Abstract
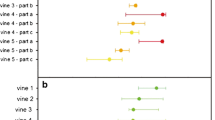
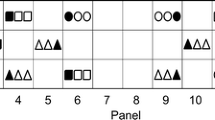
Several vineyards in Rías Baixas and one in the Ribeira Sacra (Spain) were monitored and the spatial pattern of leafroll-diseased grapevine was analysed at several dates. Unidimensional aggregation analysis (ordinary runs), bidimensional analysis, and disease gradients analysis were used as methods of study of spatial aspects of epidemics of GLRaV. At very low insect populations the mealybug Planococcus citri transmitted the GLRaV-3 from infected plants to healthy ones planted between them in an experimental plot at Beluso; an initial focus of leafroll-infected grapevines was detected 2 years after planting at the area where the vectors had been located infesting the old plants. Within 8 years some new foci appeared and coalesced, and the incidence reached >80%. In three commercial plots where no vectors were observed, the spatial analysis of the diseased plants showed three different situations. In Meaño, the study of the evolution of the spatial patterns of diseased plants between 1992 and 2005 suggested slow vectorial field transmission of GLRaV-3. In Goián the analysis for only 2 years suggested random distribution; therefore the viruses were arriving with the planting material, but the runs analysis of some lines suggested incipient spread of GLRaV-3. In Portomarín the incidence of both GLRaV-2 and 3 was low and their distribution was random, without any evidence of field spread. These examples of the study of the spatial analysis of leafroll-infected plants may be helpful to determine whether or not spread of the viruses is occurring, and the best control measures to take.




Similar content being viewed by others
References
Bonfiglioli, T., Edwards, F., Hoskins, N., & Pantaleo, A. (2003). Graft incompatibility syndrome in New Zealand Merlot vines involves another possible variant of GLRaV-2. The Australian and New Zealand Grapevine Grower and Winemaker, 472, 87–90.
Cabaleiro, C., & Segura, A. (1997a). Some characteristics of the transmission of grapevine leafroll associated virus 3 (GLRaV-3) by Planococcus citri Risso. European Journal of Plant Pathology, 103, 373–378.
Cabaleiro, C., & Segura, A. (1997b). Field transmission of Grapevine leafroll associated virus 3 by Planococcus citri Risso. Plant Disease, 81, 283–287.
Cabaleiro, C., & Segura, A. (2006). Temporal analysis of grapevine leafroll associated virus 3 (GLRaV-3) epidemics. European Journal of Plant Pathology, 114, 441–446.
Cabaleiro, C., Segura, A., & García-Berrios, J. (1999). Effect of grapevine leafroll associated virus 3 on the physiology and must of Vitis vinifera L. cv. Albariño following contamination in the field. American Journal of Enology and Viticulture, 50(1), 40–44.
Cid, M., Pereira, S., Faoro, F., Cabaleiro, C., & Segura, A. (2007). Presence of GLRaV-3 on the primary salivary glands of P. citri suggest a circulative transmission mechanism. European Journal of Plant Pathology, 118(1), 23–30.
Cid, M., Pereira, S., Gago, P., Couceiro, C., Cabaleiro, C., & Segura, A. (2006). Planococcus citri (Risso) (Homoptera: Pseudococcidae) as GLRaV-3 vector. Extended abstracts 15th ICVG, South Africa, 3–7 April 2006, 252–253.
Cohen, D., van den Brink, R., & Habili, N. (2004). Detection of leafroll virus in newly infected grapevines. The Australian and New Zealand Grapevine Grower and Winemaker. Annual Technical Issue 2004, 56–59.
Couceiro, C., Cid, M., Pereira, S., Segura, A., & Cabaleiro, C. (2006). Use of direct immunoprinting (DIP)-ELISA for the detection of grapevine leafroll viruses. Extended abstracts 15th ICVG, South Africa, 3–7 April 2006, 180–181.
Dallot, S., Gottwald, T., Labonne, G., & Quiot, J. B. (2003). Spatial pattern analysis of Sharka disease (Plum pox virus strain M) in peach orchards of southern France. Phytopathology, 93, 1543–1552.
Dimitrijevic, B. (1973). Some observations on natural spread of grapevine leafroll disease in Yugoslavia. Rivista di Patologia Vegetale (S.IV) 9, 114–119.
Engelbrecht, D. J., & Kasdorf, G. G. F. (1990). Field spread of corky bark, fleck, leafroll and Shiraz decline diseases and associated viruses in South African grapevines. Phytophylactica, 22(3), 347–354.
Epstein, A. H. (1978). Root graft transmission of tree pathogens. Annual Review of Phytopathology, 16, 181–192.
Gottwald, T. R., Cambra, M., Moreno, P., Camarasa, E., & Piquer, J. (1996). Spatial and temporal analyses of Citrus tristeza in eastern Spain. Phytopathology, 80, 45–55.
Gugerli, P. (2003). Grapevine leafroll and related viruses. Extended abstracts 14th Meeting ICVG, Locorotondo (Bari), Italia, 12–17 September, 2003, 25–31.
Habili, N., Fazeli, C. F., Ewart, A., Hamilton, R., Cirami, R., Saldarelli, P., et al. (1995). Natural spread and molecular analysis of grapevine leafroll-associated virus 3 in Australia. Phytopathology, 285, 1418–1422.
Jeger, M. J., Holt, J., Bosch, F., & Madden, L. V. (2004). Epidemiology of insect-transmitted plant viruses: Modelling disease dynamics and control interventions. Physiological Entomology, 29, 291–304.
Jordan, D. (1993). Leafroll spread in New Zealand vineyards. Australian and New Zealand Wine Industry Journal, 8(4), 322–324.
Krüger, K., Saccaggi, D., & Douglas, N. (2006). Grapevine leafroll associated virus 3- vector interactions: Transmission by the mealybugs Planococcus ficus and Pseudococcus longispinus (Hemiptera: Pseudococcidae). Extended abstracts 15th ICVG, South Africa, 3–7 April 2006, 130–131.
Madden, L. V. (2006). Botanical epidemiology: some key advances and its continuing role in disease management. European Journal of Plant Pathology, 115, 3–23.
Madden, L. V., Hughes, G., & van den Bosh, F. (2007). The study of plant disease epidemics. APS, St. Paul, Minesotta, USA
Madden, L. V., Louie, R., Abt, J. J., & Knoke, J. K. (1982). Evaluation of tests for randomness of infected plants. Phytopathology, 72, 195–198.
Madden, L. V., Pirone, T. P., & Raccah, B. (1987). Analysis of spatial patterns of virus-diseased tobacco plants. Phytopathology, 77(10), 1409–1417.
Maixner, M. (1993). PATCHY – Ein programm zur analyse rŠumlicher verteilungsmuster von Rebkrankheiten. Nachrichtenbl Deut Pflanzenschutzd, 45, 157–164.
Maixner, M., & Reinert, W. (1997). Spatio-temporal analysis of the distribution of grapevine yellows in Germany. Extended abstracts 12th Meeting ICVG, Lisbon, Portugal, 29 September–2 October 1997, 75–76.
Martin, R. R., Eastwell, K. C., Wagner, A., Lamprecht, I., & Tzanetakis, I. E. (2005). Survey for viruses of grapevine in Oregon and Washington. Plant Disease, 89, 763–766.
Monis, J., & Bestwick, R. K. (1996). Detection and localization of grapevine leafroll associated closteroviruses in greenhouse and tissue culture grown plants. American Journal of Enology and Viticulture, 47(2), 199–205.
Pietersen, G. (2006). Spatio-temporal distribution dynamics of Grapevine leafroll disease in Western Cape vineyards. Extended abstracts 15th ICVG, South Africa, 3–7 April 2006, 126–127.
Pietersen, G., Kellerman, U., Oosthulzen, W. T., & Spreeth, N. (2003). Effect of roguing on natural spread of grapevine leafroll disease in two vineyards in South Africa. 8th International Congress of Plant Pathology. Christchurch, New Zealand, 2–7 February 2003. pp 301.
Sether, D. M., Ullman, D. E., & Hu, J. S. (1998). Transmission of pineapple mealybug Wilt-associated virus by two species of mealybug (Dysmicoccus spp.). Phytopathology, 88, 1224–1230.
Tsitsipis, J. A., Angelakis, E., Margaritopoulos, J. T., Tsamandaini, K., & Zarpas, K. D. (2005). First record of the grapevine aphid Aphis illinoisensis in the island of Kriti, Greece. OEPP/EPPO. Bulletin, 35, 541–542.
Acknowledgements
This work was partly funded by grants from the Xunta de Galicia (XUGA200074A93 and PGIDITO3PXIC20001PN), the European Commission (ALVIRUS/SP1.P7), the FEDER and Ministerio de Ciencia y Tecnología (AGL2002-02438), and the Instituto Nacional de Investigación y Tecnología Agraria y Alimentaria (RTA2005-00201-C03-03).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cabaleiro, C., Couceiro, C., Pereira, S. et al. Spatial analysis of epidemics of Grapevine leafroll associated virus-3 . Eur J Plant Pathol 121, 121–130 (2008). https://doi.org/10.1007/s10658-007-9254-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-007-9254-1