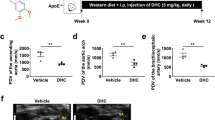
Abstract
Purpose
Hypothyroidism (HT) is associated with accelerated atherosclerosis (AS). The efficacy of traditional strategies of hypothyroid AS remains controversial. Here, we aimed to deepen the understanding of the HT-induced acceleration of AS, to decrease the residual risk of coronary artery disease (CAD) and to find a new therapeutic target.
Methods
We collected peripheral venous blood samples from 20 patients and divided them into 4 groups, namely, the normal group, the HT group, the CAD group and the HT + CAD group. Then we performed mRNA microarray analysis and bioinformatics analysis to screen the differentially expressed genes and pathways, and we also conducted validations on ApoE knockout mice models and Raw264.7 cell models.
Results
In short, (1) in the analysis between the CAD group and the HT + CAD group, we found a total of 1218 differentially expressed genes, 11 upregulated pathways and 40 downregulated pathways. (2) We validated that patients with HT and CAD had a significantly decreased expression of MAP3K7 (encoding transforming growth factor-β-activated kinase 1, TAK1) gene than normal subjects. (3) In animal and cell experiments, we found the decreased expression of TAK1 and the reduced phosphorylation of AMP-activated protein kinase (AMPK) under the hypothyroid and atherosclerotic condition. (4) Changes in the expressions of TAK1 may affect the progression of AS.
Conclusion
Taken together, these data suggest that the accelerated AS in hypothyroid patients may be due to the suppression of TAK1-AMPK pathway in macrophages. This new finding may become a novel therapeutic target in hypothyroid AS.





Similar content being viewed by others
References
Moran AE, Forouzanfar MH, Roth GA, Mensah GA, Ezzati M, Murray CJL, et al. Temporal trends in ischemic heart disease mortality in 21 world regions, 1980 to 2010: the global burden of disease 2010 study. Circulation. 2014;129(14):1483–92.
Piepoli MF, Hoes AW, Agewall S, Albus C, Brotons C, Catapano AL, et al. 2016 European guidelines on cardiovascular disease prevention in clinical practice: the sixth joint task force of the European Society of Cardiology and Other Societies on cardiovascular disease prevention in clinical practice (constituted by representatives of 10 societies and by invited experts)developed with the special contribution of the European Association for Cardiovascular Prevention & Rehabilitation (EACPR). Eur Heart J. 2016;37(29):2315–81.
Jabbar A, Pingitore A, Pearce SH, et al. Thyroid hormones and cardiovascular disease. Nat Rev Cardiol. 2017;14(1):39–55.
Ning Y, Cheng YJ, Liu LJ, Sara JDS, Cao ZY, Zheng WP, et al. What is the association of hypothyroidism with risks of cardiovascular events and mortality? A meta-analysis of 55 cohort studies involving 1,898,314 participants. BMC Med. 2017;15(1):21.
Zhang M, Sara JD, Matsuzawa Y, et al. Clinical outcomes of patients with hypothyroidism undergoing percutaneous coronary intervention. Eur Heart J. 2016;37(26):2055–65.
Sampson UK, Fazio S, Linton MF. Residual cardiovascular risk despite optimal LDL cholesterol reduction with statins: the evidence, etiology, and therapeutic challenges. Curr Atheroscler Rep. 2012;14(1):1–10.
Wong ND, Zhao Y, Quek RGW, Blumenthal RS, Budoff MJ, Cushman M, et al. Residual atherosclerotic cardiovascular disease risk in statin-treated adults: the multi-ethnic study of atherosclerosis. Journal of clinical lipidology. 2017;11(5):1223–33.
Aziz M, Kandimalla Y, Machavarapu A, Saxena A, Das S, Younus A, et al. Effect of thyroxin treatment on carotid intima-media thickness (CIMT) reduction in patients with subclinical hypothyroidism (SCH): a meta-analysis of clinical trials. J Atheroscler Thromb. 2017;24(7):643–59.
Blum MR, Gencer B, Adam L, Feller M, Collet TH, da Costa BR, et al. Impact of thyroid hormone therapy on atherosclerosis in the elderly with subclinical hypothyroidism: a randomized trial. J Clin Endocrinol Metab. 2018;103(8):2988–97.
Mooijaart SP, Du Puy RS, Stott DJ, et al. Association between levothyroxine treatment and thyroid-related symptoms among adults aged 80 years and older with subclinical hypothyroidism. Jama. 2019:1–11.
Biondi B, Cappola AR, Cooper DS. Subclinical hypothyroidism: a review. Jama. 2019;322(2):153–60.
Lu C, Xiong M, Luo Y, Li J, Zhang Y, Dong Y, et al. Genome-wide transcriptional analysis of apoptosis-related genes and pathways regulated by H2AX in lung cancer A549 cells. Apoptosis : an international journal on programmed cell death. 2013;18(9):1039–47.
Jeon US, Choi JP, Kim YS, Ryu SH, Kim YK. The enhanced expression of IL-17-secreting T cells during the early progression of atherosclerosis in ApoE-deficient mice fed on a western-type diet. Exp Mol Med. 2015;47:e163.
Bianco AC, Anderson G, Forrest D, Galton VA, Gereben B, Kim BW, et al. American Thyroid Association guide to investigating thyroid hormone economy and action in rodent and cell models. Thyroid : official journal of the American Thyroid Association. 2014;24(1):88–168.
Hu HJ, Shi ZY, Lin XL, Chen SM, Wang QY, Tang SY. Upregulation of sestrin2 expression protects against macrophage apoptosis induced by oxidized low-density lipoprotein. DNA Cell Biol. 2015;34(4):296–302.
Chang YC, Lee TS, Chiang AN. Quercetin enhances ABCA1 expression and cholesterol efflux through a p38-dependent pathway in macrophages. J Lipid Res. 2012;53(9):1840–50.
Orning P, Weng D, Starheim K, Ratner D, Best Z, Lee B, et al. Pathogen blockade of TAK1 triggers caspase-8-dependent cleavage of gasdermin D and cell death. Science. 2018;362(6418):1064–9.
Cheng Z, Chu H, Wang S, Huang Y, Hou X, Zhang Q, et al. TAK1 knock-down in macrophage alleviate lung inflammation induced by black carbon and aged black carbon. Environ Pollut. 2019;253:507–15.
Singh AK, Haque M, O'Sullivan K, et al. Suppression of monosodium urate crystal-induced inflammation by inhibiting TGF-beta-activated kinase 1-dependent signaling: role of the ubiquitin proteasome system. Cell Mol Immunol. 2019.
Ou H, Liu C, Feng W, Xiao X, Tang S, Mo Z. Role of AMPK in atherosclerosis via autophagy regulation. Sci China Life Sci. 2018;61(10):1212–21.
Aashaq S, Batool A, Andrabi KI. TAK1 mediates convergence of cellular signals for death and survival. Apoptosis : an international journal on programmed cell death. 2019;24(1–2):3–20.
Aoki Y, Belin RM, Clickner R, Jeffries R, Phillips L, Mahaffey KR. Serum TSH and total T4 in the United States population and their association with participant characteristics: National Health and nutrition examination survey (NHANES 1999-2002). Thyroid : official journal of the American Thyroid Association. 2007;17(12):1211–23.
Chaker L, Bianco AC, Jonklaas J, et al. Hypothyroidism. Lancet (London, England). 2017;390(10101):1550–62.
Yang C, Lu M, Chen W, He Z, Hou X, Feng M, et al. Thyrotropin aggravates atherosclerosis by promoting macrophage inflammation in plaques. J Exp Med. 2019;216(5):1182–98.
Bano A, Chaker L, Mattace-Raso FUS, van der Lugt A, Ikram MA, Franco OH, et al. Thyroid function and the risk of atherosclerotic cardiovascular morbidity and mortality: the Rotterdam study. Circ Res. 2017;121(12):1392–400.
Rittenhouse PA, Redei E. Thyroxine administration prevents streptococcal cell wall-induced inflammatory responses. Endocrinology. 1997;138(4):1434–9.
De Vito P, Incerpi S, Pedersen JZ, et al. Thyroid hormones as modulators of immune activities at the cellular level. Thyroid : official journal of the American Thyroid Association. 2011;21(8):879–90.
Grais IM, Sowers JR. Thyroid and the heart. Am J Med. 2014;127(8):691–8.
Taddei S, Caraccio N, Virdis A, Dardano A, Versari D, Ghiadoni L, et al. Impaired endothelium-dependent vasodilatation in subclinical hypothyroidism: beneficial effect of levothyroxine therapy. J Clin Endocrinol Metab. 2003;88(8):3731–7.
Razvi S, Ingoe L, Keeka G, Oates C, McMillan C, Weaver JU. The beneficial effect of L-thyroxine on cardiovascular risk factors, endothelial function, and quality of life in subclinical hypothyroidism: randomized, crossover trial. J Clin Endocrinol Metab. 2007;92(5):1715–23.
Andersen MN, Olsen AS, Madsen JC, et al. Long-term outcome in levothyroxine treated patients with subclinical hypothyroidism and concomitant heart disease. J Clin Endocrinol Metab. 2016;101(11):4170–7.
Ajibade AA, Wang HY, Wang RF. Cell type-specific function of TAK1 in innate immune signaling. Trends Immunol. 2013;34(7):307–16.
Ninomiya-Tsuji J, Kishimoto K, Hiyama A, Inoue JI, Cao Z, Matsumoto K. The kinase TAK1 can activate the NIK-I kappaB as well as the MAP kinase cascade in the IL-1 signalling pathway. Nature. 1999;398(6724):252–6.
Neumann D. Is TAK1 a direct upstream kinase of AMPK? International journal of molecular sciences. 2018;19(8).
Adhikari A, Xu M, Chen ZJ. Ubiquitin-mediated activation of TAK1 and IKK. Oncogene. 2007;26(22):3214–26.
Pagliari LJ, Perlman H, Liu H, Pope RM. Macrophages require constitutive NF-kappaB activation to maintain A1 expression and mitochondrial homeostasis. Mol Cell Biol. 2000;20(23):8855–65.
Mihaylova MM, Shaw RJ. The AMPK signalling pathway coordinates cell growth, autophagy and metabolism. Nat Cell Biol. 2011;13(9):1016–23.
Liao X, Sluimer JC, Wang Y, Subramanian M, Brown K, Pattison JS, et al. Macrophage autophagy plays a protective role in advanced atherosclerosis. Cell Metab. 2012;15(4):545–53.
Gautier EL, Huby T, Witztum JL, Ouzilleau B, Miller ER, Saint-Charles F, et al. Macrophage apoptosis exerts divergent effects on atherogenesis as a function of lesion stage. Circulation. 2009;119(13):1795–804.
Sergin I, Evans TD, Zhang X, Bhattacharya S, Stokes CJ, Song E, et al. Exploiting macrophage autophagy-lysosomal biogenesis as a therapy for atherosclerosis. Nat Commun. 2017;8:15750.
Sanjo H, Nakayama J, Yoshizawa T, et al. Cutting edge: TAK1 safeguards macrophages against proinflammatory cell death. Journal of Immunology (Baltimore, Md : 1950). 2019;203(4):783–8.
Day EA, Ford RJ, Steinberg GR. AMPK as a therapeutic target for treating metabolic diseases. Trends Endocrinol Metab. 2017;28(8):545–60.
Kinugawa K, Jeong MY, Bristow MR, et al. Thyroid hormone induces cardiac myocyte hypertrophy in a thyroid hormone receptor alpha1-specific manner that requires TAK1 and p38 mitogen-activated protein kinase. Molecular Endocrinology (Baltimore, Md). 2005;19(6):1618–28.
Sinha RA, Singh BK, Zhou J, Wu Y, Farah BL, Ohba K, et al. Thyroid hormone induction of mitochondrial activity is coupled to mitophagy via ROS-AMPK-ULK1 signaling. Autophagy. 2015;11(8):1341–57.
Matesanz N, Bernardo E, Acin-Perez R, et al. MKK6 controls T3-mediated browning of white adipose tissue. Nat Commun. 2017;8(1):856.
Videla LA, Vargas R, Riquelme B, et al. Thyroid hormone-induced expression of the hepatic scaffold proteins Sestrin2, β-Klotho, and FRS2α in relation to FGF21-AMPK signaling. Exp Clin Endocrinol Diabetes. 2018;126(03):182–6.
Wang P, Xu TY, Guan YF, Zhao Y, Li ZY, Lan XH, et al. Vascular smooth muscle cell apoptosis is an early trigger for hypothyroid atherosclerosis. Cardiovasc Res. 2014;102(3):448–59.
Acknowledgements
The authors thank Weiping Tang (Cnkingbio Company Ltd., Beijing, China) for bioinformatics assistance.
Funding
The work was supported by the Natural Science Foundation of China (No. 81670322); the Beijing Natural Science Foundation (No. 7182047); and the Beijing Science and Technology Nova Program Interdisciplinary Fund (No. Z181100006218133).
Author information
Authors and Affiliations
Contributions
MZ, YXY, YN, YWQ, HNZ, YHD and LYL conceived the idea and designed the study. YN and WWW performed the literature search. YXY, YN, WWW, YFJ and MLH performed the statistical analysis. YXY wrote the first draft. MZ, YXY, YFJ, XLJ and YYY made substantial contributions to study conception and design, interpretation of results and revising the manuscript critically for intellectual content. GHL provided theoretical guidance. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Informed Consent
All patients signed written informed consent.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplemental Figure S1
Microarray analysis and bioinformation analysis in human peripheral blood. Based on the KEGG database, pathways with p <0.05 and FDR < 0.05 were identified as significantly changed pathways. The y-axis shows significantly changed pathways and the x-axis, -Lg P. The larger -Lg P indicates a smaller p-value. ( a ) Significantly enriched pathways in human peripheral blood from all 4 groups (n = 5 in each group). ( b ) Significantly up-regulated pathways in human peripheral blood from the CAD group and the HT + CAD group. Fisher's exact test was used to choose the significant pathways. HT: hypothyroidism; CAD: coronary artery disease. (PNG 3681 kb)
Supplemental Figure S2
Representative images for transfection of lentivirus targeting MAP3K7 (LV- MAP3K7 ) in Raw264.7 cells. Representative images of Raw264.7 cells after 10-15 days of LV- MAP3K7 infection are shown. A fluorescence microscope was used to measure GFP (green fluorescent protein) expression (Scale bars = 100 μm). (PNG 1430 kb)
Supplemental Figure S3
Representative Oil Red O stained cells (left) and quantitative assessment (right) (n = 3). (a) 7 5Z-7-Oxozeaenol treated cells exhibited increased accumulation of lipid droplets. RAW264.7 cells were stimulated with 8 or without ox-LDL in the presence or absence of 5Z-7-Oxozeaenol. (b) After the stimulation of ox-LDL, MAP3K7 -9 overexpressing cells ( MAP3K7 -OE) showed reduced accumulation of lipid droplets than control cells (Ctrl-OE, Raw264.7 cells infected with LV-NC). ANOVA followed by Tukey test was used for statistical analysis between 11 experimental sets (Scale bars = 100 μm). ox-LDL: 100 μg/ml, 24 hours; 5Z-7-Oxozeaenol: TAK1 inhibitor, 5 μM, 1 hour; LV-NC: negative control lentivirus. (PNG 5358 kb)
Supplemental Figure S4
The top 35 genes that appear most frequently in the down-regulated pathway between CAD patients and HT + CAD patients. The y-axis shows the number of involved pathways and the x-axis, genes. HT: hypothyroidism; CAD: coronary artery disease. (PNG 4486 kb)
Table S1
(DOCX 17 kb)
Table S2
(DOCX 14 kb)
Rights and permissions
About this article
Cite this article
Yang, Y., Jia, Y., Ning, Y. et al. TAK1-AMPK Pathway in Macrophages Regulates Hypothyroid Atherosclerosis. Cardiovasc Drugs Ther 35, 599–612 (2021). https://doi.org/10.1007/s10557-020-06996-w
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-020-06996-w