Abstract
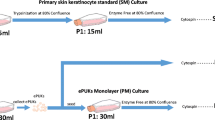
Our laboratory had developed a methodology to expand epithelial cells in culture by growing keratinocyte monolayers, under large volumes of medium that produces large numbers of keratinocytes that leave the monolayer and move into suspension. The cells have been defined as epithelial Pop Up Keratinocytes or ePUKs cells and appear to be highly suitable for clinical applications. In this publication we extend the characterization of the cells with a detailed analysis of the capabilities of the monolayer of a single culture flask to produce, over time, ePUK cells. The cells were characterized using standard epithelial markers for proliferation and differentiation. Analysis of morphology of the monolayer formed and total number of cells produced is presented for a variety of human epithelial cell strains. These keratinocytes provide an additional controlled human cell system for investigation of the mechanisms regulating epithelia cell growth and differentiation and since they are produced in large numbers, they are highly suitable for use in epithelial cell banking.







Similar content being viewed by others
References
Bavister BD, Wolf DP, Brenner CA (2005) Challenges of primate embryonic stem cell research. Clon Stem Cells 7:82–94
Dragúňová J, Kabát P, Koller J (2012) Skin explant cultures as a source of keratinocytes for cultivation. Cell Tissue Bank. doi:10.1007/s10561-012-9330-4
Duncan CO, Shelton RM, Navsaria H, Balderson DS, Papini RP, Barralet JE (2005) In vitro transfer of keratinocytes: comparison of transfer from fibrin membrane and delivery by aerosol spray. J Biomed Mater Res B Appl Biomater 73:221–228
Hodgkinson CP, Gomez JA, Mirotsou M, Dzau VJ (2010) Genetic engineering of mesenchymal stem cells and its application in human diseases therapy. Hum Gene Ther 21:1513–1526
Lei P, Andreadis ST (2008) Efficient retroviral gene transfer to epidermal stem cells. Methods Mol Biol 433:367–379
Lister R, Pelizzola M, Kida YS, Hawkins RD, Nery JR, Hon G, Antosiewicz-Bourget J, O’Malley R, Castanon R, Klugman S, Downes M, Yu R, Stewart R, Ren B, Thomson JA, Evans RM, Ecker JR (2011) Hotspots of aberrant epigenomic reprogramming in human induced pluripotent stem cells. Nature 471:68–73
Marcelo CL, Duell EA, Rhodes LM, Dunham WR (1992) In vitro model of essential fatty acid deficiency. J Invest Dermatol 99:703–708
Marcelo CL, Peramo A, Ambati A, Feinberg SE (2012) Characterization of an unique technique for culturing primary epithelial progenitor/stem-cells. BMC Dermatol 12:8
Mignone JL, Kreutziger KL, Paige SL, Murry CE (2010) Cardiogenesis from human embryoinic stem cells. Circ J 74:2517–2526
Oliveira AA Jr, Hodges HM (2005) Alzheimer’s disease and neural transplantation as prospective cell therapy. Curr Alzheimer Res 2:79–95
Pellegrini G, Dellambra R, Golisano O, Martinelli E, Fantozzi I, Bondanza S, Ponzin D, McKeon F, De Luca M (2001) P63 identifies keratinocytes stem cells. Proc Natl Acad Sci USA 98:3156–3161
Webb A, Li A, Kaur P (2004) Location and phenotype of human adult keratinocyte strain cells of the skin. Differentiation 72:387–395
Acknowledgments
This work was supported by grant R01AM-26009 to CLM.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10561_2012_9343_MOESM2_ESM.tif
Figure S1 Comparison of cell production for the pairs ePUK0-ePUK1 for different cell strains. ePUK production of frozen strains show a substantial decrease in the ePUK1 flasks. In fresh cells, neonatal strains maintain a similar production from the beginning, but oral ePUK1 cells grow more slowly (TIFF 180 kb)
Rights and permissions
About this article
Cite this article
Peramo, A., Feinberg, S.E. & Marcelo, C.L. Characterization of cultured epithelial cells using a novel technique not requiring enzymatic digestion for subculturing. Cell Tissue Bank 14, 423–435 (2013). https://doi.org/10.1007/s10561-012-9343-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-012-9343-z