Summary
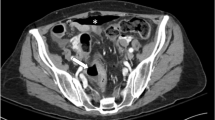
Background Bevacizumab (Bev) is generally well-tolerated, and Bev-associated intestinal perforation (BAP) is a rare albeit serious side effect in cases of non-small cell lung cancer (NSCLC). Therefore, the present study aimed to identify clinical predictors of BAP to help predict and manage the development of life-threatening intestinal complications among patients receiving Bev. Methods This retrospective study evaluated demographic, clinical, and treatment factors for patients with NSCLC who were treated with Bev between February 2010 and August 2015 at our center. Results We identified 314 regimens (208 patients; median age: 65 years; 115 women) for analysis, which included 119 first-line regimens, 74 s-line regimens, and 121 third-line or later regimens. BAP occurred in 7 cases (2.23% among all regimens and 3.37% among all patients), which generally occurred during first- or second-line treatment and was caused by ulcerative colitis (1 case), colon diverticulitis (1 case), and idiopathic perforations (5 cases). Univariate analyses revealed that BAP was significantly associated with deteriorating PS during the first cycle of chemotherapy (odd ratio [OR]: 11.07, 95% confidence interval [CI]: 2.37–51.63, p = 0.0022), grade ≥ 3 diarrhea (OR: 11.37, 95% CI: 2.37–54.50, p = 0.0024), febrile neutropenia (OR: 9.16, 95% CI: 1.98–42.49, p = 0.0047), and stomatitis (OR: 4.60, 95% CI: 1.01–21.04, p = 0.0492). Conclusions Among patients with NSCLC, BAP was associated with deteriorating PS during the first cycle of chemotherapy, grade ≥ 3 diarrhea, febrile neutropenia, and stomatitis. Therefore, careful observation is needed for patients with NSCLC who receive Bev in any line of treatment, especially if they develop serious side effects that affect their PS or mucous membrane.
Similar content being viewed by others
References
Hoffman PC, Mauer AM, Vokes EE (2002) Lung cancer. Lancet 355:479–485
Sandler A, Gray R, Perry MC, Brahmer J, Schiller JH, Dowlati A, Lilenbaum R, Johnson DH (2006) Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer. N Engl J Med 355:2542–2550
Reck M, von Pawel J, Zatloukal P, Ramlau R, Gorbounova V, Hirsh V, Leighl N, Mezger J, Archer V, Moore N, Manegold C (2009) Phase III trial of cisplatin plus gemcitabine with either placebo or bevacizumab as first-line therapy for nonsquamous non-small-cell lung cancer: AVAil. J Clin Oncol 27:1227–1234
Takeda M, Okamoto I, Yamanaka T, Nakagawa K, Nakanishi Y (2012) Impact of treatment with bevacizumab beyond disease progression: a randomized phase II study of docetaxel with or without bevacizumab after platinum-based chemotherapy plus bevacizumab in patients with advanced nonsquamous non-small cell lung cancer (WJOG 5910L). BMC Cancer 12:327
Hurwitz H, Fehrenbacher L, Novotny W, Cartwright T, Hainsworth J, Heim W, Berlin J, Baron A, Griffing S, Holmgren E, Ferrara N, Fyfe G, Rogers B, Ross R, Kabbinavar F (2004) Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med 350:2335–2342
Tewari KS, Sill MW, Long HJ 3rd, Penson RT, Huang H, Ramondetta LM, Landrum LM, Oaknin A, Reid TJ, Leitao MM, Michael HE, Monk BJ (2014) Improved survival with bevacizumab in advanced cervical cancer. N Engl J Med 370:734–743
Bennouna J, Sastre J, Arnold D, Österlund P, Greil R, Van Cutsem E, von Moos R, Viéitez JM, Bouché O, Borg C, Steffens CC, Alonso-Orduña V, Schlichting C, Reyes-Rivera I, Bendahmane B, André T, Kubicka S, ML18147 Study Investigators (2013) Continuation of bevacizumab after first progression in metastatic colorectal cancer (ML18147): a randomised phase 3 trial. Lancet Oncol 14:29–37
Burger RA (2007) Experience with bevacizumab in the management of epithelial ovarian cancer. J Clin Oncol 25:2902–2908
Sliesoraitis S, Tawfik B (2011) Bevacizumab-induced bowel perforation. J Am Osteopath Assoc 111:437–441
Badgwell BD, Camp ER, Feig B, Wolff RA, Eng C, Ellis LM, Cormier JN (2008) Management of bevacizumab-associated bowel perforation: a case series and review of the literature. Ann Oncol 19:577–582
Hapani S, Chu D, Wu S (2009) Risk of gastrointestinal perforation in patients with cancer treated with bevacizumab: a meta-analysis. Lancet Oncol 10:559–568
Abu-Hejleh T, Mezhir JJ, Goodheart MJ, Halfdanarson TR (2012) Incidence and management of gastrointestinal perforation from bevacizumab in advanced cancers. Curr Oncol Rep 14:277–284
Burger RA, Brady MF, Bookman MA, Monk BJ, Walker JL, Homesley HD, Fowler J, Greer BE, Boente M, Fleming GF, Lim PC, Rubin SC, Katsumata N, Liang SX (2014) Risk factors for GI adverse events in a phase III randomized trial of bevacizumab in first-line therapy of advanced ovarian cancer: a gynecologic oncology group study. J Clin Oncol 32:1210–1217
Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, Verweij J, Van Glabbeke M, van Oosterom AT, Christian MC, Gwyther SG (2000) New guidelines to evaluate the response to treatment in solid tumors. J Natl Cancer Inst 92:205–216
Tamiya M, Tamiya A, Yamadori T, Nakao K, Asami K, Yasue T, Otsuka T, Shiroyama T, Morishita N, Suzuki H, Okamoto N, Okishio K, Kawaguchi T, Atagi S, Kawase I, Hirashima T (2013) Phase2 study of bevacizumab with carboplatin-paclitaxel for non-small cell lung cancer with malignant pleural effusion. Med Oncol 30:676
Kitamura K, Kubota K, Ando M, Takahashi S, Nishijima N, Sugano T, Toyokawa M, Miwa K, Kosaihira S, Noro R, Minegishi Y, Seike M, Yoshimura A, Gemma A (2013) Bevacizumab plus chemotherapy for advanced non-squamous non-small-cell lung cancer with malignant pleural effusion. Cancer Chemother Pharmacol 71:457–461
Schellhaas E, Loddenkemper C, Schmittel A, Buhr HJ, Pohlen U (2009) Bowel perforation in non-small cell lung cancer after bevacizumab therapy. Investig New Drugs 27:184–187
Badgwell B, Feig BW, Ross MI, Mansfield PF, Wen S, Chang GJ (2007) Pneumoperitoneum in the cancer patient. Ann Surg Oncol 14:3141–3147
Choi YI, Lee SH, Ahn BK, Baek SU, Park SJ, Kim YS, Shin SH (2008) Intestinal perforation in colorectal cancers treated with bevacizumab (Avastin). Cancer Res Treat 40:33–35
Heinzerling JH, Huerta S (2006) Bowel perforation from bevacizumab for the treatment of metastatic colon cancer: incidence, etiology, and management. Curr Surg 63:334–337
Traina TA, Rugo HS, Dickler M (2007) Bevacizumab for advanced breast cancer. Hematol Oncol Clin North Am 21:303–319
Tol J, Cats A, Mol L, Koopman M, Bos MM, van der Hoeven JJ, Antonini NF, van Krieken JH, Punt CJ (2008) Gastrointestinal ulceration as a possible side effect of bevacizumab which may herald perforation. Investig New Drugs 26:393–397
Richardson DL, Backes FJ, Hurt JD, Seamon LG, Copeland LJ, Fowler JM, Cohn DE, O'Malley DM (2010) Which factors predict bowel complications in patients with recurrent epithelial ovarian cancer being treated with bevacizumab? Gynecol Oncol 118:47–51
Kaye SB (2007) Bevacizumab for the treatment of epithelial ovarian cancer: will this be its finest hour? J Clin Oncol 25:5150–5152
Kindler HL, Friberg G, Singh DA, Locker G, Nattam S, Kozloff M, Taber DA, Karrison T, Dachman A, Stadler WM, Vokes EE (2005) Phase II trial of bevacizumab plus gemcitabine in patients with advanced pancreatic cancer. J Clin Oncol 23:8033–8040
Kamba T, Tam BY, Hashizume H, Haskell A, Sennino B, Mancuso MR, Norberg SM, O'Brien SM, Davis RB, Gowen LC, Anderson KD, Thurston G, Joho S, Springer ML, Kuo CJ, McDonald DM (2006) VEGF-dependent plasticity of fenestrated capillaries in the normal adult microvasculature. Am J Physiol Heart Circ Physiol 290:H560–H576
Lordick F, Geinitz H, Theisen J, Sendler A, Sarbia M (2006) Increased risk of ischemic bowel complications during treatment with bevacizumab after pelvic irradiation: report of three cases. Int J Radiat Oncol Biol Phys 64:1295–1298
Collins D, Ridgway PF, Winter DC, Fennelly D, Evoy D (2009) Gastrointestinal perforation in metastatic carcinoma: a complication of bevacizumab therapy. Eur J Surg Oncol 35:444–446
Acknowledgments
The authors thank all the participants of this study.
Funding
The authors did not receive any funding for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Tamiya, M., Suzuki, H., Shiroyama, T. et al. Clinical predictors of bevacizumab-associated intestinal perforation in non-small cell lung cancer. Invest New Drugs 36, 696–701 (2018). https://doi.org/10.1007/s10637-018-0581-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-018-0581-1