Abstract
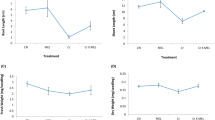
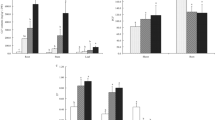
To evaluate the ameliorating effects of hydrogen peroxide (H2O2, 200 μM) on hexavalent chromium [Cr(VI)] toxicity in canola (Brassica napus L.), we focused on the plant growth, chlorophyll content, thiol contents, lipid peroxidation, antioxidant enzymes, and the expression of metallothionein protein (BnMP1) mRNA. Cr(VI) at 50 μM significantly decreased the plant growth (fresh and dry weights). The decrease in growth was accompanied by increased lipid peroxidation and decreased chlorophyll content in leaves. Hydrogen peroxide pretreatment, however, enhanced plant growth parameters and led to the reduced levels of lipid peroxidation and higher levels of pigment. In addition, H2O2 pretreatment increased Cr accumulation in aerial parts of seedlings. The tendency of increase in thiol content under Cr(VI) stress was further increased with H2O2 pretreatment. The activities of antioxidant enzymes such as superoxide dismutase (SOD), ascorbate peroxidase (APX), guaiacol peroxidase (POD) and catalase (CAT) were differentially altered. SOD and POD activities increased under Cr(VI) stress, whereas APX and CAT activities decreased. The SOD and CAT activities remained unaffected in both durations due to H2O2 pretreatment, but activities of APX and POD were promoted in the Cr(VI)-stressed seedlings. Metallothioneins are a family of low-molecular-weight Cys-rich proteins and are thought to play a possible role in metal metabolism or detoxification. In real-time quantitative PCR analysis, the expression level of BnMP1 mRNA was increased at 1 day after treatment (DAT), whereas it was decreased at 7 DAT in Cr(VI)-stressed seedlings. At 1 DAT, pretreatment of H2O2 before Cr(VI) stress reduced the expression of BnMP1 mRNA as compared to Cr(VI) stress alone, but this effect was not significant. At 7 DAT, H2O2 pretreatment alleviated the Cr(VI) stress-mediated decrease in the expression of BnMP1 mRNA. These results suggest that H2O2 may act as a signal that triggers defense mechanisms which in turn protects canola seedlings from Cr(VI)-induced oxidative damage.






Similar content being viewed by others
References
Aebi H (1984) Catalase in vitro. Method Enzymol 105:121–126
Asada K (1992) Ascorbate peroxidase-a hydrogen peroxide scavenging enzyme in plants. Physiol Plant 85:235–241
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287
Becquer T, Quantin C, Sicot M, Boudot JP (2003) Chromium availability in ultramafic soils from New Caledonia. Sci Total Environ 301:251–261
Chance B, Maehly AC (1955) Assay of catalase and perxoidase. In: Colowick SP, Kaplan NO (eds) Methods in enzymology, 2nd edn. Academic Press, New York, pp 764–775
Chao YY, Chen CY, Huang WD, Kao CH (2010) Salicylic acid-mediated hydrogen peroxide accumulation and protection against Cd toxicity in rice leaves. Plant Soil 329:327–337
Chen L, Ren F, Zhong H, Jiang W, Li X (2010) Identification and expression analysis of genes in response to high-salinity and drought stresses in Brassica napus. Acta Biochim Biophys Sin 42:154–164
Cobbett CS (2000) Phytochelatins and their roles in heavy metal detoxification. Plant Physiol 123:825–832
Cobbett C, Goldsbrough P (2002) Phytochelatins and metallothioneins: roles in heavy metal detoxification and homeostasis. Annu Rev Plant Biol 53:159–182
Dube BK, Tewari K, Chatterjee J, Chatterjee C (2003) Excess chromium alters uptake and translocation of certain nutrients in citrullus. Chemosphere 53:1147–1153
Fargašová A (2012) Plants as models for chromium and nickel risk assessment. Ecotoxicology 21:1476–1483
Foyer CH, Looez-Delgado H, Dat JF, Scott IM (1997) Hydrogen peroxide and glutathione-associated mechanisms of acclamatory stress tolerance and signaling. Physiol Plant 100:241–254
Goupil P, Souguir D, Ferjani E, Faure O (2009) Expression of stress-related genes in tomato plants exposed to arsenic and chromium in nutrient solution. J Plant Physiol 166:1446–1452
Harmens H, Den Hartog PR, ten Bookum WM, Verkleij JAC (1993) Increased zinc tolerance in Silene vulgaris (Moench) Garcke is not due to increased production of phytochelatins. Plant Physiol 103:1305–1309
Hassinen VH, Tervahauta I, Schat H, Kärenlampi SO (2011) Plant metallothioneins-metal chelators with ROS scavenging activity? Plant Biol 13:225–232
Hayat S, Khalique G, Irfan M, Wani AS, Tripathi BN, Ahmad A (2012) Physiological changes induced by chromium stress in plants: an overview. Protoplasma 249:599–611
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198
Hernández-Allica J, Becerril JM, Garbisu C (2008) Assessment of the phytoextraction potential of high biomass crop plants. Environ Pollut 152:32–40
Hsu YT, Kao CH (2007) Heat shock-mediated H2O2 accumulation and protection against Cd toxicity in rice seedlings. Plant Soil 300:137–147
Hu Y, Ge Y, Zhang C, Ju T, Cheng W (2009) Cadmium toxicity and translocation in rice seedlings are reduced by hydrogen peroxide pretreatment. Plant Growth Regul 59:51–61
Huang G-Y, Wang Y-S, Ying GG (2011) Cadmium-inducible BgMT2, a type 2 metallothionein gene from mangrove species (Bruguiera gymnorrhiza), its encoding protein shows metal-binding ability. J Exp Mar Biol Ecol 405:128–132
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7:405–410
Nagalakshmi N, Prasad MNV (2001) Responses of glutathione cycle enzymes and glutathione metabolism to copper stress in Scenedesmus bijugatus. Plant Sci 160:291–299
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880
Navabpour S, Morris K, Allen R, Harrison E, A-H-Mackerness S, Buchanan-Wollaston V (2003) Expression of senescence-enhanced genes in response to oxidative stress. J Exp Bot 54:2285–2292
Noctor G, Foyer CH (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Physiol Plant Mol Biol 49:249–279
Prasad TK, Anderson MD, Martin BA, Stewart CR (1994) Evidence for chilling-induced oxidative stress in maize seedlings and a regulatory role for hydrogen peroxide. Plant Cell 6:65–74
Shams KM, Tichy G, Fischer A, Sager M, Peer T, Bashar A, Filip K (2010) Aspects of phytoremediation for chromium contaminated sites using common plants Urtica dioica, Brassica napus and Zea mays. Plant Soil 328:175–189
Shanker AK, Cervantes C, Loza-Tavera H, Avudainayagam S (2005) Chromium toxicity in plants. Environ Int 31:739–753
Uchida A, Jagendorf AT, Hibino T, Takabe T, Takabe T (2002) Effects of hydrogen peroxide and nitric oxide on both salt and heat stress tolerance in rice. Plant Sci 163:515–523
Vajpayee P, Tripathi RD, Rai UN, Ali MB, Singh SN (2000) Chromium(VI) accumulation reduces chlorophyll biosynthesis, nitrate reductase activity and protein content in Nymphaea alba L. Chemosphere 41:1075–1082
Wellburn AR (1994) The spectral determination of chlorophylls a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. J Plant Physiol 144:307–313
Xu Q, Xu X, Zhao Y, Jiao K, Herbert SJ, Hao L (2008) Salicylic acid, hydrogen peroxide and calcium-induced saline tolerance associated with endogenous hydrogen peroxide homeostasis in naked oat seedlings. Plant Growth Regul 54:249–259
Yu R, Ji J, Yuan X, Song Y, Wang C (2012) Accumulation and translocation of heavy metals in the canola (Brassica napus L.)–soil system in Yangtze River Delta China. Plant Soil 353:33–45
Zhang H, Lian C, Shen Z (2009) Proteomic identification of small, copper-responsive proteins in germinating embryos of Oryza sativa. Ann Bot 103:923–930
Zhang FQ, Wang YS, Sun CC, Lou ZP, Dong JD (2012) A novel metallothionein gene from a mangrove plant Kandelia candel. Ecotoxicology 21:1633–1641
Acknowledgments
The authors wish to thank Afyon Kocatepe University Research Fund (Project Nos. 11.FENED.02 and 11.FEN.BIL.14) for financial support. The authors also wish to thank Afyon Kocatepe University’s Foreign Language Support Unit for some language editing.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yıldız, M., Terzi, H. & Bingül, N. Protective role of hydrogen peroxide pretreatment on defense systems and BnMP1 gene expression in Cr(VI)-stressed canola seedlings. Ecotoxicology 22, 1303–1312 (2013). https://doi.org/10.1007/s10646-013-1117-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-013-1117-2