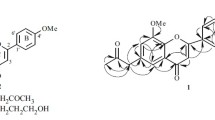
Two new isoflavones, 6-hydroxy-7-(3-hydroxypropyl)-4′-methoxyisoflavone (1) and 6,4′-dihydroxy-7-(3-hydroxypropyl)-isoflavone (2), were isolated from the flowers of Rosa damascena. Their structures were elucidated by spectroscopic methods, including extensive 1D and 2D NMR techniques. Compounds 1 and 2 were evaluated for their anti-methicillin-resistant Staphylococcus aureus (anti-MRSA) activity. The results revealed that compounds 1 and 2 showed weak inhibition with IZD of 11.6 ± 1.2 and 11.2 ± 1.0 mm, respectively. Compounds 1 and 2 were also tested for their antioxidant activity and showed good activity with an IC50 value of 4.2 and 4.0 μg/mL, respectively.

Similar content being viewed by others
References
The Dictionary of Roses in Colour, Ed. by S. M. Gault and P. M. Synge, Ebury Press, London, 1971.
Flora of China, Vol. 37, Ed. by L. Putian and Y. Jiang, Chinese Science Press, Beijing, 1985, p. 401.
X. H. Zhang, Z. Z. Xie, Chin. J. Shandong. Forest. Sci. Tech., 42, 5 (2012).
J. L. Lu, Chin. Flow. Hortic., 11, 26 (2012).
K. Shibata, A Cyclopedia of Useful Plant and Plant Products, Enlarged and Revised Edition, The Hokuryukan, Tokyo, 1957, p. 612.
V. Cunja, M. Mikulic-Petkovsek, F. Stampar, and V. Schmitzer, J. Am. Soc. Hortic. Sci., 139, 157 (2014).
S. Ochir, B. Park, M. Nishizawa, T. Kanazawa, M. Funaki, and T. Yamagishi, J. Nat. Med., 64, 383 (2010).
K. H. Park, K. Sung, S. E. Choi, J. H. Kwon, H. Myung, and M. W. Lee, Chem. Pharm. Bull., 58, 1227 (2010).
E. A. Porter, A. A. van den Bos, G. C. Kite, N. C. Veitch, and M. S. Simmonds, Phytochemistry, 81, 90 (2012).
E. K. Kwon, D. Y. Lee, H. Lee, D. O. Kim, N. Baek, Y. E. Kim, and H. Y. Kim, J. Agric. Food. Chem., 58, 882 (2010).
X. M. Gao, L. Y. Yang, L. D. Shu, Y. Q. Shen, Y. J. Zhang, and Q. F. Hu, Heterocycles, 85, 1925 (2012).
Y. K. Li, J. Q. Sun, X. M. Gao, and C. Lei, Helv. Chim. Acta, 97, 414 (2014).
Ali Mostafavi and Daryoush Afzali, Chem. Nat. Compd., 45, 110 (2008).
N. Zeng, Y. Shen, L. Z. Li, W. H. Jiao, P. Y. Gao, S. J. Song, W. S. Chen, and H. W. Lin, J. Nat. Prod., 74, 732 (2011).
E. S. Abdel-Hameed, S. A. Bazaid, and H. A. Hagag, J. Essent. Oil. Res., 28, 121 (2016).
M. Zhou, K. Zhou, P. He, K. M. Wang, R. Z. Zhu, Y. D. Wang, W. Dong, G. P. Li, H. Y. Yang, Y. Q. Ye, G. Du, X. M. Li, and Q. F. Hu, Planta Med., 82, 414 (2016).
L. Li, Q. P. Shen, C. B. Liu, Y. Wang, J. J. Yao, T. Zhang, F. M. Zhang, P. He, X. X. Si, Z. H. Liu, M. M. Miao, and G. Y. Yang, Phytochem. Lett., 13, 156 (2015).
S. Mulla, A. Kumar, and S. Rajdev, Clinical and Laboratory Standards Institute (CLSI), Performance Standards for Antimicrobial Susceptibility Testing: Eighteenth Informational Supplement M100-S18, 3rd edn., Wayne, Pennsylvania, 2008.
S. Takamatsu, A. M. Galal, S. A. Ross, D. Ferreira, M. A. Elsohly, A. R. Ibrahim, and F. S. El-Feraly, Phytother. Res., 17, 963 (2003).
Acknowledgment
This research was supported by the Research Foundation of China Tobacco Company (No. 110201502006), the Research Foundation of China Tobacco Yunnan Industrial Co., Ltd. (Nos. 2015539200340277, 2016JC03, and JSZX20151008-52), the National Natural Science Foundation of China (Nos. 81660717 and 21462051), and the Natural Science Foundation of Yunnan Province (No. 2014FD033).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 3, May–June, 2019, pp. 386–389.
Rights and permissions
About this article
Cite this article
Xiang, HY., Xing, HH., Li, J. et al. Two New Isoflavones from the Flowers of Rosa damascena and Their Biological Activities. Chem Nat Compd 55, 449–452 (2019). https://doi.org/10.1007/s10600-019-02711-w
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-019-02711-w