Abstract
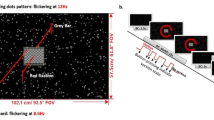
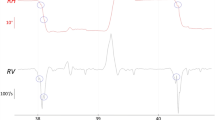
Purpose Intra-ocular pressure (IOP) increases to double that of its normal level under full-body inversion, in part simulating high IOPs found in glaucoma and ocular hypertension. Inversion also simulates negative g-forces experienced in aerobatic maneuvers such as those produced during aerial combat. Studies using achromatic pattern-reversal visual evoked potentials (VEPs) have shown losses in response amplitude when subjects are inverted and IOP is increased. In other studies, chromatic, pattern-onset VEPs have been shown to be a sensitive and objective indicator of ocular and systemic pathology. Thus, chromatic pattern-onset VEPs may also show changes in amplitude and/or latency when subjects are subjected to full-body inversion. Methods In this study we employed chromatic, pattern-onset VEPs to determine if there were changes in response latency for the different visual pathways with healthy subjects during full-body inversion. Stimuli were 1 cpd horizontal sine-wave patterns presented in an onset mode (100 ms on/400 ms off). Subjects (n = 7) were tested at low to medium contrast levels, which were subjectively equated across chromatic pathways (S, LM, and achromatic). Patterns were presented using LCD goggles. Results All subjects showed a small but statistically significant increase in latency in the large negative component (CII) for both L − M and S − (L + M) pathways during inversion. The achromatic pathway also showed a statistically significant increase in latency to the positive component (CI) during inversion. Conclusions These results demonstrate that changes in ocular and/or systemic physiology during full-body inversion can result in increased latencies of chromatic and achromatic pattern-onset VEPs.



Similar content being viewed by others
Abbreviations
- IOP:
-
Intra-ocular pressure
- RVPP:
-
Retinal vascular profusion pressure
- VEP:
-
Visual evoked potential
References
Grieshaber MC, Flammer J (2005) Blood flow in glaucoma. Curr Opin Ophthalmol 16:79–83. doi:10.1097/01.icu.0000156134.38495.0b
Linder BJ, Trick GL (1987) Simulation of spaceflight with whole-body head down tilt: influence on intraocular pressure and retinocortical processing. Aviat Space Environ Med 58(9 Pt 2):A139–A142
Draeger J, Hanke K (1986) Postural variations of intraocular pressure—preflight experiments for the D1 mission. Ophthalmic Res 18(1):55–60
Hawkins AS, Szlyk JP, Ardickas Z, Alexander KR, Wilensky JT (2003) Comparison of contrast sensitivity, visual field testing in patients with glaucoma. J Glaucoma 12(2):134–138. doi:10.1097/00061198-200304000-00008
Linder J, Trick G, Wolf L (1988) Altering body position affects intraocular pressure and visual function. Invest Ophthalmol Vis Sci 29(10):1492–1497
Flammer J (1994) The vascular concept in glaucoma. Surv Ophthalmol 38(Suppl):3–6. doi:10.1016/0039-6257(94)90041-8
Flammer J, Drance SM (1984) Correlation between color vision scores and quantitative perimetry in suspected glaucoma. Arch Ophthalmol 102(1):38–39
Flammer J, Orgül S, Costa VP, Orzalesi N, Krieglstein GK, Serra LM, Renard JP, Stefánsson E (2002) The impact of ocular blood flow in glaucoma. Prog Retin Eye Res 21:359–393. doi:10.1016/S1350-9462(02)00008-3
Flammer J (2001) Glaucoma. A guide for patients. An introduction for care-providers. A reference for quick information, 1st edn. Huber, Bern, Gottingen/Toronto/Seattle
Weinreb R, Cook J, Friberg T (1984) Effect of inverted body position on interlobular pressure. Am J Ophthalmol 98:784–787
Friberg T, Weinreb R (1985) Ocular manifestations of gravity inversion. JAMA 253(12):1755–1757. doi:10.1001/jama.253.12.1755
Kothe AC, Lovasik JV (1989) A parametric evaluation of retinal vascular perfusion pressure and visual neural function in man. Electroencephalogr Clin Neurophysiol 75:185–199. doi:10.1016/0013-4694(90)90172-G
Friberg TR, Sanborn G (1985) Optic nerve dysfunction during gravity inversion. Pattern reversal visual evoked potentials. Arch Ophthalmol 103(11):1687–1689
Greensteitn VC, Hood DC, Ritch R, Steinberger D, Carr RE (1989) S (blue) cone pathway vulnerability in retinitis pigmentosa diabetes and glaucoma. Invest Ophthalmol Vis Sci 30:1732–1737
Crognale MA, Rabin J, Switkes E, Adams AJ (1993) Selective loss of S-pathway sensitivity in central serous chroidpathy revealed by spatio-chromatic visual evoked cortical potentials (VECP) In: Drum B (ed) Colour vision deficiencies XI, pp 229–239
Schneck ME, Lee E, Fortune B, Switkes EW, Crognale M (1997) Spatio-chromatic VEPs in recovered optic neuritis and multiple sclerosis. In: Cavonius CR (ed) Color vision deficiencies XIII, pp 179–185
Rabin J, Switkes E, Crognale MA, Schneck M (1994) Visual evoked potentials in three-dimensional color space: correlates of spatio-chromatic processing. Vis Res 34:2657–2671. doi:10.1016/0042-6989(94)90222-4
Cotter SA, Lee DY, French AL (1999) Evaluation of a new color vision test: “color vision testing made easy”. Opt Vis Sci 76(9):631–636. doi:10.1097/00006324-199909000-00020
MacLeod DI, Boynton RM (1978) Chromaticity diagram showing cone excitation by stimuli of equal luminance. J Opt Soc Am 69:1183–1186. doi:10.1364/JOSA.69.001183
Derrington AM, Krauskopf J, Lennie P (1984) Chromatic mechanisms in lateral geniculate nucleus of macaque. J Physiol 357:241–265
Wagner B, Boynton RM (1972) Comparison of four methods of heterochromatic photometry. J Opt Soc Am 62:1508–1515. doi:10.1364/JOSA.62.001508
Switkes E, Crognale MA (1999) Comparison of color and luminance contrasts: apples versus oranges? Vis Res 39:1823–1831. doi:10.1016/S0042-6989(98)00219-3
Berninger TA, Arden GB, Hogg CR, Frumkes T (1989) Separable evoked retinal and cortical potentials from each major visual pathway: preliminary results. Br J Ophthalmol 73(7):502–511. doi:10.1136/bjo.73.7.502
Kulikowski JJ, Robson AG, McKeefry DJ (1996) Specificity and selectivity of chromatic visual evoked potentials. Vis Res 36(21):3397–3401. doi:10.1016/0042-6989(96)00055-7
Switkes E, Crognale M, Rabin J, Schneck ME, Adams AJ (1996) Reply to ‘Specificity and selectivity of chromatic visual evoked potentials’. Vis Res 36:3403–3405. doi:10.1016/0042-6989(96)00056-9
Porciatti V, Sartucci F (1996) Retinal and cortical evoked response to chromatic contrast stimuli. Specific losses in both eyes of patients with multiple sclerosis and unilateral optic neuritis. Brain 199:723–740. doi:10.1093/brain/119.3.723
Harding GFA, Odom JV, Spileers W, Spekreijse H (1996) Standard for visual evoked potentials 1995. Vis Res 36:3567–3572. doi:10.1016/0042-6989(96)00125-3
Tekavcic-Pompe M, Tekavcic I (2008) Color vision in the tritan axis is predominantly affected at high altitude. High Alt Med Biol 9(1):38–42. doi:10.1089/ham.2008.1034
Gobba F (2000) Color vision: a sensitive indicator for exposure to neurotoxins. Neurotoxicology 21(5):857–862
Pokorny J, Pinckers S (eds) (1979) Congenital and acquired color vision defects. Grune & Straton, New York
Mollon JD (1982) Color vision. Ann Rev Physiol 33:41–85
Sanberg MA, Berson EL (1977) Blue and green cone mechanisms in retinitis pigmentosa. Invest Ophthalmol Vis Sci 16:149–157
Ventura Df, Gualtieri M, Oliveria AG, Costa MF, Quiros P, Sadun F, de Negri AM, Salomao SR, Berezovsky A, Sherman J, Sadun AA, Carelli V (2007) Male prevalence of acquired color vision defects in asymptomatic carriers of Lever’s hereditary optic neuropathy. Invest Ophthalmol Vis Sci 48(4):2362–2370
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Highsmith, J., Crognale, M.A. Changes in chromatic pattern-onset VEP with full-body inversion. Doc Ophthalmol 119, 59–66 (2009). https://doi.org/10.1007/s10633-009-9170-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10633-009-9170-0