2020). Water hyacinth (Eichhornia crassipes) biochar as an alternative cathode electrocatalyst in an air-cathode single chamber microbial fuel cell. International Journal of Hydrogen Energy, 45(10), 5911–5927.
Badessa, T. S., Wakuma, E., & Yimer, A. M. (2020). Bio-sorption for effective removal of chromium(VI) from wastewater using Moringa stenopetala seed powder (MSSP) and banana peel powder (BPP). BMC Chemistry, 14(1), 71.
Cao, J., He, G., Ning, X., Wang, C., Fan, L., Yin, Y., & Cai, W. (2021). Hydroxypropyl chitosan-based dual self-healing hydrogel for adsorption of chromium ions. International Journal of Biological Macromolecules, 174, 89–100.
Chavez, N. N. G., Ragaza, J. A., Corre, V. L., Serrano, A. E., & Traifalgar, R. F. M. (2016). Effects of water hyacinth ( Eichhornia crassipes) leaf protein concentrate as soybean protein replacement in white shrimp Litopenaeus vannamei (Boone) postlarvae diet. Aquaculture Research, 47(8), 2642–2649.
Dönmez, G., & Aksu, Z. (2002). Removal of chromium(VI) from saline wastewaters by Dunaliella species. Process Biochemistry, 38(5), 751–762.
Dutta, S., Srivastava, S. K., & Gupta, A. K. (2021). Polypyrrole–polyaniline copolymer coated green rice husk ash as an effective adsorbent for the removal of hexavalent chromium from contaminated water. Materials Advances, 2(7), 2431–2443.
Geng, J., Yin, Y., Liang, Q., Zhu, Z., & Luo, H. (2019). Polyethyleneimine cross-linked graphene oxide for removing hazardous hexavalent chromium: Adsorption performance and mechanism. Chemical Engineering Journal, 361, 1497–1510.
Ho, Y.-S. (2004). Selection of optimum sorption isotherm. Carbon, 42(10), 2115–2116.
Huang, Y., Li, S., Chen, J., Zhang, X., & Chen, Y. (2014). Adsorption of Pb(II) on mesoporous activated carbons fabricated from water hyacinth using H3PO4 activation: Adsorption capacity, kinetic and isotherm studies. Applied Surface Science, 293, 160–168.
Jia, B., Li, G., Cao, E., Luo, J., Zhao, X., & Huang, H. (2023). Recent progress of antibacterial hydrogels in wound dressings. Materials Today Bio, 19, 100582.
Khalil, U., Shakoor, M. B., Ali, S., Ahmad, S. R., Rizwan, M., Alsahli, A. A., & Alyemeni, M. N. (2021). Selective removal of hexavalent chromium from wastewater by rice husk: Kinetic, isotherm and spectroscopic investigation. Water, 13(3), 263.
Kokab, T., Ashraf, H. S., Shakoor, M. B., Jilani, A., Ahmad, S. R., Majid, M., Ali, S., et al. (2021). Effective removal of Cr(VI) from wastewater using biochar derived from walnut shell. International Journal of Environmental Research and Public Health, 18(18), 9670.
Kong, D., He, L., Li, H., Zhang, F., & Song, Z. (2021). Preparation and characterization of graphene oxide/chitosan composite aerogel with high adsorption performance for Cr(VI) by a new crosslinking route. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 625, 126832.
Kouotou, D., Ghalit, M., Ndi, J. N., Martinez, L. M. P., Ouahabi, M. E., Ketcha, J. M., & Gharibi, E. K. (2021). Removal of metallic trace elements (Pb2+, Cd2+, Cu2+, and Ni2+) from aqueous solution by adsorption onto cerium oxide modified activated carbon. Environmental Monitoring and Assessment, 193(8), 467.
Kumar, P., & Chauhan, M. S. (2019). Adsorption of chromium (VI) from the synthetic aqueous solution using chemically modified dried water hyacinth roots. Journal of Environmental Chemical Engineering, 7(4), 103218.
Kwak, H. W., Lee, H., & Lee, K. H. (2020). Surface-modified spherical lignin particles with superior Cr(VI) removal efficiency. Chemosphere, 239, 124733.
Lu, Z., Zhang, H., Shahab, A., Zhang, K., Zeng, H., Bacha, A.-U.-R., Nabi, I., et al. (2021). Comparative study on characterization and adsorption properties of phosphoric acid activated biochar and nitrogen-containing modified biochar employing Eucalyptus as a precursor. Journal of Cleaner Production, 303, 127046.
Mekonnen, E., Yitbarek, M., & Soreta, T. R. (2015). Kinetic and thermodynamic studies of the adsorption of Cr(VI) onto some selected local adsorbents. South African Journal of Chemistry, 68, 45–52 Retrieved May 23, 2023, from http://ref.scielo.org/fdww4r
Naseem, T., Bibi, F., Arif, S., Waseem, M., Haq, S., Azra, M. N., Liblik, T., et al. (2022). Adsorption and kinetics studies of Cr (VI) by graphene oxide and reduced graphene oxide-zinc oxide nanocomposite. Molecules, 27(21), 7152.
Parameswari, E., Premalatha, R. P., Davamani, V., & Sebastian, S. P. (2021). Biosequestration of chromium (VI) from aqueous medium using carbonaceous adsorbents derived from Eichhornia crassipes (preprint). In Review. Retrieved May 23, 2023, from https://www.researchsquare.com/article/rs-595032/v1
Rajeshwari, K., Latha, S., Gomathi, T., Sangeetha, K., & Sudha, P. N. (2018). Adsorption of heavy metal Cr (VI) by a ternary biopolymer blend. Materials Today: Proceedings, 5(6), 14628–14638.
Ren, L., Xu, J., Zhang, Y., Zhou, J., Chen, D., & Chang, Z. (2019). Preparation and characterization of porous chitosan microspheres and adsorption performance for hexavalent chromium. International Journal of Biological Macromolecules, 135, 898–906.
Sahoo, S. K., Panigrahi, G. K., Sahoo, J. K., Pradhan, A. K., Purohit, A. K., & Dhal, J. P. (2021). Electrospun magnetic polyacrylonitrile-GO hybrid nanofibers for removing Cr(VI) from water. Journal of Molecular Liquids, 326, 115364.
Sahoo, S. K., Panigrahi, G. K., Sahu, M. K., Arzoo, A., Sahoo, J. K., Sahoo, A., Pradhan, A. K., et al. (2022). Biological synthesis of GO-MgO nanomaterial using Azadirachta indica leaf extract: A potential bio-adsorbent for removing Cr(VI) ions from aqueous media. Biochemical Engineering Journal, 177, 108272.
Samuel, M. S., Bhattacharya, J., Raj, S., Santhanam, N., Singh, H., & Pradeep Singh, N. D. (2019). Efficient removal of Chromium(VI) from aqueous solution using chitosan grafted graphene oxide (CS-GO) nanocomposite. International Journal of Biological Macromolecules, 121, 285–292.
Shang, M., Liu, Y., Liu, S., Zeng, G., Tan, X., Jiang, L., Huang, X., et al. (2016). A novel graphene oxide coated biochar composite: Synthesis, characterization and application for Cr( vi ) removal. RSC Advances, 6(88), 85202–85212.
Sharma, N., Sharma, V., Jain, Y., Kumari, M., Gupta, R., Sharma, S. K., & Sachdev, K. (2017). Synthesis and characterization of graphene oxide (GO) and reduced graphene oxide (rGO) for gas sensing application. Macromolecular Symposia, 376(1), 1700006.
Song, Z., Li, W., Liu, W., Yang, Y., Wang, N., Wang, H., & Gao, H. (2015). Novel magnetic lignin composite sorbent for chromium(vi) adsorption. RSC Advances, 5(17), 13028–13035.
Sun, Y., Liu, X., Lv, X., Wang, T., & Xue, B. (2021). Synthesis of novel lignosulfonate-modified graphene hydrogel for ultrahigh adsorption capacity of Cr(VI) from wastewater. Journal of Cleaner Production, 295, 126406.
Vilela, P. B., Dalalibera, A., Duminelli, E. C., Becegato, V. A., & Paulino, A. T. (2019). Adsorption and removal of chromium (VI) contained in aqueous solutions using a chitosan-based hydrogel. Environmental Science and Pollution Research, 26(28), 28481–28489.
Yan, Z., Wu, T., Fang, G., Ran, M., Shen, K., & Liao, G. (2021). Self-assembly preparation of lignin–graphene oxide composite nanospheres for highly efficient Cr(vi) removal. RSC Advances, 11(8), 4713–4722.
Yang, J., Yu, M., & Chen, W. (2015). Adsorption of hexavalent chromium from aqueous solution by activated carbon prepared from longan seed: Kinetics, equilibrium and thermodynamics. Journal of Industrial and Engineering Chemistry, 21, 414–422.
Yifru, A., Mekonnen, N., Mehretie, S., & Admassie, S. (2022). Polypyrrole–polyaniline-water hyacinth leaf protein concentrate composite for the removal of Cr(VI) from aqueous solution: Kinetics, isotherm and thermodynamic studies. Bulletin of the Chemical Society of Ethiopia, 36(3), 571–584.
Yu, P., Wang, H.-Q., Bao, R.-Y., Liu, Z., Yang, W., Xie, B.-H., & Yang, M.-B. (2017). Self-assembled sponge-like chitosan/reduced graphene oxide/montmorillonite composite hydrogels without cross-linking of chitosan for effective Cr(VI) sorption. ACS Sustainable Chemistry & Engineering, 5(2), 1557–1566.
Yuan, R., Yuan, J., Wu, Y., Ju, P., Ji, L., Li, H., Chen, L., et al. (2018). Graphene oxide-monohydrated manganese phosphate composites: Preparation via modified Hummers method. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 547, 56–63.
Yusuff, A. S. (2019). Adsorption of hexavalent chromium from aqueous solution by Leucaena leucocephala seed pod activated carbon: Equilibrium, kinetic and thermodynamic studies. Arab Journal of Basic and Applied Sciences, 26(1), 89–102.
Zhang, L., Luo, H., Liu, P., Fang, W., & Geng, J. (2016). A novel modified graphene oxide/chitosan composite used as an adsorbent for Cr(VI) in aqueous solutions. International Journal of Biological Macromolecules, 87, 586–596.
Zhang, M., Liu, Y., Li, T., Xu, W., Zheng, B., Tan, X., Wang, H., et al. (2015). Chitosan modification of magnetic biochar produced from Eichhornia crassipes for enhanced sorption of Cr(vi) from aqueous solution. RSC Advances, 5(58), 46955–46964.
Zhou, J.-M., Jiang, Z.-C., Qin, X.-Q., Zhang, L.-K., Huang, Q.-B., Xu, G.-L., & Dionysiou, D. D. (2020). Efficiency of Pb, Zn, Cd, and Mn removal from karst water by Eichhornia crassipes. International Journal of Environmental Research and Public Health, 17(15), 5329.
Funding
This work was supported by the International Science Program (ISP), Uppsala University, Sweden.
Author information
Authors and Affiliations
Contributions
Asmamaw Taye; Conceived and designed the experiments; Performed the experiments; Analyzed and interpreted the data; Contributed reagents, materials, analysis tools or data; Wrote the paper. Solomon Mehretie, Shimelis Admassie, Alemayehu Yifru: Conceived and designed the experiments; Analyzed and interpreted the data; Contributed reagents, materials, analysis tools or data; Wrote the paper. Negash Getachew runs the XRD experiments.
Corresponding author
Ethics declarations
All authors have read, understood, and complied as applicable with the statement on “Ethical responsibilities of Authors” as found in the Instructions for Authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Taye, A., Yifru, A., Getachew, N. et al. Adsorption of hexavalent chromium using Water Hyacinth Leaf Protein Concentrate/Graphene Oxide hydrogel. Environ Monit Assess 195, 1342 (2023). https://doi.org/10.1007/s10661-023-11960-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-023-11960-w