Abstract
The Kentucky Arrow Darter (Etheostoma sagitta spilotum) is federally listed as threatened and known only from five tributary systems in the Kentucky River system, Kentucky, USA. Recent surveys revealed considerable population decline, with individuals detected in only 45% of known historical localities. Impacts of these declines on the genetic structure of E. s. spilotum may be exacerbated by limited dispersal capabilities and small initial population size resulting in high levels of isolation among extant populations. Long standing genetic isolation may also be evident in contemporary genetic signatures; populations that have undergone historic isolation may be prime candidates for intensive conservation efforts. To address contemporary and historic genetic isolation, we generated genotypic (11 microsatellite loci) and mtDNA sequence (ND2 gene) data from multiple locations spanning the taxon’s range. We recovered seven haplotypes with low divergence levels, shared among and within multiple Kentucky River tributary systems, indicating absence of long-standing isolation among localities examined. In contrast, microsatellite data suggested all nine populations are functionally isolated, with little to no admixture among populations, even among those within the same tributary system. The drastic decline of E. s. spilotum populations has likely combined with limited dispersal, resulting in extensive contemporary genetic isolation among extant populations. Conservation management plans to enhance stability and maintain survivability of E. s. spilotum must address the severe genetic isolation identified here and work towards increasing gene flow among extant populations.



Similar content being viewed by others
References
Amos JN, Harrisson KA, Radford JQ, White M, Newell G, Mac Nally R, Sunnucks P, Pavlova A (2014) Species- and sex-specific connectivity effects of habitat fragmentation in a suite of woodland birds. Ecology 95:1556–1568. https://doi.org/10.1890/13-1328.1
Austin JD, Jelks HL, Tate B, Johnson AR (2011) Jordan F (2011) Population genetic structure and conservation genetics of threatened Okaloosa darters (Etheostoma okaloosae). Conserv Genet 12:981–989. https://doi.org/10.1007/s10592-011-0201-5
Baxter J (2015) Distribution, movement, and ecology of Etheostoma spilotum (Gilbert), the Kentucky Arrow Darter, in Gilberts Big Creek and Elisha Creek, Red Bird River Basin, Clay and Leslie Counties, Kentucky. Unpublished M.S. Thesis, Eastern Kentucky University, Richmond, KY
Belkhir K, Porsa P, Chikhi L, Raufaste N, Bonhomme F (2004) GENETIX 4.05, logical sous Windows TM our la genetique des populations. Laboratorire Genome, Populations, Interactions, CNRS UMR 5171, Universite de Montpellier II, Montpellier, France
Beneteau CL, Mandrak NE (2009) Heath DD (2009) The effects of river barriers and range expansion on the population genetic structure and stability in greenside darter (Etheostoma blennioides) populations. Conserv Genet 10:477–487. https://doi.org/10.1007/s10592-008-9627-9
Blakney JR, Loxterman JL, Keeley ER (2014) Range-wide comparisons of northern Leatherside Chub populations reveal historical and contemporary patterns of genetic variation. Conserv Genet 2014(15):757–770. https://doi.org/10.1007/s10592-014-0576-1
Bohanak AJ (1999) Dispersal, gene flow, and population structure. Q Rev Biol 74:21–45
Boutin-Ganache I, Raposo M, Raymond M, Deschepper CF (2001) M13-tailed primers improve the readability and usability of microsatellite analyses performed with two different allele-sizing methods. Biotechniques 31:24–28. https://doi.org/10.2144/01311bm02
Burr BM, Warren ML Jr (1986) A distributional atlas of Kentucky fishes, vol 4. Kentucky Nature Preserves Commission, Frankfort
Cabin RJ, Mitchell RJ (2000) To Bonferroni or not to Bonferroni: when and how are the questions. Bull Ecol Soc Am 81(3):246–248
Castelloe J, Templeton AR (1994) Root probabilities for intraspecific gene trees under neutral coalescent theory. Mol Phylogenet Evol 3:102–113. https://doi.org/10.1006/mpev.1994.1013
Chiucchi JE, Gibbs HL (2010) Similarity of historical and contemporary gene flow among highly fragmented populations of an endangered rattlesnake. Mol Ecol 19:5345–5358. https://doi.org/10.1111/j.1365-294X.2010.04860.x
Cornuet JM, Luikart G (1996) Description and power analysis of two tests for detecting recent population bottlenecks from allele frequency data. Genetics 144:2001–2014
Coulon A, Cosson JF, Angibault JM, Cargnelutti F, Galan M, Morellet N, Petit E, Aulagnier S, Hewison AJM (2004) Landscape connectivity influences gene flow in a roe deer population inhabiting a fragmented landscape: an individual-based approach. Mol Ecol 13:2841–2850. https://doi.org/10.1111/j.1365-294X.2004.02253.x
Davis DJ, Wieman AC, Berendzen PB (2015) The influence of historical and contemporary landscape variables on the spatial genetic structure of the Rainbow Darter (Etheostoma caeruleum) in tributaries of the upper Mississippi River. Conserv Genet 16:167–179. https://doi.org/10.1007/s10592-014-0649-1
DeWoody JA, Avise JC (2000) Microsatellite variation in marine, freshwater and anadramous fishes compared with other animals. Fish Biol 56:461–473. https://doi.org/10.1111/j.1095-8649.2000.tb00748.x
Di Rienzo A, Peterson AC, Garza JC, Valdes AM, Slatkin M, Freimer NB (1994) Mutational processes of simple-sequence repeat loci in human populations. Proc Natl Acad Sci 91:3166–3170
Do C, Waples RS, Peel D, Macbeth GM, Tillett BJ, Ovenden JR (2014) NeEstimator V2: re-implementation fo software for the estimation of contemporary effective population size (Ne) from genetic data. Mol Ecol Resour 14:209–214. https://doi.org/10.1111/1755-0998.12157
Earl DA, vonHoldt BM (2012) STRUCTURE HARVESTER: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv Genet Resour 4:359–361. https://doi.org/10.1007/s12686-011-9548-7
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620. https://doi.org/10.1111/j.1365-294X.2005.02553.x
Ewers RM, Didham RK (2006) Confounding factors in the detection of species response to habitat fragmentation. Biol Rev 81:117–142. https://doi.org/10.1017/S1464793105006949
Fagan WF (2002) Connectivity, fragmentation, and extinction risk in dendriditic metapopulations. Ecology 83:3243–3249. https://doi.org/10.1890/0012-9658(2002)083%5b3243:CFAERI%5d2.0.CO;2
Fagan WF, Unmack P, Burgess C, Minckley WL (2002) Rarity, fragmentation, and extinction risk in desert fishes. Ecology 83:3250–3256. https://doi.org/10.1890/0012-9658(2002)083%5b3250:RFAERI%5d2.0.CO;2
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Evol Sys 34:487–515. https://doi.org/10.1146/annurev.ecolsys.34.011802.132419
Falush D, Stephens M, Pritchard JK (2003) Influence of population structure using multilocus genotype data: linked loci and correlated allele frequencies. Genetics 164:1567–1587
Falush D, Stephens M, Pritchard JK (2007) Inference of population structure using multilocus genotype data: dominant markers and null alleles. Mol Ecol Notes 7:574–578. https://doi.org/10.1111/j.1471-8286.2007.01758.x
Fluker BL, Kuhajda BR, Lang NJ, Harris PM (2010) Low genetic diversity and small long-term population sizes in the spring endemic Watercress Darter, Etheostoma nuchale. Conserv Genet 11:2267–2279. https://doi.org/10.1007/s10592-010-0111-y
Frankham R (1998) Inbreeding and extinction: island populations. Conserv Biol 12:665–675. https://doi.org/10.1111/j.1523-1739.1998.96456.x
Frankham R (2005) Genetics and extinction. Biol Conserv 126:131–140. https://doi.org/10.1016/j.biocon.2005.05.002
Frankham R, Lees K, Montgomery ME, England PR, Lowe EH, Briscoe DA (1999) Do population size bottlenecks reduce evolutionary potential? Anim Conserv 2:255–260. https://doi.org/10.1111/j.1469-1795.1999.tb00071.x
Frankham R, Ballou JD, Briscoe DA (2002) Introduction to Conservation Genetics. Cambridge University Press, Cambridge UK
Frankham R, Bradshaw CJA, Brook BW (2014) Genetics in conservation management: revised recommendations for the 50/500 rules, Red List criteria and population viability analyses. Biol Conserv 170:56–63. https://doi.org/10.1016/j.biocon.2013.12.036
Fu YX (1997) Statistical tests of neutrality of mutations against population growth, hitchhicking and background selection. Genetics 147:915–925
Gabel JM, Dakin EE, Freeman BJ, Porter BA (2008) Isolation and identification of eight microsatellite loci in the Cherokee Darter (Etheostoma scotti) and their variability in other members of genera Etheostoma, Ammocrypta, and Percina. Mol Ecol Resour 8:149–151. https://doi.org/10.1111/j.1471-8286.2007.01903.x
George AL, Kuhajda BR, Williams JD, Cantrell MA, Rakes PL, Shute JR (2009) Guidelines for propagation and translocation for freshwater fish conservation. Fisheries 34:529–545. https://doi.org/10.1577/1548-8446-34.11.529
Ginson R, Walter RP, Mandrak NE, Beneteau CL, Heath DD (2015) Hierarchical analysis of genetic structure in the habitat-specialist Eastern Sand Darter (Ammocrypta pellucida). Ecol Evol 5:695–708. https://doi.org/10.1002/ece3.1392
Gotelli NJ, Taylor CM (1999) Testing metapopulation models with stream-fish assemblages. Evol Ecol Res 1:835–845
Hartl DL, Clark AG (1997) Principles of Population Genetics, 3rd edn. Sinauer Associates Inc., Sunderland, MA
Hedgecock D (1994) Does variance in reproductive success limit effective population size of marine organisms? In: Beaumont A (ed) Genetics and Evolution of Aquatic Organisms. Chapman & Hall, London, pp 122–134
Hendrick PW (1995) Gene flow and genetic restoration: the Florida Panther as a case study. Conserv Biol 5:996–1007. https://doi.org/10.1046/j.1523-1739.1995.9050988.x-i1
Hitt NP, Floyd M, Compton M, McDonald K (2016) Threshold responses of Blackside Dace (Chrosomus cumberlandensis) and Kentucky Arrow Darter (Etheostoma spilotum) to stream conductivity. Southeast Nat 15:41–60. https://doi.org/10.1656/058.015.0104
Hollinsgworth P, Near TJ (2009) Temporal diversification and microendemism in Eastern Highland endemic barcheek darters (Percidae: Etheostomatinae). Evolution 63:228–243. https://doi.org/10.1111/j.1558-5646.2008.00531.x
Jakobsson M, Rosenberg NA (2007) CLUMPP: a cluster matching and permutation program for dealing with label switching and multimodality in analysis of population structure. Bioinformatics 23:1801–1806. https://doi.org/10.1093/bioinformatics/btm233
Jelks HL, Walsh SJ, Burkhead NM, Contreras-Balderas S et al (2008) Conservation status of imperiled North American freshwater and diadromous fishes. Fisheries 33:372–407
Jensen JL, Bohonak AJ, Kelley ST (2005) Isolation by distance, web service. BMC Genet 6:1–6. https://doi.org/10.1186/1471-2156-6-13
KDFWR (2005) Kentucky’s Comprehensive Wildlife Conservation Strategy. KDFWR, Frankfort, Kentucky. http://fw.ky.gov/kfwis/stwg/
Kocher TD, Conroy JA, McKaye KR, Stauffer JR, Lockwood SF (1995) Evolution of NADH dehydrogenase subunit 2 in East African cichlid fish. Mol Phylogenet Evol 4:420–432
Kottelat M, Freyhof J (2007) Handbook of European freshwater fishes. Kottelat, Berlin
Krabbenhoft TJ, Rhode FC, Leibman AN, Quattro JM (2008) Concordant mitochondrial and nuclear DNA partitions define evolutionary significant units in the imperiled Pinewoods Darter, Etheostoma mariae (Pisces: Percidae). Copeia 2008:909–915
Kuehne RA, Barbour RW (1983) The American Darters. The University of Kentucky Press, Lexington
Lang NJ, Echelle AA (2011) Novel phylogeographic patterns in a lowland fish, Etheostoma proeliare (Percidae). Southeast Nat 10:133–144
Librado P, Rozas J (2009) DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25:1451–11452. https://doi.org/10.1093/bioinformatics/btp187
Lui J, Wilson M, Hu G, Liu J, Wu J, Yu M (2018) How does habitat fragmentation affect the biodiversity and ecosystem functioning relationship? Landsc Ecol 33(3):341–1352. https://doi.org/10.1007/s10980-018-0620-5
Luikart G, Cornuet J (1998) Empirical evaluation of a test for identifying recently bottlenecked populations from allele frequency data. Conserv Biol 12:228–237. https://doi.org/10.1111/j.1523-1739.1998.96388.x
McCraney WT, Goldsmith G, Jacobs DK, Kinzinger AP (2010) Rampant drift in artificially fragmented populations of the endangered Tidewater Goby (Eucyclogobius newberryi). Mol Ecol 19:3315–3327. https://doi.org/10.1111/j.1365-294X.2010.04755.x
Morales P, Vila I, Poulin E (2011) Genetic structure in remanant populations of an endangered cypriniodontid fish Orestias ascotanensis, edemic to the Ascotán salt pan of the Altiplano. Conserv Genet 12:1639–1643. https://doi.org/10.1007/s10592-011-0245-6
Newman D, Pilson D (1997) Increased probability of extinction due to decreased genetic effective population size: experimental populations of Clarkia pulchella. Evolution 51:354–362. https://doi.org/10.1111/j.1558-5646.1997.tb02422.x
Oosterhout CV, Hutchinson WF, Wills DP, Shipley P (2004) MICRO-CHECKER: software for identifying and correcting genotyping errors in microsatellite data. Mol Ecol Resour 4:535–538. https://doi.org/10.1111/j.1471-8286.2004.00684.x
Page LM (1983) Handbook of Darters. Publications Inc, Neptune
Pavlova A, Beheregaray LB, Coleman R, Gilligan D, Harrisson KA, Ingram BA, Kearns J, Lamb AM, Lintermans M, Lyon J, Nguyen TTT, Sasaki M, Tonkin Z, Yen JDL, Sunnucks P (2017) Severe consequences of habitat fragmentation on genetic diversity of an endangered Australian freshwater fish: a call for assisted gene flow. Evol Appl 10:531–550. https://doi.org/10.1111/eva.12484
Peakall R, Smouse PE (2006) GENALEX 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol Ecol Resour 6:288–295. https://doi.org/10.1111/j.1471-8286.2005.01155.x
Peakall R, Smouse PE (2012) GenAlEx 6.5: genetic analysis in Excel. Population genetic software for teaching and research -an update. Bioinformatics 28:2537–2539. https://doi.org/10.1093/bioinformatics/bts460
Peery MZ, Kirby R, Reid BN, Stoelting R, Doucet-Beer E, Robinson S, Vasquez-Carrillo C, Pauli JN, Paslboll J (2012) Reliability of genetic bottleneck tests for detecting recent population declines. Mol Ecol 21:3403–3418. https://doi.org/10.1111/j.1365-294X.2012.05635.x
Porter BA, Fiumera AC, Avise JC (2002) Egg mimicry and allopaternal care: two mate-attracting tactics by which nesting striped darter (Etheostoma virgatum) males enhance reproductive success. Behav Ecol Sociobiol 51:350–359. https://doi.org/10.1007/s00265-002-0456-4
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Ramos-Onsins SE, Rozas J (2002) Statistical properties of new neutrality tests against population growth. Mol Biol Evol 23:1642. https://doi.org/10.1093/oxfordjournals.molbev.a004034
Reed DH, Frankham R (2003) Correlation between fitness and genetic diversity. Conserv Biol 17:230–237. https://doi.org/10.1046/j.1523-1739.2003.01236.x
Roberts JH, Angermeier PL, Hallerman EM (2013) Distance, dams, and drift: what structures populations of an endangered benthic stream fish? Freshw Biol 58:2050–2064. https://doi.org/10.1111/fwb.12190
Robinson JD, Simmons JW, Williams AS, Moyer GR (2013) Population structure and genetic diversity in the endangered bluemask darter (Etheostoma akatulo). Conserv Genet 14:79–92. https://doi.org/10.1007/s10592-012-0427-x
Rogers AR, Harpending H (1992) Population growth makes waves in the distribution of pairwise genetic differences. Mol Biol Evol 9:552–569. https://doi.org/10.1093/oxfordjournals.molbev.a040727
Rosenberg NA (2004) Distruct: a program for the graphical display of population structure. Mol Ecol Resour 4:137–138. https://doi.org/10.1046/j.1471-8286.2003.00566.x
Rousset F (2008) GENEPOP’007: a complete re-implementation of the GENEPOP software for Windows and Linux. MolEcol Resour 8:103–106. https://doi.org/10.1111/j.1471-8286.2007.01931.x
Savage WK, Fremier AK, Shaffer BH (2010) Landscape genetics of alpine Sierra Nevada salamanders reveal extreme population subdivision in space and time. Mol Ecol 19:3301–3314. https://doi.org/10.1111/j.1365-294X.2010.04718.x
Sommer S (2005) The importance of immune gene variability (MHC) in evolutionary ecology and conservation. Front Zool 2:16–33. https://doi.org/10.1186/1742-9994-2-16
Spielman D, Brook BW, Frankham R (2004) Most species are not driven to extinction before genetic factors impact them. Proc Natl Acad Sci 101:15261–15264. https://doi.org/10.1073/pnas.0403809101
Sterling KA, Reed DH, Noonan BP, Warren ML (2012) Genetic effects of habitat fragmentation and population isolation on Etheostoma raneyi (Percidae). Conserv Genet 13:859–872. https://doi.org/10.1007/s10592-012-0335-0
Switzer JF, Welsh SA, King TL (2008) Microsatellite DNA primers for the Candy Darter, Etheostoma osburni, and Variegate Darter, Etheostoma variatum, and cross-species amplification in other darters (Percidae). Mol Ecol Resour 8:335–338. https://doi.org/10.1111/j.1471-8286.2007.01946.x
Tajima F (1989) Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics 123:585–595
Templeton AR, Crandall KA, Sing CF (1992) A cladistic analysis of phenotypic associations with haplotypes inferred from restriction endonuclease mapping and DNA sequence data. III. Cladogram estimation. Genetics 132:619–633
Thomas MR (2008) Distribution and status of the Kentucky Arrow Darter (Etheostoma sagitta spilotum). Project Report, Kentucky Department of Fish and Wildlife Resources, Frankfort, KY, State and Tribal Wildlife Grant T-9-RSI-1
Turner TF (2001) Comparative study of larval transport and gene flow in darters. Copeia 3:766–774
Turner TF, Robison HW (2006) Genetic diversity of the Caddo Madtom, Noturus taylori, with comments on factors that promote genetic divergence in fishes endemic to the Ouachita Highlands. Southwest Nat 51:338–345. https://doi.org/10.1894/0038-4909(2006)51%5b338:GDOTCM%5d2.0.CO;2
Turner TF, Trexler JC (1998) Ecological and historical associations of gene flow in darters (Teleostei: Percidae). Evolution 52:1781–1801. https://doi.org/10.1111/j.1558-5646.1998.tb02256.x
Turner TF, Trexler JC, Kuhn DN, Robison HW (1996) Life history variation and comparative phylogeography of darters (Pisces: Percidae) from the North American Central Highlands. Evolution 50:2023–2036. https://doi.org/10.1111/j.1558-5646.1996.tb03589.x
USFWS (2010) Endangered and threatened wildlife and plants; review of native species that are candidates for listing as endangered or threatened; annual notice of findings on resubmitted petitions; annual description of progress on listing actions. Fed Reg 75:69222–69294
USFWS (2012) Genetic assessment of Abrams Creek reintroduction program for the federally threatened Yellowfin Madtom (Noturus flavipinnis), and endangered Smoky Madtom (Noturus baileyi) and Citico Darter (Etheostoma sitikuense). Project Report, Warm Springs, GA USFWS Conservation Genetics Lab
USFWS (2013) Range-wide conservation strategy for the Kentucky Arrow Darter (Etheostoma s. spilotum Gilbert). Frankfort, Kentucky
USFWS (2015) Endangered and threatened wildlife and plants; threatened species status for Kentucky Arrow Darter with 4(d) rule. Fed Reg 80:60962–60988
Waples RS, Do C (2008) LDNE: a program for estimating effective population size from data on linkage disequilibrium. Mol Ecol Resour 8:753–756. https://doi.org/10.1111/j.1755-0998.2007.02061.x
Waples RS, England PR (2011) Estimating contemporary effective population size on the basis of linkage disequilibrium in the face of migration. Genetics 189:633–644. https://doi.org/10.1534/genetics.111.132233
Warren ML Jr, Burr BM, Walsh SJ et al (2000) Diversity, distribution, and conservation status of the native freshwater fishes of the Southern United States. Fisheries 25:7–31
Weeks AR, Sgro CM, Young AG, Frankham R, Mitchell NJ, Miller KA, Byrne M, Coates DJ, Elderidge MDB, Sunnucks P, Breed MF, James EA, Hoffmann AA (2011) Assessing the benefits and riskds of translocations in changing environments: a genetic perspective. Evol Appl 4:709–725. https://doi.org/10.1111/j.1752-4571.2011.00192.x
Weir BS, Cockerham CC (1984) Estimating F-statistics for the analysis of population structure. Evolution 38:1358–1370. https://doi.org/10.2307/2408641
Whiteley AR, Fitzpatrick SW, Funk WC, Tallmon DA (2015) Genetic rescue to the rescue. TREE 30:42–149. https://doi.org/10.1016/j.tree.2014.10.009
Wigginton JE, Cutler DJ, Abecasis GR (2005) A note on exact tests of Hardy-Weinberg equilibrium. Am J Hum Genet 76:887–893. https://doi.org/10.1086/429864
Williamson-Natesan E (2005) Comparison of methods for detecting bottlenecks from microsatellite loci. Conserv Genet 6:551–562. https://doi.org/10.1007/s10592-005-9009-5
Young A, Clarke G (2000) Genetics, demography, and the viability of fragmented populations. Cambridge University Press, UK
Acknowledgements
Funding and resources provided through the Kentucky Department of Fish and Wildlife Resources (KDFWR), US Fish and Wildlife Service (USFWS) Section 6 funding, Austin Peay State University (APSU), and the APSU Center of Excellence for Field Biology. We thank S. Carr (KDFWR) for invaluable assistance facilitating contracts and other technical issues throughout the project; J. Johansen (TTU, APSU) for maps and georeferencing; S. Woltmann and (APSU) J. Johansen for helpful discussion related to data analyses and interpretation and comments on the manuscript; and E. Bloom (APSU), M. Lewis (APSU), K. Pilcher (APSU), and J. Johansen for lab assistance. S. Harrel (EKU) assisted in tissue collections. The findings and conclusions in this article are those of the authors and do not necessarily represent the views of the United States Fish and Wildlife Service.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10592_2019_1188_MOESM1_ESM.tif
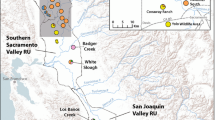
Supplementary material 1 (TIFF 117535 kb). Online Resource 1. Geographic distribution of Kentucky Arrow Darter (KAD), Etheostoma sagitta spilotum. Circles represent all historical localities known for the species; colors reflect presence (green) or absence (red) at a site as a result of recent survey efforts (2007-2013) to document occurrence (modified from USFWS 2013). Publicly owned lands are shown to demonstrate E. s. spilotum occurrence is now primarily restricted to these areas where mining activities, for example, have been less pervasive
10592_2019_1188_MOESM2_ESM.docx
Supplementary material 2 (DOCX 16 kb). Online Resource 2.. Locality information for populations examined in assessments of genetic diversity of the Kentucky Arrow Darter, Etheostoma sagitta spilotum. All sites were located in the Kentucky River system of Kentucky (Fig. 1). The population identifier (Pop. ID) number corresponds to those used throughout the document. SFK = South Fork Kentucky River; MFK = Middle Fork Kentucky River; NFK = North Fork Kentucky River
10592_2019_1188_MOESM3_ESM.eps
Supplementary material 3 (EPS 11161 kb). Online Resource 3. Population number (K) inferred from STRUCTURE analysis of Kentucky Arrow Darter, Etheostoma sagitta spilotum: (A) mean Log Likelihood; and (B) ΔK estimates. Both recover nine populations
Rights and permissions
About this article
Cite this article
Blanton, R.E., Cashner, M.F., Thomas, M.R. et al. Increased habitat fragmentation leads to isolation among and low genetic diversity within populations of the imperiled Kentucky Arrow Darter (Etheostoma sagitta spilotum). Conserv Genet 20, 1009–1022 (2019). https://doi.org/10.1007/s10592-019-01188-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-019-01188-y