Abstract
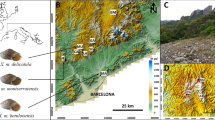
Population genetics of threatened species provides information about evolutionary pressures over those populations and thus may inform conservation management strategies. However, conservation genetics still has a low impact on conservation practices. This study’s aim is to integrate genetics in the conservation management of the only-known population of an extremely narrow-range endemic Corsican snail – Tyrrhenaria ceratina –, whose distribution area is restricted to the Ricantu site in Corsica. Using non-invasive DNA samples of 210 individuals, we amplified 13 microsatellites loci to assess the population viability, genetic structure and demographic history of the population, along with the estimation of the historical and contemporary gene flow between identified genetic clusters. We also estimated the dispersal ability of the species. Our results showed a surprisingly high genetic diversity, along with a pattern of isolation by distance (IBD) and a strongly spatialized genetic structure. Furthermore, we underlined a low functional connectivity, along with evidence of a recent decline in the population size, which are both likely due to a historical fragmentation between the sampled areas, caused by anthropization. Overall, this study allows to provide a first insight about the functioning of the population, to guide future conservation actions for the species.



Similar content being viewed by others
References
Allendorf FW, Hohenlohe PA, Luikart G (2010) Genomics and the future of conservation genetics. Nat Rev Genet 11(10):697–709. https://doi.org/10.1038/nrg2844
Araki H, Waples R, Blouin M (2007) A potential bias in the temporal method for estimating Ne in admixed populations under natural selection. Publications, Agencies and Staff of the U.S. Department of Commerce. https://digitalcommons.unl.edu/usdeptcommercepub/464
Arnaud J-F, Madec L, Guiller A, Bellido A (2001) Spatial analysis of allozyme and microsatellite DNA polymorphisms in the land snail Helix aspersa (Gastropoda: Helicidae). Mol Ecol 10(6):1563–1576. https://doi.org/10.1046/j.1365-294X.2001.01292.x
Arnaud JF, Madec L, Guiller A, Deunff J (2003) Population genetic structure in a human-disturbed environment: a case study in the land snail Helix aspersa (Gastropoda: Pulmonata). Heredity 90(6):451–458. https://doi.org/10.1038/sj.hdy.6800256
Beaumont MA, Zhang W, Balding DJ (2002) Approximate bayesian computation in population genetics. Genetics 162(4):2025–2035. https://doi.org/10.1093/genetics/162.4.2025
Beerli P (2006) Comparison of bayesian and maximum-likelihood inference of population genetic parameters. Bioinf (Oxford England) 22:341–345. https://doi.org/10.1093/bioinformatics/bti803
Bender DJ, Contreras TA, Fahrig L (1998) Habitat loss and Population decline: a Meta-analysis of the Patch size Effect. Ecology 79(2):517–533. https://doi.org/10.1890/0012-9658(1998)079[0517:HLAPDA]2.0.CO;2
Bouchet P, Ripken T, Recorbet B (1997) Rediscovery of the corsican land snail Helix ceratina on the verge of extinction. La Terre et la Vie 52:97–111
Caughley G (1994) Directions in Conservation Biology. J Anim Ecol 63(2):215–244. https://doi.org/10.2307/5542
Caziot E (1903) Etude sur la faune des Mollusques vivants terrestres et fluviatiles de l’Ile de Corse. Bulletin de la Société des Sciences Historiques et Naturelles de la Corse, 22 (Fasc. 266/269): 354 pp., 2 pl. http://gallica.bnf.fr/ark:/12148/bpt6k5725738n/f9
Caziot E (1911) Etude révisionnelle des mollusques quaternaires des brèches de Toga, Bastia (Corse). Bull de la Société géologique de France 11(1911):239–249
Charrier M, Nicolai A, Dabard MP, Crave A (2013) Plan national d’action 2013–2017 en faveur de l’Hélix de corse tyrrhenaria ceratina. https://doi.org/10.13140/RG.2.1.4816.4004
Chevalier L, Charrier M (2002) Etude de la démographie de Helix ceratina et des interactions nutritionnelles entre cet escargot et la végétation su dite du Campo dell’Oro à Ajaccio. Rapport final, étude financée par la DIREN Corse sud et l’OEC, 153 p. et annexes
Coates DJ, Byrne M, Moritz C (2018) Genetic diversity and conservation units: dealing with the species-population continuum in the age of genomics. Front Ecol Evol 6:165. https://doi.org/10.3389/fevo.2018.00165
Cornuet J-M, Santos F, Beaumont MA, Robert CP, Marin J-M, Balding DJ, Guillemaud T, Estoup A (2008) Inferring population history with DIY ABC: a user-friendly approach to approximate bayesian computation. Bioinf (Oxford England) 24(23):2713–2719. https://doi.org/10.1093/bioinformatics/btn514
Crnokrak P, Roff DA (1999) Inbreeding depression in the wild. Heredity 83(3):260–270. https://doi.org/10.1038/sj.hdy.6885530
Cucherat X (2019) Mission d’assistance pour le monitoring de l’hélix de corse (Helix ceratina)
Dahirel M, Ansart A, Madec L (2013) Stage- and weather-dependent dispersal in the brown garden snail Cornu aspersum. Population Ecology, 56, 227-237.7https://doi.org/10.1007/s10144-013-0407-0
Diamond JM (1972) Biogeographic Kinetics: Estimation of Relaxation Times for Avifaunas of Southwest Pacific Islands. Proceedings of the National Academy of Sciences Nov 1972, 69 (11) 3199–3203; https://doi.org/10.1073/pnas.69.11.3199
Do C, Waples RS, Peel D, Macbeth GM, Tillett BJ, Ovenden JR (2014) NeEstimator v2: re-implementation of software for the estimation of contemporary effective population size (ne) from genetic data. Mol Ecol Resour 14(1):209–214. https://doi.org/10.1111/1755-0998.12157
Earl DA, vonHoldt BM (2012) STRUCTURE HARVESTER: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv Genet Resour 4(2):359–361. https://doi.org/10.1007/s12686-011-9548-7
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software structure: a simulation study. Mol Ecol 14(8):2611–2620. https://doi.org/10.1111/j.1365-294X.2005.02553.x
Fagan WF, Holmes EE (2006) Quantifying the extinction vortex. Ecol Lett 9(1):51–60. https://doi.org/10.1111/j.1461-0248.2005.00845.x
Frankham R (1996) Relationship of genetic variation to Population size in Wildlife. Conserv Biol 10(6):1500–1508. https://doi.org/10.1046/j.1523-1739.1996.10061500.x
Gabriel W, Bürger R (1994) Extinction risk by mutational meltdown: Synergistic effects between population regulation and genetic drift. In V. Loeschcke, S. K. Jain, and J. Tomiuk (Eds.), Conservation Genetics (pp. 69–84). Birkhäuser. https://doi.org/10.1007/978-3-0348-8510-2_7
Gaggiotti OE (2003) Genetic threats to population persistence. Ann Zool Fenn 40(2):155–168. https://www.jstor.org/stable/23736522
Galleras F, Freytet A (2014) Réhabilitation écologique et paysagère du site du Ricantu. Doublevébé Récup., p 50
Gargominy O, Tercerie S, Régnier C, Ramage T, Dupont P, Daszkiewicz P, Poncet L (2021) TAXREF v15, référentiel taxonomique pour la France: méthodologie, mise en oeuvre et diffusion Rapport Patrinat. Paris, Muséum national d’Histoire naturelle. p. 63
Goudet J (2005) HIERFSTAT, a package for R to compute and test hierarchical F-statistics. Mol Ecol Notes 5:184–186. https://doi.org/10.1111/j.1471-8286.2004.00828.x
Halley JM, Monokrousos N, Mazaris AD, Vokou D (2017) Extinction debt in plant communities: where are we now? J Veg Sci 28(3):459–461. https://doi.org/10.1111/jvs.12538
Hall SJG, Lenstra JA, Deeming DC (2012) Prioritization based on neutral genetic diversity may fail to conserve important characteristics in cattle breeds. J Anim Breed Genet 129(3):218–225. https://doi.org/10.1111/j.1439-0388.2011.00949.x
Hylander K, Ehrlén J (2013) The mechanisms causing extinction debts. Trends in ecology and evolution 28(6):341–346. https://doi.org/10.1016/j.tree.2013.01.010
IUCN (2012) IUCN Red List categories and criteria. https://portals.iucn.org/library/node/10315
IUCN (2021) The IUCN Red List of Threatened Species. https://www.iucnredlist.org
Jakobsson M, Rosenberg NA (2007) CLUMPP: a cluster matching and permutation program for dealing with label switching and multimodality in analysis of population structure. Bioinformatics 23(14):1801–1806. https://doi.org/10.1093/bioinformatics/btm233
Jarne P, Théron A (2001) Genetic structure in natural populations of flukes and snails: A practical approach and review. Parasitology, 123 Suppl, S27-40. https://doi.org/10.1017/s0031182001007715
Johnson SG (2005) Age, phylogeography and population structure of the microendemic banded spring snail, Mexipyrgus churinceanus. Mol Ecol 14(8):2299–2311. https://doi.org/10.1111/j.1365-294x.2005.02580.x
Kohn MH, Murphy WJ, Ostrander EA, Wayne RK (2006) Genomics and conservation genetics. Trends in Ecology and Evolution 21(11):629–637. https://doi.org/10.1016/j.tree.2006.08.001
Korábek O, Kosová T, Dolejš P, Petrusek A, Neubert E, Juřičková L (2021) Geographic isolation and human-assisted dispersal in land snails: A Mediterranean story of Helix borealis and its relatives (Gastropoda: Stylommatophora: Helicidae). Zoological Journal of the Linnean Society, zlaa186. https://doi.org/10.1093/zoolinnean/zlaa186
Kramarenko S (2014). Active and passive dispersal of terrestrial mollusks: Areview//Ruthenica.-2014https://www.academia.edu/7147639/Kramarenko_S_S_Active_and_passive_dispersal_of_terrestrial_mollusks_a_review_Ruthenica_2014_V_24_1_P_1_14
Lande R (1998) Anthropogenic, ecological and genetic factors in extinction and conservation. Popul Ecol 40(3):259–269. https://doi.org/10.1007/BF02763457
Lande R, Shannon S (1996) The role of genetic variation in Adaptation and Population Persistence in a changing environment. Evolution 50(1):434–437. https://doi.org/10.1111/j.1558-5646.1996.tb04504.x
Landguth EL, Cushman SA, Schwartz MK, McKelvey KS, Murphy M, Luikart G (2010 Oct) Quantifying the lag time to detect barriers in landscape genetics. Mol Ecol 19(19):4179–4191. https://doi.org/10.1111/j.1365-294X.2010.04808.xEpub 2010 Sep 6. PMID: 20819159
Lydeard C, Cowie RH, Ponder WF, Bogan AE, Bouchet P, Clark SA, Cummings KS, Frest TJ, Gargominy O, Herbert DG, Hershler R, Perez KE, Roth B, Seddon M, Strong EE, Thompson FG (2004) The Global Decline of Nonmarine Mollusks. BioScience,54(4),321–330. https://doi.org/10.1641/0006-3568(2004)054[0321:TGDONM]2.0.CO;2
Millette KL, Gonzalez A, Cristescu ME (2020) Breaking ecological barriers: anthropogenic disturbance leads to habitat transitions, hybridization, and high genetic diversity. Sci Total Environ 740:140046. https://doi.org/10.1016/j.scitotenv.2020.140046
O’Brien SJ, Johnson WE, Driscoll CA, Dobrynin P, Marker L (2017) Conservation Genetics of the Cheetah: Lessons learned and New Opportunities. J Hered 108(6):671–677. https://doi.org/10.1093/jhered/esx047
Palstra FP, Ruzzante DE (2008) Genetic estimates of contemporary effective population size: what can they tell us about the importance of genetic stochasticity for wild population persistence? Mol Ecol 17(15):3428–3447. https://doi.org/10.1111/j.1365-294X.2008.03842.x
Paradis E (2010) Pegas: an R package for population genetics with an integrated–modular approach. Bioinformatics 26(3):419–420. https://doi.org/10.1093/bioinformatics/btp696
Paradis G, Maurin A, Piazza C (2011) Etude phytosociologique et cartographie de la végétation du site Natura 2000 « Ricanto-Campo dell’Oro » (Ajaccio, Corse). Bulletin de la Société Botanique du Centre-Ouest – Nouvelle Série, 41: 139–232
Pritchard JK, Stephens M, Donnelly P (2000) Inference of Population structure using multilocus genotype data. Genetics 155(2):945–959. https://doi.org/10.1093/genetics/155.2.945
Qiu C, Lu D-B, Deng Y, Zou H-Y, Liang Y-S, Webster JP (2019) Population genetics of Oncomelania hupensis snails, intermediate hosts of Schistosoma japonium, from emerging, re-emerging or established habitats within China. Acta Trop 197:105048. https://doi.org/10.1016/j.actatropica.2019.105048
Raup DM (1994) The role of extinction in evolution. Proceedings of the National Academy of Sciences, 91(15), 6758–6763. https://doi.org/10.1073/pnas.91.15.6758
Reed DH, Frankham R (2003) Correlation between fitness and genetic diversity. Conserv Biol 17(1):230–237. https://doi.org/10.1046/j.1523-1739.2003.01236.x
Régnier C, Fontaine B, Bouchet P (2009) Not knowing, not Recording, Not Listing: numerous unnoticed mollusk extinctions. Conserv Biol 23(5):1214–1221. https://doi.org/10.1111/j.1523-1739.2009.01245.x
Rousset F (1997) Genetic differentiation and estimation of Gene Flow from F-Statistics under isolation by Distance. Genetics 145(4):1219–1228. https://doi.org/10.1093/genetics/145.4.1219
Salvi D, Harris DJ, Bombi P, Carretero MA, Bologna MA (2010) Mitochondrial phylogeography of the Bedriaga’s rock lizard, Archaeolacerta bedriagae (Reptilia: Lacertidae) endemic to Corsica and Sardinia. Mol Phylogenet Evol 56(2):690–697. https://doi.org/10.1016/j.ympev.2010.03.017
Samarasin P, Shuter BJ, Wright SI, Rodd FH (2017) The problem of estimating recent genetic connectivity in a changing world. Conserv Biol 31(1):126–135. https://doi.org/10.1111/cobi.12765
Schmidt DJ, Espinoza T, Connell M, Hughes JM (2018) Conservation genetics of the Mary River turtle (Elusor macrurus) in natural and captive populations. Aquat Conserv 28:115–123. https://doi.org/10.1002/aqc.2851
Sharma S, Dutta T, Maldonado JE, Wood TC, Panwar HS, Seidensticker J (2013) Forest corridors maintain historical gene flow in a tiger metapopulation in the highlands of central India. Proceedings. Biological Sciences, 280(1767), 20131506. https://doi.org/10.1098/rspb.2013.1506
Slatkin M (2008) Linkage disequilibrium—understanding the evolutionary past and mapping the medical future. Nat Rev Genet 9(6):477–485. https://doi.org/10.1038/nrg2361
Teixeira JC, Huber CD (2021) The inflated significance of neutral genetic diversity in conservation genetics. Proceedings of the National Academy of Sciences, 118(10), e2015096118. https://doi.org/10.1073/pnas.2015096118
Triantis KA, Parmakelis A, Cameron RAD (2009) Understanding fragmentation: snails show the way. J Biogeogr 36(11):2021–2022. https://doi.org/10.1111/j.1365-2699.2009.02211.x
van Strien M, Holderegger R, Van Heck H (2015) Isolation-by-distance in landscapes: considerations for landscape genetics. Heredity 114:27–37. https://doi.org/10.1038/hdy.2014.62
Vähä J-P, Erkinaro J, Niemelä E, Primmer CR (2007) Life-history and habitat features influence the within-river genetic structure of Atlantic salmon. Mol Ecol 16(13):2638–2654. https://doi.org/10.1111/j.1365-294X.2007.03329.x
Väla Ü, Einarsson A, Waits L, Ellegren H (2008) To what extent do microsatellite markers reflect genome-wide genetic diversity in natural populations? Mol Ecol 17(17):3808–3817. https://doi.org/10.1111/j.1365-294X.2008.03876.x
Waples RS, Antao T, Luikart G (2014) Effects of overlapping generations on linkage disequilibrium estimates of Effective Population size. Genetics 197(2):769–780. https://doi.org/10.1534/genetics.114.164822
Waples RS, Do C (2010) Linkage disequilibrium estimates of contemporary ne using highly variable genetic markers: a largely untapped resource for applied conservation and evolution. Evol Appl 3(3):244–262. https://doi.org/10.1111/j.1752-4571.2009.00104.x
Weir B, Cockerham C (1984) Estimating F-Statistics for the Analysis of Population-Structure. Evolution 38: 1358–1370. Evolution, 38, 1358–1370. https://doi.org/10.2307/2408641
Wiehn J, Kopp K, Rezzonico S, Karttunen S, Jokela J (2002) Family-level covariation between parasite resistance and mating system in a hermaphroditic freshwater snail. Evolution 56(7):1454–1461. https://doi.org/10.1111/j.0014-3820.2002.tb01457.x
Willi Y, Torsten N, Kristensen CM, Sgrò AR, Weeks M, Ørsted, Ary A, Hoffmann (2022) Proceedings of the National Academy of Sciences 119 (1) e2105076119; https://doi.org/10.1073/pnas.2105076119
Wilson GA, Rannala B (2003) Bayesian inference of recent Migration Rates using Multilocus genotypes. Genetics 163:1177–1191. https://doi.org/10.1093/genetics/163.3.1177
Witzenberger K, Hochkirch A (2011) Ex situ conservation genetics: a review of molecular studies on the genetic consequences of captive breeding programmes for endangered animal species. Biodivers Conserv 20:1843–1861. https://doi.org/10.1007/s10531-011-0074-4
Wright S (1943) Isolation by Distance. Genetics 28(2):114–138. https://doi.org/10.1093/genetics/28.2.114
Acknowledgements
We thank all the volunteers who realized the sample collection, especially the team of the Centre Permanent d’Initiatives pour l’Environnement d’Ajaccio mobilized for the project “Aio lumacchi, Allez les escargots” funded by Office Français de la Biodiversité and the wardens of the Collectivité Territoriale de Corse. We also thank DREAL Corse for funding support.
Handling and sampling of this protected species were performed under permits: Arrêtés préfectoraux 2 A 2018-01-29-001, 2 A 2020-03-25-001 and 2 A-2021-02-25-003.
Funding
This work was supported by the Direction Régionale de l’Environnement de l’Aménagement et du Logement de Corse, and by the Conservatoire du Littoral.
Author information
Authors and Affiliations
Contributions
AG, MD and PP conceived the idead and designed methodology; CN, XC, MD, LC and AG collected the data, LC and SD did the laboratory work. LC, AC and PP analysed and interpreted the data; LC and AG led the writing of the manuscript. All authors contributed critically to the drafts and gave final approval for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Camus, L., Poli, P., Delaugerre, MJ. et al. Unexpected and spatially structured genetic diversity of the relict population of the endangered corsican land snail Tyrrhenaria ceratina. Conserv Genet 24, 661–672 (2023). https://doi.org/10.1007/s10592-023-01535-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-023-01535-0