Abstract
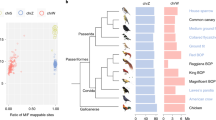
A hallmark feature of the male-specific region of the human Y chromosome is the presence of large and near-identical palindromes. These palindromes are maintained in a state of near identity via gene conversion between the arms of the palindrome, and both neutral and selection-based theories have been proposed to explain their enrichment on the human Y and X chromosomes. While those proposed theories would be applicable to sex chromosomes in other species, it has not been established whether near-identical palindromes are a common feature of sex chromosomes in a broader range of taxa, including other tetrapods. Here, we report the genomic sequencing and features of a 279-kb region of the non-recombining portion of the W chromosome spanning the CHD1W locus in a New World sparrow, the white-throated sparrow (Zonotrichia albicollis), and the corresponding region on the Z chromosome. As has been observed for other Y and W chromosomes, we detected a high repetitive element content (51%) and low gene content on the white-throated sparrow W chromosome. In addition, we identified a 22-kb near-identical (>99%) palindrome on the W chromosome that flanks the 5′ end of the CHD1W gene. Signatures of gene conversion were readily detected between the arms of this palindrome, as was the presence of this palindrome in other New World sparrows and blackbirds. Near-identical palindromes are therefore present on the avian W chromosome and may persist due to the same forces proposed for the enrichment of these elements on the human sex chromosomes.




Similar content being viewed by others
Abbreviations
- BAC:
-
Bacterial artificial chromosome
- CHD1W:
-
Chromo-helicase DNA-binding protein, W chromosome
- CHD1Z:
-
Chromo-helicase DNA-binding protein, Z chromosome
- CHORI:
-
Children’s Hospital Oakland Research Institute
- dNTP:
-
deoxy-nucleotide triphosphate
- HINTW:
-
Histidine triad nucleotide binding protein W
- kb:
-
Kilobase pairs
- MSCI:
-
Meiotic sex chromosome inactivation
- MSY:
-
Male-specific region of the Y chromosome
- PCR:
-
Polymerase chain reaction
- RASA1:
-
RAS p21 protein activator (GTPase activating protein) 1
- RGMB:
-
RGM domain family, member B
- UWBM:
-
University of Washington Burke Museum
References
Agate RJ, Choe M, Arnold AP (2004) Sex differences in structure and expression of the sex chromosome genes CHD1Z and CHD1W in zebra finches. Mol Biol Evol 21:384–396
Altschul SF, Madden TL, Schaffer AA et al (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Bachtrog D, Hom E, Wong KM, Maside X, De Jong P (2008) Genomic degradation of a young Y chromosome in Drosophila miranda. Genome Biol 9:R30
Backstrom N, Ceplitis H, Berlin S, Ellegren H (2005) Gene conversion drives the evolution of HINTW, an ampliconic gene on the female-specific avian W chromosome. Mol Biol Evol 22:1992–1999
Bagnall RD, Ayres KL, Green PM, Giannelli F (2005) Gene conversion and evolution of Xq28 duplicons involved in recurring inversions causing severe hemophilia A. Genome Res 15:214–223
Balakrishnan CN, Edwards SV (2009) Nucleotide variation, linkage disequilibrium and founder-facilitated speciation in wild populations of the zebra finch (Taeniopygia guttata). Genetics 181:645–660
Blakesley RW, Hansen NF, Mullikin JC et al (2004) An intermediate grade of finished genomic sequence suitable for comparative analyses. Genome Res 14:2235–2244
Burge C, Karlin S (1997) Prediction of complete gene structures in human genomic DNA. J Mol Biol 268:78–94
Caceres M, Sullivan RT, Thomas JW (2007) A recurrent inversion on the eutherian X chromosome. Proc Natl Acad Sci USA 104:18571–18576
Charlesworth B, Charlesworth D (2000) The degeneration of Y chromosomes. Philos Trans R Soc Lond B Biol Sci 355:1563–1572
Charlesworth D, Charlesworth B, Marais G (2005) Steps in the evolution of heteromorphic sex chromosomes. Heredity 95:118–128
Ellegren H, Carmichael A (2001) Multiple and independent cessation of recombination between avian sex chromosomes. Genetics 158:325–331
Ezaz T, Moritz B, Waters P, Marshall Graves JA, Georges A, Sarre SD (2009) The ZW sex microchromosomes of an Australian dragon lizard share no homology with those of other reptiles or birds. Chromosome Res 17:965–973
Fridolfsson AK, Ellegren H (2000) Molecular evolution of the avian CHD1 genes on the Z and W sex chromosomes. Genetics 155:1903–1912
Griffiths R, Double MC, Orr K, Dawson RJ (1998) A DNA test to sex most birds. Mol Ecol 7:1071–1075
Hillier LW, Miller W, Birney E et al (2004) Sequence and comparative analysis of the chicken genome provide unique perspectives on vertebrate evolution. Nature 432:695–716
Hori T, Asakawa S, Itoh Y, Shimizu N, Mizuno S (2000) Wpkci, encoding an altered form of PKCI, is conserved widely on the avian W chromosome and expressed in early female embryos: implication of its role in female sex determination. Mol Biol Cell 11:3645–3660
Hughes JF, Skaletsky H, Pyntikova T et al (2010) Chimpanzee and human Y chromosomes are remarkably divergent in structure and gene content. Nature 463:536–539
Huson DH, Bryant D (2006) Application of phylogenetic networks in evolutionary studies. Mol Biol Evol 23:254–267
Itoh Y, Kampf K, Arnold AP (2008) Molecular cloning of zebra finch W chromosome repetitive sequences: evolution of the avian W chromosome. Chromosoma 117:111–121
Itoh Y, Kampf K, Arnold AP (2009) Disruption of FEM1C-W gene in zebra finch: evolutionary insights on avian ZW genes. Chromosoma 118:323–334
Jeanmougin F, Thompson JD, Gouy M, Higgins DG, Gibson TJ (1998) Multiple sequence alignment with Clustal X. Trends Biochem Sci 23:403–405
Kondo M, Hornung U, Nanda I et al (2006) Genomic organization of the sex-determining and adjacent regions of the sex chromosomes of medaka. Genome Res 16:815–826
Kumar S, Tamura K, Nei M (2004) MEGA3: integrated software for molecular evolutionary genetics analysis and sequence alignment. Brief Bioinform 5:150–163
Lahn BT, Page DC (1999) Four evolutionary strata on the human X chromosome. Science 286:964–967
Lakich D, Kazazian HH Jr, Antonarakis SE, Gitschier J (1993) Inversions disrupting the factor VIII gene are a common cause of severe haemophilia A. Nat Genet 5:236–241
Lange J, Skaletsky H, Van Daalen SK et al (2009) Isodicentric Y chromosomes and sex disorders as byproducts of homologous recombination that maintains palindromes. Cell 138:855–869
Mank JE, Ellegren H (2007) Parallel divergence and degradation of the avian W sex chromosome. Trends Ecol Evol 22:389–391
Marshall Graves JA (2008) Weird animal genomes and the evolution of vertebrate sex and sex chromosomes. Annu Rev Genet 42:565–586
Matsubara K, Tarui H, Toriba M et al (2006) Evidence for different origin of sex chromosomes in snakes, birds, and mammals and step-wise differentiation of snake sex chromosomes. Proc Natl Acad Sci USA 103:18190–18195
Nam K, Ellegren H (2008) The chicken (Gallus gallus) Z chromosome contains at least three nonlinear evolutionary strata. Genetics 180:1131–1136
Ohno S (1967) Sex chromosomes and sex-linked genes. Springer-Verlag, New York
Rossiter JP, Young M, Kimberland ML et al (1994) Factor VIII gene inversions causing severe hemophilia A originate almost exclusively in male germ cells. Hum Mol Genet 3:1035–1039
Rozen S, Skaletsky H, Marszalek JD et al (2003) Abundant gene conversion between arms of palindromes in human and ape Y chromosomes. Nature 423:873–876
Sawyer S (1989) Statistical tests for detecting gene conversion. Mol Biol Evol 6:526–538
Schoenmakers S, Wassenaar E, Hoogerbrugge JW, Laven JS, Grootegoed JA, Baarends WM (2009) Female meiotic sex chromosome inactivation in chicken. PLoS Genet 5:e1000466
Schwartz S, Zhang Z, Frazer KA et al (2000) PipMaker—a web server for aligning two genomic DNA sequences. Genome Res 10:577–586
Skaletsky H, Kuroda-Kawaguchi T, Minx PJ et al (2003) The male-specific region of the human Y chromosome is a mosaic of discrete sequence classes. Nature 423:825–837
Sperber G, Lovgren A, Eriksson NE, Benachenhou F, Blomberg J (2009) RetroTector online, a rational tool for analysis of retroviral elements in small and medium size vertebrate genomic sequences. BMC Bioinform 10(6):S4
Sullivan RT, Morehouse CB, Thomas JW (2008) Uprobe 2008: an online resource for universal overgo hybridization-based probe retrieval and design. Nucleic Acids Res 36:W149–W153
Thomas JW (2008) Comparative physical mapping: universal overgo hybridization probe design and BAC library hybridization. In: Murphy WJ (ed) Methods in molecular biology: phylogenomics. Humana Press, Totowa, pp 119–132
Thomas JW, Prasad AB, Summers TJ et al (2002) Parallel construction of orthologous sequence-ready clone contig maps in multiple species. Genome Res 12:1277–1285
Vibranovski MD, Koerich LB, Carvalho AB (2008) Two new Y-linked genes in Drosophila melanogaster. Genetics 179:2325–2357
Warburton PE, Giordano J, Cheung F, Gelfand Y, Benson G (2004) Inverted repeat structure of the human genome: the X-chromosome contains a preponderance of large, highly homologous inverted repeats that contain testes genes. Genome Res 14:1861–1869
Warren WC, Clayton DF, Ellegren H et al (2010) The genome of a songbird. Nature 464:757–762
Wheelan SJ, Church DM, Ostell JM (2001) Spidey: a tool for mRNA-to-genomic alignments. Genome Res 11:1952–1957
Wilson MA, Makova KD (2009) Genomic analyses of sex chromosome evolution. Annu Rev Genomics Hum Genet 10:333–354
Yu Q, Hou S, Hobza R et al (2007) Chromosomal location and gene paucity of the male specific region on papaya Y chromosome. Mol Genet Genomics 278:177–185
Yuri T, Mindell DP (2002) Molecular phylogenetic analysis of Fringillidae, “New World nine-primaried oscines” (Aves: Passeriformes). Mol Phylogenet Evol 23:229–243
Zhang Z, Schwartz S, Wagner L, Miller W (2000) A greedy algorithm for aligning DNA sequences. J Comput Biol 7:203–214
Zink RM, Dittmann DL, Rootes WL (1991) Mitochrondrial DNA variation and the phylogeny of Zonotrichia. Auk 108:578–584
Internet references
Smit AFA, Hubley R, Green P (1996–2004) RepeatMasker Open-3.0. http://www.repeatmasker.org
Acknowledgements
The authors thank Judith E. Mank for helpful comments and discussions, Mario Cáceres for comments, Cheryl T. Strauss for technical writing edits, Donna L. Maney for the dark-eyed junco DNA, Greg K. Tharp for computer support, the BC Cancer Agency Genome Sciences Centre, Vancouver, Canada, for generation of the BAC-end sequences, and members of the NIH Sequencing Center including E.D. Green, R. Blakesley, G. Bouffard, and J. McDowell. DNA samples (excluding the dark-eyed junco) were provided by The Burke Museum of Natural History and Culture. J.K.D. and J.W.T. were supported by a grant from the National Institutes of Health (1R21MH082046), and the NIH Intramural Sequencing Center was supported in part by the Intramural Research Program of the National Human Genome Research Institute of the National Institutes of Health.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
Responsible Editor: Mary E. Delany
Rights and permissions
About this article
Cite this article
Davis, J.K., Thomas, P.J., NISC Comparative Sequencing Program. et al. A W-linked palindrome and gene conversion in New World sparrows and blackbirds. Chromosome Res 18, 543–553 (2010). https://doi.org/10.1007/s10577-010-9134-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10577-010-9134-y