Summary
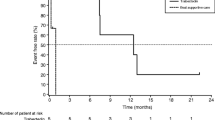
Currently, there is no gold standard treatment for Extraskeletal Myxoid Chondrosarcomas (EMC) making wide margin surgical resection the most effective alternative treatment. Nevertheless, in previous preclinical studies our lab demonstrated the potential of the hypoxia-activated prodrug (HAP) ICF05016 on EMC murine model inoculated with the H-EMC-SS human cell line. The aim of this study was to assess, in vivo, the relevance of the combination of this HAP with External Beam Radiotherapy (EBR). Firstly EMC-bearing mice were treated with 6 Gy or 12 Gy of EBR (single 6 MV photon). Then for combination of HAP and EBR, animals received 6 doses of ICF05016 (46.8 μmol/kg, intravenously) at 4-day intervals, with 6 Gy EBR performed 24 h after the 3rd dose of HAP. Animals were monitored throughout the study for clinical observations (tumour growth, side effects) and survival studies were performed. From tumour samples, PCNA, Ki-67 and p21 expressions were used as markers of proliferation and cell cycle arrest. Statistical significances were determined using Kruskall-Wallis and log rank tests. The radiosensitivity of the EMC model was demonstrated at 12 Gy with significant inhibition of tumour growth. Then, the HAP strategy potentiated EBR efficacy at a lower dose (6 Gy) by improving survival without generating side effects. Thus, results of this study showed the potential interest of ICF05016 for the combination with EBR in the management of EMC.





Similar content being viewed by others
Data availability
The datasets used and/or analysed during this study are available from the corresponding author on request.
Abbreviations
- AUC :
-
Area Under the Curve
- EBR :
-
External Beam Radiotherapy
- EMC :
-
Extraskeletal Myxoid Chondrosarcomas
- HAP :
-
Hypoxia-Activated Prodrug
- IV :
-
Intravenous
- SC :
-
Subcutaneously
- SCID :
-
Severe Combined Immunodeficiency
- SD :
-
Standard Deviation
- STS :
-
Soft Tissue Sarcomas
- TGI :
-
Tumour Growth Inhibition
- TSA :
-
Tyramide Signal Amplification
References
Aigner T, Oliveira AM, Nascimento AG (2004) Extraskeletal myxoid chondrosarcomas do not show a chondrocytic phenotype. Mod Pathol 17(2):214–221
Sasbou Y, Rhanim A, Mhammdi Y, Nkaoui M, El Bardouni A, Berrada MS et al (2015) Chondrosarcome myxoïde extra-osseux: à propos d’un cas et revue de la littérature. Pan Afr Med J 20:360
Davis EJ, Wu Y-M, Robinson D, Schuetze SM, Baker LH, Athanikar J, Cao X, Kunju LP, Chinnaiyan AM, Chugh R (2017) Next generation sequencing of extraskeletal myxoid chondrosarcoma. Oncotarget 8(13):21770–21777
Elsayed AG, Al-Qawasmi L, Katz H, Lebowicz Y (2018) Extraskeletal Chondrosarcoma: long-term follow-up of a patient with metastatic disease. Cureus 10(5):e2709
Drilon AD, Popat S, Bhuchar G, D’Adamo DR, Keohan ML, Fisher C et al (2008) Extraskeletal myxoid chondrosarcoma: a retrospective review from 2 referral centers emphasizing long-term outcomes with surgery and chemotherapy. Cancer 113(12):3364–3371
Kawaguchi S, Wada T, Nagoya S, Ikeda T, Isu K, Yamashiro K, Kawai A, Ishii T, Araki N, Myoui A, Matsumoto S, Umeda T, Yoshikawa H, Hasegawa T, Multi-Institutional Study of 42 Cases in Japan (2003) Extraskeletal myxoid chondrosarcoma: a multi-institutional study of 42 cases in Japan. Cancer 97(5):1285–1292
Ogura K, Fujiwara T, Beppu Y, Chuman H, Yoshida A, Kawano H, Kawai A (2012) Extraskeletal myxoid chondrosarcoma: a review of 23 patients treated at a single referral center with long-term follow-up. Arch Orthop Trauma Surg 132(10):1379–1386
Stacchiotti S, Pantaleo MA, Astolfi A, Dagrada GP, Negri T, Dei Tos AP, Indio V, Morosi C, Gronchi A, Colombo C, Conca E, Toffolatti L, Tazzari M, Crippa F, Maestro R, Pilotti S, Casali PG (2014) Activity of sunitinib in extraskeletal myxoid chondrosarcoma. Eur J Cancer 50(9):1657–1664
Morioka H, Takahashi S, Araki N, Sugiura H, Ueda T, Takahashi M et al (2016) Results of sub-analysis of a phase 2 study on trabectedin treatment for extraskeletal myxoid chondrosarcoma and mesenchymal chondrosarcoma. BMC Cancer 16:479
Kemmerer EJ, Gleeson E, Poli J, Ownbey RT, Brady LW, Bowne WB (2018) Benefit of radiotherapy in Extraskeletal Myxoid Chondrosarcoma: a propensity score weighted population-based analysis of the SEER database. Am J Clin Oncol 41(7):674–680
Mery B, Espenel S, Guy J-B, Rancoule C, Vallard A, Aloy M-T, Rodriguez-Lafrasse C, Magné N (2018) Biological aspects of chondrosarcoma: leaps and hurdles. Crit Rev Oncol Hematol 126:32–36
Peeters SGJA, Zegers CML, Biemans R, Lieuwes NG, van Stiphout RGPM, Yaromina A, Sun JD, Hart CP, Windhorst AD, van Elmpt W, Dubois LJ, Lambin P (2015) TH-302 in combination with radiotherapy enhances the therapeutic outcome and is associated with pretreatment [18F]HX4 hypoxia PET imaging. Clin Cancer Res 21(13):2984–2992
Voissiere A, Weber V, Gerard Y, Rédini F, Raes F, Chezal J-M, Degoul F, Peyrode C, Miot-Noirault E (2017) Proteoglycan-targeting applied to hypoxia-activated prodrug therapy in chondrosarcoma: first proof-of-concept. Oncotarget 8(56):95824–95840
Jackson RK, Liew LP, Hay MP (2019) Overcoming Radioresistance: small molecule Radiosensitisers and hypoxia-activated Prodrugs. Clin Oncol (R Coll Radiol) 31(5):290–302
Mistry IN, Thomas M, Calder EDD, Conway SJ, Hammond EM (2017) Clinical advances of hypoxia-activated Prodrugs in combination with radiation therapy. Int J Radiat Oncol Biol Phys 98(5):1183–1196
Hajj C, Russell J, Hart CP, Goodman KA, Lowery MA, Haimovitz-Friedman A, Deasy JO, Humm JL (2017) A combination of radiation and the hypoxia-activated Prodrug Evofosfamide (TH-302) is efficacious against a human Orthotopic pancreatic tumor model. Transl Oncol 10(5):760–765
https://cancer.sanger.ac.uk/cell_lines/sample/overview?id=907290, accessed 29 July 2020
Nytko KJ, Grgic I, Bender S, Ott J, Guckenberger M, Riesterer O et al (2017) The hypoxia-activated prodrug evofosfamide in combination with multiple regimens of radiotherapy. Oncotarget 8(14):23702–23712
Acknowledgements
The authors thank Soubeyrand-Damon Christelle for technical assistance and the ANIPATH platform (UMR 6293 CNRS / U1103 INSERM GReD, Clermont-Ferrand, France) for the immunohistochemical studies.
Funding
The work was supported by the PRT-k Translational Cancer Research INCa-DGOS program (D-TECT project).
Author information
Authors and Affiliations
Contributions
All authors read and approved the manuscript.
Biology experimentations: ME, VA, DV, CE.
Drug synthesis: GY, CJM, WV.
Head of team: DF, MNE.
Methodology: ME, CE, MNE.
Supervision: CE, MNE.
First writing: ME, WV, CE, MNE.
Corresponding author
Ethics declarations
Conflict of interest
Elise Maubert declares that she has no conflict of interest.
Valérie Weber declares that she has no conflict of interest.
Aurélien Voissière declares that he has no conflict of interest.
Yvain Gérard declares that he has no conflict of interest.
Véronique Dedieu declares that she has no conflict of interest.
Françoise Degoul declares that she has no conflict of interest.
Jean-Michel Chezal declares that he has no conflict of interest.
Emmanuel Chautard declares that he has no conflict of interest.
Elisabeth Miot-Noirault declares that she has no conflict of interest.
Ethics approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All animal studies were carried out in accordance with directive 2010/63/EU after approval by the institutional review board (C2E2A, authorization #9954).
Informed consent
For this type of study, formal consent is not required.
Code availability
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Maubert, E., Weber, V., Voissière, A. et al. Relevance of the combination of external beam radiotherapy with the hypoxia-activated prodrug ICF05016 in an experimental model of extraskeletal myxoid chondrosarcoma. Invest New Drugs 39, 295–303 (2021). https://doi.org/10.1007/s10637-020-01002-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-020-01002-4