Abstract
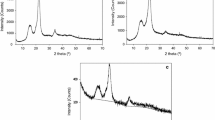
Microcrystalline cellulose (MCC) particles were subjected to hydrothermal treatment using an autoclave with temperatures ranging from 200 to 250 °C and reaction times ranging from 20 to 100 min. The structure and chemical composition of the reacted solid phase was analyzed by X-ray diffraction, thermo-gravimetric analysis, FTIR spectroscopy and 13C-NMR spectroscopy. The relative composition of the water-soluble products was determined by one-dimensional 1H-NMR and two-dimensional homo and hetero-nuclear NMR spectroscopy. Within the experimental temperature and treatment time ranges, the crystallinity of the reacted solid phase was found to be mostly dependent on the treatment temperature while the aqueous solution was found to change with both temperature and treatment time. At the maximum temperature employed in this study (250 °C), the solid products are similar to amorphous oxidized carbon with glucose as the main water-soluble product. At lower temperatures the particles are unconverted MCC and the liquid products are primarily levulinic acid, formic acid and acetic acid with smaller quantities of 5-hydroxymethyl-furfural and glucose. Heterogeneous and liquid phase reaction-schemes are proposed to explain the observed solid and water-soluble products as a function of temperature and treatment time.













Similar content being viewed by others
References
Agarwal UP, Reiner RS, Ralph SA (2010) Cellulose I crystallinity determination using FT-Raman spectroscopy: univariate and multivariate methods. Cellulose 17:721–733
Atalla RH, Gast JC, Sindorf DW, Bartuska VJ, Maciel GE (1980) C-13 NMR-spectra of cellulose polymorphs. J Am Chem Soc 102:3249–3251
Bansal P, Hall M, Realff MJ, Lee JH, Bommarius AS (2010) Multivariate statistical analysis of X-ray data from cellulose: a new method to determine degree of crystallinity and predict hydrolysis rates. Biores Tech 101:4461–4471
Cantero DA, Dolores Bermejo M, Jose Cocero M (2013) Kinetic analysis of cellulose depolymerization reactions in near critical water. J Supercrit Fluids 75:48–57
Deguchi S, Tsujii K, Horikoshi K (2008) Crystalline-to-amorphous transformation of cellulose in hot and compressed water and its implications for hydrothermal conversion. Green Chem 10:191–196
Driemeier C, Calligaris GA (2011) Theoretical and experimental developments for accurate determination of crystallinity of cellulose I materials. J Appl Cryst 44:184–192
Earl WL, Vanderhart DL (1980) High-resolution, magic angle sample spinning C-13 NMR of solid cellulose-I. J Am Chem Soc 102:3251–3252
Ehara K, Saka S (2002) A comparative study on chemical conversion of cellulose between the batch-type and flow-type systems in supercritical water. Cellulose 9:301–311
French AD, Santiago Cintrón M (2013) Cellulose polymorphy, crystallite size, and the Segal crystallinity index. Cellulose 20:583–588
Gallezot P (2012) Conversion of biomass to selected chemical products. Chem Soc Rev 41:1538–1558
Horii F, Hirai A, Kitamaru R (1987) CP/MAS C-13 NMR-spectra of the crystalline components of native celluloses. Macromolecules 20:2117–2120
Huber GW (2008) Breaking the chemical and engineering barriers to lignocellulosic biofuels. National Science Foundation Report, USA
Kabyemela BM, Adschiri T, Malaluan RM, Arai K (1997) Kinetics of glucose epimerization and decomposition in subcritical and supercritical water. Ind Eng Chem Res 36:1552–1558
Kamio E, Takahashi S, Noda H, Fukuhara C, Okamura T (2006) Liquefaction of cellulose in hot compressed water under variable temperatures. Ind Eng Chem Res 45:4944–4953
Kamio E, Sato H, Takahashi S, Noda H, Fukuhara C, Okamura T (2008) Liquefaction kinetics of cellulose treated by hot compressed water under variable temperature conditions. J Mater Sci 43:2179–2188
Kumar S, Gupta RB (2008) Hydrolysis of microcrystalline cellulose in subcritical and supercritical water in a continuous flow reactor. Ind Eng Chem Res 47:9321–9329
Leppanen K, Andersson S, Torkkeli M, Knaapila M, Kotelnikova N, Serimaa R (2009) Structure of cellulose and microcrystalline cellulose from various wood species, cotton and flax studied by X-ray scattering. Cellulose 16:999–1015
Masson JF, Manley RS (1992) Solid-state NMR of some cellulose synthetic polymer blends. Macromolecules 25:589–592
Mettler MS, Mushrif SH, Paulsen AD, Javadekar AD, Vlachos DG, Dauenhauer PJ (2012) Revealing pyrolysis chemistry for biofuels production: conversion of cellulose to furans and small oxygenates. Energy Environ Sci 5:5414–5424
Moon RJ, Martini A, Nairn J, Simonsen J, Youngblood J (2011) Cellulose nanomaterials review: structure, properties and nanocomposites. Chem Soc Rev 40:3941–3994
Newman RH, Hemmingson JA (1995) C-13 NMR distinction between categories of molecular order and disorder in cellulose. Cellulose 2:95–110
Nishiyama Y, Langan P, Chanzy H (2002) Crystal structure and hydrogen-bonding system in cellulose 1 beta from synchrotron X-ray and neutron fiber diffraction. J Am Chem Soc 124:9074–9082
NIST (2005) Standard reference database number 69, NIST chemistry WebBook, National Institute of Standards and Technology, Gaithersburg MD, 20899. http://webbook.nist.gov
Park S, Baker JO, Himmel ME, Parilla PA, Johnson DK (2010) Cellulose crystallinity index: measurement techniques and their impact on interpreting cellulase performance. Biotechnol Biofuels 3:1–10
Sasaki M, Fang Z, Fukushima Y, Adschiri T, Arai K (2000) Dissolution and hydrolysis of cellulose in subcritical and supercritical water. Ind Eng Chem Res 39:2883–2890
Scherrer P (1918) Bestimmung der Größe und der inneren Struktur von Kolloidteilchen mittels Röntgenstrahlen. Nachr Ges Wiss Gottingen 26:98–100
Schwanninger M, Rodrigues JC, Pereira H, Hinterstoisser B (2004) Effects of short-time vibratory ball milling on the shape of FT-IR spectra of wood and cellulose. Vib Spectrosc 36:23–40
Seehra MS, Akkineni LP, Yalamanchi M, Singh V, Poston J (2012) Structural characteristics of nanoparticles produced by hydrothermal pretreatment of cellulose and their applications for electrochemical hydrogen generation. Int J Hydrog Energy 37:9514–9523
Segal L, Creely JJ, Martin AE, Conrad CM (1959) An empirical method for estimating the degree of crystallinity of native cellulose using the X-ray diffractometer. Text Res J 29:786–794
Srokol Z, Bouche AG, van Estrik A, Strik RCJ, Maschmeyer T, Peters JA (2004) Hydrothermal upgrading of biomass to biofuel; studies on some monosaccharide model compounds. Carbohydr Res 339:1717–1726
Thygesen A, Oddershede J, Lilholt H, Thomsen AB, Stahl K (2005) On the determination of crystallinity and cellulose content in plant fibres. Cellulose 12:563–576
Titirici M-M, Antonietti M, Baccile N (2008) Hydrothermal carbon from biomass: a comparison of the local structure from poly-to monosaccharides and pentoses/hexoses. Green Chem 10:1204–1212
Toor SS, Rosendahl L, Rudolf A (2011) Hydrothermal liquefaction of biomass: a review of subcritical water technologies. Energy 36:2328–2342
Yaws CL, Narasimhan PK, Gabbula C (2009) Yaws' handbook of Antoine coefficients for vapor pressure (2nd Electronic Edition). Knovel. Online
Yu Y, Wu H (2010) Significant differences in the hydrolysis behavior of amorphous and crystalline portions within microcrystalline cellulose in hot-compressed water. Ind Eng Chem Res 49:3902–3909
Acknowledgments
We gratefully acknowledge financial support from West Virginia University. MSS also acknowledges financial support from the U.S. Department of Energy (Contract # DE-FC26-05NT42456) during the initial stages of the project. Funding for solution NMR instrumentation was provided by the National Science Foundation (CHE-1228336).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Seehra, M.S., Popp, B.V., Goulay, F. et al. Hydrothermal treatment of microcrystalline cellulose under mild conditions: characterization of solid and liquid-phase products. Cellulose 21, 4483–4495 (2014). https://doi.org/10.1007/s10570-014-0424-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-014-0424-y