Abstract
Background
Currently, the mechanisms of impaired gut mucosal immunity in sepsis remain unclear. Gut immunoglobulin A (IgA) is an important defense mechanism against invasive pathogens, and CD4+ T cells regulate the IgA response.
Aim
We aimed to verify the hypothesis indicating that CD4+ T pyroptosis induced by lipopolysaccharide (LPS) leads to an impaired gut IgA response and subsequent bacterial translocation and organ damage.
Methods
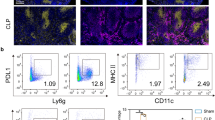
Cultured CD4+ T cells and mice were manipulated with LPS, and pyroptosis was improved by A438079 or adoptive CD4+ T cell transfer. The changes demonstrated in pyroptosis-related molecules, cytotoxicity and CD4+ T cells were examined to determine CD4+ T pyroptosis. The changes demonstrated in IgA+ B cells, AID (key enzyme for immunoglobulins) and IgA production and function were examined to evaluate the IgA response. Serum biomarkers, bacterial colonies and survival analysis were detected for bacterial translocation and organ damage.
Results
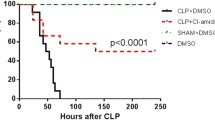
LPS attack induced CD4+ T pyroptosis, as evidenced by increased expression of P2X7, Caspase-11 and cleaved GSDMD, which elevated cytotoxicity and decreased CD4+ T cells. Decreased CD4+ T subsets (Foxp3+ T and Tfh cells) influenced the IgA response, as evidenced by lower AID expression, which decreased IgA+ B cells and IgA production and function. A438079 or cell transfer improved the IgA response but failed to reduce the translocation of gut pathogens, damage to the liver and kidney, and mortality of mice.
Conclusion
LPS attack results in CD4+ T pyroptosis. Improvement of pyroptosis restores the mucosal IgA response but fails to ameliorate bacterial translocation and organ damage.






Similar content being viewed by others
References
Mira JC, Gentile LF, Mathias BJ et al. Sepsis pathophysiology, chronic critical illness, and persistent inflammation-immunosuppression and catabolism syndrome. Crit Care Med. 2017;45:253–262.
Amornphimoltham P, Yuen PST, Star RA, Leelahavanichkul A. Gut leakage of fungal-derived inflammatory mediators: part of a gut-liver-kidney axis in bacterial sepsis. Dig Dis Sci. 2019;64:2416–2428.
Longhitano Y, Zanza C, Thangathurai D et al. Gut alterations in septic patients: a biochemical literature review. Rev Recent Clin Trials 2020;15:289–297.
Wang C, Li Q, Ren J. Microbiota-immune interaction in the pathogenesis of gut-derived infection. Front Immunol. 2019;10:1873.
Pabst O. New concepts in the generation and functions of IgA. Nat Rev Immunol. 2012;12:821–832.
Cabrera-Perez J, Badovinac VP, Griffith TS. Enteric immunity, the gut microbiome, and sepsis: rethinking the germ theory of disease. Exp Biol Med (Maywood). 2017;242:127–139.
Ates I, Dogan N, Aksoy M, Halıcı Z, Gündogdu C, Keles MS. The protective effects of IgM-enriched immunoglobulin and erythropoietin on the lung and small intestine tissues of rats with induced sepsis: biochemical and histopathological evaluation. Pharm Biol. 2015;53:78–84.
Jorgensen I, Miao EA. Pyroptotic cell death defends against intracellular pathogens. Immunol Rev. 2015;265:130–142.
Santana PT, Benjamim CF, Martinez CG, Kurtenbach E, Takiya CM, Coutinho-Silva R. The P2X7 receptor contributes to the development of the exacerbated inflammatory response associated with sepsis. J Innate Immun. 2015;7:417–427.
Yang D, He Y, Muñoz-Planillo R, Liu Q, Núñez G. Caspase-11 requires the Pannexin-1 channel and the Purinergic P2X7 pore to mediate pyroptosis and endotoxic shock. Immunity 2015;43:923–932.
Guo R, Zhao G, Bai G et al. Depletion of mTOR ameliorates CD4+ T cell pyroptosis by promoting autophagy activity in septic mice. Int Immunopharmacol. 2023;124:110964.
Fagarasan S, Kawamoto S, Kanagawa O, Suzuki K. Adaptive immune regulation in the gut: T cell-dependent and T cell-independent IgA synthesis. Annu Rev Immunol. 2010;28:243–273.
Zhang X-Y, Chen Xi, Zhang H-F et al. Propofol does not reduce pyroptosis of enterocytes and intestinal epithelial injury after lipopolysaccharide challenge. Dig Dis Sci. 2018;63:81–91.
Fuc E, Złotkowska D, Wasilewska E, Wróblewska B. OVA-experienced CD4+ T cell transfer and chicken protein challenge affect the immune response to OVA in a murine model. Int J Mol Sci. 2021;22:6573.
Chang Z, Zhang Y, Lin M et al. Improvement of gut-vascular barrier by terlipressin reduces bacterial translocation and remote organ injuries in gut-derived sepsis. Front Pharmacol. 2022;13:1019109.
Zhang X-Y, Liu Z-M, Zhang H-F et al. TGF-β1 improves mucosal IgA dysfunction and dysbiosis following intestinal ischaemia-reperfusion in mice. J Cell Mol Med. 2016;20:1014–1023.
Zhang XY, Liu ZM, Wen SH et al. Dexmedetomidine administration before, but not after, ischemia attenuates intestinal injury induced by intestinal ischemia-reperfusion in rats. Anesthesiology 2012;116:1035–1046.
Mouries J, Brescia P, Silvestri A et al. Microbiota-driven gut vascular barrier disruption is a prerequisite for non-alcoholic steatohepatitis development. J Hepatol. 2019;71:1216–1228.
Vincent J-L, Opal SM, Marshall JC, Tracey KJ. Sepsis definitions: time for change. Lancet 2013;381:774–775.
Boomer JS, To K, Chang KC et al. Immunosuppression in patients who die of sepsis and multiple organ failure. JAMA 2011;306:2594–2605.
Osterberg J, Johnsson C, Gannedahl G, Westlund A, Haglund U. Alterations in mucosal immune cell distribution in septic rats. Shock 1997;7:182–185.
Proietti M, Cornacchione V, Jost TR et al. ATP-gated ionotropic P2X7 receptor controls follicular T helper cell numbers in Peyer’s patches to promote host-microbiota mutualism. Immunity 2014;41:789–801.
Liu Y, Zhang Y, Liu Q et al. Inhibition of DDX3X ameliorated CD4+ T cells pyroptosis and improves survival in septic mice. Mol Immunol. 2023;154:54–60.
Lai Y-N, Yeh S-L, Lin M-T, Shang H-F, Yeh C-L, Chen W-J. Glutamine supplementation enhances mucosal immunity in rats with Gut-Derived sepsis. Nutrition 2004;20:286–291.
Fan J, Li G, Lidong Wu et al. Parenteral glutamine supplementation in combination with enteral nutrition improves intestinal immunity in septic rats. Nutrition 2015;31:766–774.
Zeng Q, He X, Puthiyakunnon S et al. Probiotic mixture golden Bifido prevents neonatal Escherichia coli K1 translocation via enhancing intestinal defense. Front Microbiol. 2017;8:1798.
Dong X, Hao Tu, Bai X, Qin S, Li Z. Intrinsic/extrinsic apoptosis and pyroptosis contribute to the selective depletion of B cell subsets in septic shock patients. Shock 2023;60:345–353.
Arulkumaran N, Sixma ML, Pollen S et al. P2X7 receptor antagonism ameliorates renal dysfunction in a rat model of sepsis. Physiol Rep. 2018;6:13622.
Zou Z, Wang Q, Zhou M et al. Protective effects of P2X7R antagonist in sepsis-induced acute lung injury in mice via regulation of circ_0001679 and circ_0001212 and downstream Pln, Cdh2, and Nprl3 expression. J Gene Med. 2020;22:e3261.
Huang J, Wang Z, Zhang X et al. Lipidomics study of sepsis-induced liver and lung injury under anti-HMGB1 intervention. J Proteome Res. 2023;22:1881–1895.
Xu-Yu Zhang Su, Guan H-F, Li R-Y, Liu Z-M. Activation of PD-1 protects intestinal immune defense through IL-10/miR-155 pathway after intestinal ischemia reperfusion. Dig Dis Sci. 2018;63:3307–3316.
Acknowledgments
The authors thank Dr. Yu-zhuang Li and Ying-shi chen (Zhongshan School of Medicine, Sun Yat-sen University) for their professional advice in Flow cytometry. Reprints will not be available from the authors.
Funding
This work was supported by grants from Natural Science Foundation of Guangdong Province, China (No. 2021A1515010990 to Xu-yu Zhang). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, cultured cell and animal experiments, data collection and analysis were performed by Z-ML, YZ, HZ, XC, HP and XZ. The first draft of the manuscript was written by Z-ML and XZ, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, ZM., Zhang, Yn., Zhang, Hf. et al. Restoration of the Mucosal IgA Response by Improving CD4+ T Pyroptosis Fails to Attenuate Gut Bacterial Translocation and Organ Damage After LPS Attack. Dig Dis Sci 69, 798–810 (2024). https://doi.org/10.1007/s10620-024-08278-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-024-08278-z