Abstract
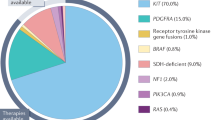
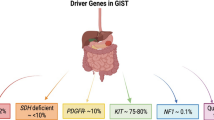
Gastrointestinal stromal tumors (GIST) are rare neoplasms arising from the interstitial cell of Cajal in the gastrointestinal tract. Two thirds of GIST in adult patients have c-Kit mutation and smaller fractions have platelet derived growth factor receptor alpha (PDGFRA) mutation. Surgery is the only curative treatment for localized disease. Imatinib improves survival when used adjuvantly and in advanced disease. Several targeted therapies have also improved survival in GIST patients after progression on imatinib including sunitinib and regorafenib. Recently, United States Federal and Drug Administration (FDA) approved two new tyrosine kinase inhibitors for the treatment of heavily pretreated advanced/unresectable GIST including avapritinib (a selective inhibitor for PDGFRA exon 18 mutation including D842V mutations) and ripretinib (a broad-spectrum kinase inhibitor of c-Kit and PDGFRA). In this article, we will provide a comprehensive review of GIST including the current standard of care treatment and exploring future paradigm shifts in therapy.


Similar content being viewed by others
References
Miettinen, M., & Lasota, J. (2001). Gastrointestinal stromal tumors-Definition, clinical, histological, immunohistochemical, and molecular genetic features and differential diagnosis. Virchows Archiv.
Miettinen, M., Sarlomo-Rikala, M., & Lasota, J. (1999). Gastrointestinal stromal tumors: Recent advances in understanding of their biology. Human Pathology.
Nilsson, B., Bümming, P., Meis-Kindblom, J. M., et al. (2005). Gastrointestinal stromal tumors: The incidence, prevalence, clinical course, and prognostication in the preimatinib mesylate era-A population-based study in western Sweden. Cancer.
Szucs, Z., Thway, K., Fisher, C., et al. (2017). Molecular subtypes of gastrointestinal stromal tumors and their prognostic and therapeutic implications. Oncol: Futur.
Newman, P. L., Wadden, C., & Fletcher, C. D. M. (1991). Gastrointestinal stromal tumours: Correlation of immunophenotype with clinicopathological features. The Journal of Pathology.
Sircar, K., Hewlett, B. R., Huizinga, J. D., Chorneyko, K., Berezin, I., & Riddell, R. H. (1999). Interstitial cells of cajal as precursors of gastrointestinal stromal tumors. The American Journal of Surgical Pathology.
Kollár, A., Aguiar, P. N., Forones, N. M., & De Mello, R. A. (2019). Gastrointestinal stromal tumor (GIST): Diagnosis and treatment. In International Manual of Oncology Practice.
Ma, G. L., Murphy, J. D., Martinez, M. E., & Sicklick, J. K. (2015). Epidemiology of gastrointestinal stromal tumors in the era of histology codes: Results of a population-based study. Cancer Epidemiology, Biomarkers & Prevention.
Tran, T., Davila, J. A., & El-Serag, H. B. (2005). The epidemiology of malignant gastrointestinal stromal tumors: An analysis of 1, 458 cases from 1992 to 2000. The American Journal of Gastroenterology.
Tryggvason, G., Gíslason, H. G., Magnússon, M. K., & Jónasson, J. G. (2005). Gastrointestinal stromal tumors in Iceland, 1990-2003: The Icelandic GIST study, a population-based incidence and pathologic risk stratification study. International Journal of Cancer.
Goettsch, W. G., Bos, S. D., Breekveldt-Postma, N., Casparie, M., Herings, R. M. C., & Hogendoorn, P. C. W. (2005). Incidence of gastrointestinal stromal tumours is underestimated: Results of a nation-wide study. European Journal of Cancer.
Tzen, C. Y., Wang, J. H., Huang, Y. J., et al. (2007). Incidence of gastrointestinal stromal tumor: A retrospective study based on immunohistochemical and mutational analyses. Digestive Diseases and Sciences.
Kawanowa, K., Sakuma, Y., Sakurai, S., et al. (2006). High incidence of microscopic gastrointestinal stromal tumors in the stomach. Human Pathology.
Agaimy, A., Wünsch, P. H., Hofstaedter, F., et al. (2007). Minute gastric sclerosing stromal tumors (GIST tumorlets) are common in adults and frequently show c-KIT mutations. The American Journal of Surgical Pathology.
Mussi, C., Schildhaus, H. U., Gronchi, A., Wardelmann, E., & Hohenberger, P. (2008). Therapeutic consequences from molecular biology for gastrointestinal stromal tumor patients affected by neurofibromatosis type 1. Clinical Cancer Research.
Comandone, A., Gasperoni, S., Manetti, R., & Tonelli, P. (2013). Gastrointestinal stromal tumors: Surgical and medical therapy. In What’s New in Surgical Oncology.
Agaram, N. P., Laquaglia, M. P., Ustun, B., et al. (2008). Molecular characterization of pediatric gastrointestinal stromal tumors. Clinical Cancer Research.
Pappo, A. S., & Janeway, K. A. (2009). Pediatric gastrointestinal stromal tumors. Hematology/Oncology Clinics of North America.
Cenaj, O., Jo, V. Y., & Doyle, L. A. (2017). Surgical pathology of gastrointestinal stromal tumors: Correlation with clinical and molecular subtypes. In Gastrointestinal stromal tumors.
Nishida, T., Goto, O., Raut, C. P., & Yahagi, N. (2016). Diagnostic and treatment strategy for small gastrointestinal stromal tumors. Cancer.
Fletcher, C. D. M., Berman, J. J., Corless, C., et al. (2002). Diagnosis of gastrointestinal stromal tumors: A consensus approach. Human Pathology.
Foo, W. C., Liegl-Atzwanger, B., & Lazar, A. J. (2012). Pathology of gastrointestinal stromal tumors. Clinical Medicine Insights Pathology.
Medeiros, F., Corless, C. L., Duensing, A., et al. (2004). KIT-negative gastrointestinal stromal tumors: Proof of concept and therapeutic implications. The American Journal of Surgical Pathology.
Novelli, M., Rossi, S., Rodriguez-Justo, M., et al. (2010). DOG1 and CD117 are the antibodies of choice in the diagnosis of gastrointestinal stromal tumours. Histopathology.
Miettinen, M., Wang, Z. F., & Lasota, J. (2009). DOG1 antibody in the differential diagnosis of gastrointestinal stromal tumors: A study of 1840 cases. The American Journal of Surgical Pathology.
West, R. B., Corless, C. L., Chen, X., et al. (2004). The novel marker, DOG1, is expressed ubiquitously in gastrointestinal stromal tumors irrespective of KIT or PDGFRA mutation status. The American Journal of Pathology.
Kang, G. H., Srivastava, A., Kim, Y. E., et al. (2011). DOG1 and PKC-are useful in the diagnosis of KIT-negative gastrointestinal stromal tumors. Modern Pathology.
Miettinen, M., & Lasota, J. (2006). Gastrointestinal stromal tumors: Review on morphology, molecular pathology, prognosis, and differential diagnosis. Archives of Pathology & Laboratory Medicine.
Anderson, W., O’Sullivan, B., Hughes, F., et al. (2017). Microscopic gastrointestinal stromal tumours: a clinical and molecular study of 13 cases. Histopathology.
Hirota, S., Isozaki, K., Moriyama, Y., et al. (1998). Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors. Science (80- ).
Wozniak, A., Rutkowski, P., Piskorz, A., et al. (2012). Prognostic value of KIT/PDGFRA mutations in gastrointestinal stromal tumours (GIST): Polish Clinical GIST Registry experience. Annals of Oncology.
Debiec-Rychter, M., Sciot, R., Le Cesne, A., et al. (2006). KIT mutations and dose selection for imatinib in patients with advanced gastrointestinal stromal tumours. European Journal of Cancer.
Rossi, F., Ehlers, I., Agosti, V., et al. (2006). Oncogenic Kit signaling and therapeutic intervention in a mouse model of gastrointestinal stromal tumor. Proceedings of the National Academy of Sciences of the United States of America.
Chi, P., Chen, Y., Zhang, L., et al. (2010). ETV1 is a lineage survival factor that cooperates with KIT in gastrointestinal stromal tumours. Nature.
Ran, L., Sirota, I., Cao, Z., et al. (2015). Combined inhibition of MAP kinase and KIT signaling synergistically destabilizes ETV1 and suppresses GIST tumor growth. Cancer Discovery.
Wardelmann, E., Losen, I., Hans, V., et al. (2003). Deletion of Trp-557 and Lys-558 in the juxtamembrane domain of the c-kit protooncogene is associated with metastatic behavior of gastrointestinal stromal tumors. International Journal of Cancer.
Heinrich, M. C., Corless, C. L., Duensing, A., et al. (2003). PDGFRA activating mutations in gastrointestinal stromal tumors. Science (80- ).
Wozniak, A., Rutkowski, P., Schöffski, P., et al. (2014). Tumor genotype is an independent prognostic factor in primary gastrointestinal stromal tumors of gastric origin: A European multicenter analysis based on ConticaGIST. Clinical Cancer Research.
Corless, C. L., & Heinrich, M. C. (2008). Molecular pathobiology of gastrointestinal stromal sarcomas. Annual Review of Pathology: Mechanisms of Disease.
Niinuma, T., Suzuki, H., & Sugai, T. (2018). Molecular characterization and pathogenesis of gastrointestinal stromal tumor. Translation of Gastroenterology and Hepatology.
Lasota, J., Dansonka-Mieszkowska, A., Sobin, L. H., & Miettinen, M. (2004). A great majority of GISTs with PDGFRA mutations represent gastric tumors of low or no malignant potential. Laboratory Investigation.
Sakurai, S., Hasegawa, T., Sakuma, Y., et al. (2004). Myxoid epithelioid gastrointestinal stromal tumor (GIST) with mast cell infiltrations: A subtype of GIST with mutations of platelet-derived growth factor receptor alpha gene. Human Pathology.
Corless, C. L., Fletcher, J. A., & Heinrich, M. C. (2004). Biology of gastrointestinal stromal tumors. Journal of Clinical Oncology.
Gill, A. J., Chou, A., Vilain, R., et al. (2010). Immunohistochemistry for SDHB divides gastrointestinal stromal tumors (GISTs) into 2 distinct types. The American Journal of Surgical Pathology.
Daniels, M., Lurkin, I., Pauli, R., et al. (2011). Spectrum of KIT/PDGFRA/BRAF mutations and Phosphatidylinositol-3-Kinase pathway gene alterations in gastrointestinal stromal tumors (GIST). Cancer Letters.
Pantaleo, M. A., Nannini, M., Corless, C. L., & Heinrich, M. C. (2015). Quadruple wild-type (WT) GIST: Defining the subset of GIST that lacks abnormalities of KIT, PDGFRA, SDH, or RAS signaling pathways. Cancer Medicine.
Janeway, K. A., Liegl, B., Harlow, A., et al. (2007). Pediatric KIT-wild-type and platelet-derived growth factor receptor α-wild-type gastrointestinal stromal tumors share KIT activation but not mechanisms of genetic progression with adult gastrointestinal stromal tumors. Cancer Research.
Kinoshita, K., Hirota, S., Isozaki, K., et al. (2004). Absence of c-kit gene mutations in gastrointestinal stromal tumours from neurofibromatosis type I patients. The Journal of Pathology.
Stratakis, C. A., & Carney, J. A. (2009). The triad of paragangliomas, gastric stromal tumours and pulmonary chondromas (Carney triad), and the dyad of paragangliomas and gastric stromal sarcomas (Carney-Stratakis syndrome): Molecular genetics and clinical implications. Journal of Internal Medicine.
Belinsky, M. G., Rink, L., & von Mehren, M. (2013). Succinate dehydrogenase deficiency in pediatric and adult gastrointestinal stromal tumors. Frontiers in Oncology.
Roberts, P. J., & Eisenberg, B. (2002). Clinical presentation of gastrointestinal stromal tumors and treatment of operable disease. European Journal of Cancer.
Miettinen, M., Sobin, L. H., & Lasota, J. (2005). Gastrointestinal stromal tumors of the stomach: A clinicopathologic, immunohistochemical, and molecular genetic study of 1765 cases with long-term follow-up. The American Journal of Surgical Pathology.
Miettinen, M., Makhlouf, H., Sobin, L. H., & Lasota, J. (2006). Gastrointestinal stromal tumors of the jejunum and ileum: A clinicopathologic, immunohistochemical, and molecular genetic study of 906 cases before imatinib with long-term follow-up. The American Journal of Surgical Pathology.
Maynard, M. A., Marino-Enriquez, A., Fletcher, J. A., et al. (2014). Thyroid hormone inactivation in gastrointestinal stromal tumors. The New England Journal of Medicine.
Scarpa, M., Bertin, M., Ruffolo, C., Polese, L., D’Amico, D. F., & Angriman, I. (2008). A systematic review on the clinical diagnosis of gastrointestinal stromal tumors. Journal of Surgical Oncology.
Hwang, J. H., Rulyak, S. D., & Kimmey, M. B. (2006). American Gastroenterological Association Institute technical review on the management of gastric subepithelial masses. Gastroenterology.
Watson, R. R., Binmoeller, K. F., Hamerski, C. M., et al. (2011). Yield and performance characteristics of endoscopic ultrasound-guided fine needle aspiration for diagnosing upper GI tract stromal tumors. Digestive Diseases and Sciences.
Mehren, M. V., Randall, R. L., Benjamin, R. S., et al. (2018). Soft tissue sarcoma, version 2.2018: Clinical practice guidelines in oncology. Journal National Comprehensive Cancer Network.
Tio, T. L., Tytgat, G. N. J., & den Hartog Jager, F. C. A. (1990). Endoscopic ultrasonography for the evaluation of smooth muscle tumors in the upper gastrointestinal tract: An experience with 42 cases. Gastrointestinal Endoscopy.
Demetri, G. D., Benjamin, R. S., Blanke, C. D., et al. (2007). NCCN task force report: Management of patients with Gastrointestinal Stromal Tumor (GIST)-Update of the NCCN clinical practice guidelines. Journal National Comprehensive Cancer Network.
Gayed, I., Vu, T., Iyer, R., et al. (2004). The role of 18F-FDG PET in staging and early prediction of response to therapy of recurrent gastrointestinal stromal tumors. Journal of Nuclear Medicine.
Kamiyama, Y., Aihara, R., Nakabayashi, T., et al. (2005). 18F-fluorodeoxyglucose positron emission tomography: Useful technique for predicting malignant potential of gastrointestinal stromal tumors. World Journal of Surgery.
Nishida, T. (2017). Therapeutic strategies for wild-type gastrointestinal stromal tumor: Is it different from KIT or PDGFRA-mutated GISTs? Translation of Gastroenterology and Hepatology.
Miettinen, M., Wang, Z. F., Sarlomo-Rikala, M., Osuch, C., Rutkowski, P., & Lasota, J. (2011). Succinate dehydrogenase-deficient GISTs: A clinicopathologic, immunohistochemical, and molecular genetic study of 66 gastric GISTs with predilection to young age. The American Journal of Surgical Pathology.
Haller, F., Moskalev, E. A., Faucz, F. R., et al. (2014). Aberrant DNA hypermethylation of SDHC: A novel mechanism of tumor development in Carney triad. Endocrine-Related Cancer.
Gaal, J., Stratakis, C. A., Carney, J. A., et al. (2011). SDHB immunohistochemistry: A useful tool in the diagnosis of Carney-Stratakis and Carney triad gastrointestinal stromal tumors. Modern Pathology.
Jasperson, K. W., Kohlmann, W., Gammon, A., et al. (2014). Role of rapid sequence whole-body MRI screening in SDH-associated hereditary paraganglioma families. Familial Cancer.
Agaram, N. P., Wong, G. C., Guo, T., et al. (2008). Novel V600E BRAF mutations in imatinib-naive and imatinib-resistant gastrointestinal stromal tumors. Genes, Chromosomes and Cancer.
Miettinen, M., & Lasota, J. (2006). Gastrointestinal stromal tumors: Pathology and prognosis at different sites. Seminars in Diagnostic Pathology.
Rubin, B. P., Blanke, C. D., Demetri, G. D., et al. (2010). Protocol for the examination of specimens from patients with Gastrointestinal Stromal Tumor. Archives of Pathology & Laboratory Medicine.
Huang, H. Y., Li, C. F., Huang, W. W., et al. (2007). A modification of NIH consensus criteria to better distinguish the highly lethal subset of primary localized gastrointestinal stromal tumors: A subdivision of the original high-risk group on the basis of outcome. Surgery.
Crosby, J. A., Catton, C. N., Davis, A., et al. (2001). Malignant gastrointestinal stromal tumors of the small intestine: A review of 50 cases from a prospective database. Annals of Surgical Oncology.
Besana-Ciani, I., Boni, L., Dionigi, G., Benevento, A., & Dionigi, R. (2003). Outcome and long term results of surgical resection for gastrointestinal stromal tumors (GIST). Scandinavian Journal of Surgery.
Langer, C., Gunawan, B., Schüler, P., Huber, W., Füzesi, L., & Becker, H. (2003). Prognostic factors influencing surgical management and outcome of gastrointestinal stromal tumours. The British Journal of Surgery.
Carboni, F., Carlini, M., Scardamaglia, F., et al. (2003). Gastrointestinal stromal tumors of the stomach. A ten-year surgical experience. Journal of Experimental & Clinical Cancer Research.
Emory, T. S., Sobin, L. H., Lukes, L., Lee, D. H., & O’Leary, T. J. (1999). Prognosis of gastrointestinal smooth-muscle (stromal) tumors: Dependence on anatomic site. The American Journal of Surgical Pathology.
Joensuu, H., Vehtari, A., Riihimäki, J., et al. (2012). Risk of recurrence of gastrointestinal stromal tumour after surgery: An analysis of pooled population-based cohorts. The Lancet Oncology.
Kukar, M., Kapil, A., Papenfuss, W., Groman, A., Grobmyer, S. R., & Hochwald, S. N. (2015). Gastrointestinal stromal tumors (GISTs) at uncommon locations: A large population based analysis. Journal of Surgical Oncology.
Hølmebakk, T., Bjerkehagen, B., Boye, K., Bruland, Stoldt, S., & Sundby Hall, K. (2016). Definition and clinical significance of tumour rupture in gastrointestinal stromal tumours of the small intestine. The British Journal of Surgery.
Joensuu, H. (2008). Risk stratification of patients diagnosed with gastrointestinal stromal tumor. Human Pathology.
Kim, T. W., Lee, H., Kang, Y. K., et al. (2004). Prognostic significance of c-kit mutation in localized gastrointestinal stromal tumors. Clinical Cancer Research.
Taniguchi, M., Nishida, T., Hirota, S., et al. (1999). Effect of c-kit mutation on prognosis of gastrointestinal stromal tumors. Cancer Research.
Antonescu, C. R., Sommer, G., Sarran, L., et al. (2003). Association of KIT exon 9 mutations with nongastric primary site and aggressive behavior: KIT mutation analysis and clinical correlates of 120 gastrointestinal stromal tumors. Clinical Cancer Research.
Emile, J. F., Théou, N., Tabone, S., et al. (2004). Clinicopathologic, phenotypic, and genotypic characteristics of gastrointestinal mesenchymal tumors. Clinical Gastroenterology and Hepatology.
Martín, J., Poveda, A., Llombart-Bosch, A., et al. (2005). Deletions affecting codons 557-558 of the c-KIT gene indicate a poor prognosis in patients with completely resected gastrointestinal stromal tumors: A study by the Spanish Group for Sarcoma Research (GEIS). Journal of Clinical Oncology.
Heinrich, M. C., Corless, C. L., Demetri, G. D., et al. (2003). Kinase mutations and imatinib response in patients with metastatic gastrointestinal stromal tumor. Journal of Clinical Oncology.
Sepe, P. S., & Brugge, W. R. (2009). A guide for the diagnosis and management of gastrointestinal stromal cell tumors. Nature Reviews. Gastroenterology & Hepatology.
Hwang, J. H., & Kimmey, M. B. (2004). The incidental upper gastrointestinal subepithelial mass. Gastroenterology.
DeMatteo, R. P., Lewis, J. J., Leung, D., Mudan, S. S., Woodruff, J. M., & Brennan, M. F. (2000). Two hundred gastrointestinal stromal tumors: Recurrence patterns and prognostic factors for survival. Annals of Surgery.
Boikos, S. A., Pappo, A. S., Killian, J. K., et al. (2016). Molecular subtypes of KIT/PDGFRA wild-type gastrointestinal stromal tumors a report from the national institutes of health gastrointestinal stromal tumor clinic. JAMA Oncology.
Catena, F., Di Battista, M., Ansaloni, L., et al. (2012). Microscopic margins of resection influence primary gastrointestinal stromal tumor survival. Onkologie.
Edmonson, J. H., Marks, R. S., Buckner, J. C., & Mahoney, M. R. (2002). Contrast of response to dacarbazine, mitomycin, doxorubicin, and cisplatin (DMAP) plus GM-CSF between patients with advanced malignant gastrointestinal stromal tumors and patients with other advanced leiomyosarcomas. Cancer Investigation.
Plaat, B. E. C., Hollema, H. H., Molenaar, W. M., et al. (2000). Soft tissue leiomyosarcomas and malignant gastrointestinal stromal tumors: Differences in clinical outcome and expression of multidrug resistance proteins. Journal of Clinical Oncology.
Eisenberg, B. L., Harris, J., Blanke, C. D., et al. (2009). Phase II trial of neoadjuvant/adjuvant imatinib mesylate (IM) for advanced primary and metastatic/recurrent operable gastrointestinal stromal tumor (GIST): Early results of RTOG 0132/ACRIN 6665. Journal of Surgical Oncology.
Fiore, M., Palassini, E., Fumagalli, E., et al. (2009). Preoperative imatinib mesylate for unresectable or locally advanced primary gastrointestinal stromal tumors (GIST). European Journal of Surgical Oncology.
Hohenberger, P., Langer, C., Wendtner, C. M., et al. (2012). Neoadjuvant treatment of locally advanced GIST: Results of APOLLON, a prospective, open label phase II study in KIT- or PDGFRA-positive tumors. JCO.
Kurokawa, Y., Yang, H. K., Cho, H., et al. (2017). Phase II study of neoadjuvant imatinib in large gastrointestinal stromal tumours of the stomach. British Journal of Cancer.
McAuliffe, J. C., Hunt, K. K., Lazar, A. J. F., et al. (2009). A randomized, phase II study of preoperative plus postoperative imatinib in GIST: Evidence of rapid radiographic response and temporal induction of tumor cell apoptosis. Annals of Surgical Oncology.
Blesius, A., Cassier, P. A., Bertucci, F., et al. (2011). Neoadjuvant imatinib in patients with locally advanced non metastatic GIST in the prospective BFR14 trial. BMC Cancer.
Eisenberg, B. L., & Judson, I. (2004). Surgery and imatinib in the management of GIST: Emerging approaches to adjuvant and neoadjuvant therapy. Annals of Surgical Oncology.
Gold, J. S., & DeMatteo, R. P. (2006). Combined surgical and molecular therapy: The gastrointestinal stromal tumor model. Annals of Surgery.
DeMatteo, R. P., Ballman, K. V., Antonescu, C. R., et al. (2013). Long-term results of adjuvant imatinib mesylate in localized, high-risk, primary gastrointestinal stromal tumor: ACOSOG Z9000 (Alliance) intergroup phase 2 trial. Annals of Surgery.
Corless, C. L., Ballman, K. V., Antonescu, C. R., et al. (2014). Pathologic and molecular features correlate with long-term outcome after adjuvant therapy of resected primary GI stromal tumor: The ACOSOG Z9001 trial. Journal of Clinical Oncology.
Casali, P. G., Le Cesne, A., Velasco, A. P., et al. (2015). Time to definitive failure to the first tyrosine kinase inhibitor in localized GI stromal tumors treated with imatinib as an adjuvant: A European Organisation for Research and Treatment of Cancer Soft Tissue and Bone Sarcoma Group Intergroup Randomized Tr. Journal of Clinical Oncology.
Joensuu, H., Eriksson, M., Hall, K. S., et al. (2012). One vs three years of adjuvant imatinib for operable gastrointestinal stromal tumor: A randomized trial. JAMA - Journal of American Medicine Association.
Joensuu, H., Eriksson, M., Sundby Hall, K., et al. (2016). Adjuvant imatinib for high-risk GI stromal tumor: Analysis of a randomized trial. Journal of Clinical Oncology.
Joensuu, H., Wardelmann, E., Sihto, H., et al. (2017). Effect of KIT and PDGFRA mutations on survival in patients with gastrointestinal stromal tumors treated with adjuvant imatinib: An exploratory analysis of a randomized clinical trial. JAMA Oncology.
Raut, C. P., Espat, N. J., Maki, R. G., et al. (2017). Extended treatment with adjuvant imatinib (IM) for patients (pts) with high-risk primary gastrointestinal stromal tumor (GIST): The PERSIST-5 study. Journal of Clinical Oncology.
Demetri, G. D., Von Mehren, M., Blanke, C. D., et al. (2002). Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. The New England Journal of Medicine.
Van Oosterom, A. T., Judson, I., Verweij, J., et al. (2001). Safety and efficacy of imatinib (STI571) in metastatic gastrointestinal stromal tumours: A phase I study. Lancet.
Verweij, J., Van Oosterom, A., Blay, J. Y., et al. (2003). Imatinib mesylate (STI-571 Glivec®, GleevecTM) is an active agent for gastrointestinal stromal tumours, but does not yield responses in other soft-tissue sarcomas that are unselected for a molecular target: Results from an EORTC Soft Tissue and Bone Sarcom. European Journal of Cancer.
Verweij, J., Casali, P. G., Zalcberg, J., et al. (2004). Progression-free survival in gastrointestinal stromal tumours with high-dose imatinib: Randomised trial. Lancet.
Blanke, C. D., Rankin, C., Demetri, G. D., et al. (2008). Phase III randomized, intergroup trial assessing imatinib mesylate at two dose levels in patients with unresectable or metastatic gastrointestinal stromal tumors expressing the kit receptor tyrosine kinase: S0033. Journal of Clinical Oncology.
Heinrich, M. C., Owzar, K., Corless, C. L., et al. (2008). Correlation of kinase genotype and clinical outcome in the North American intergroup phase III trial of imatinib mesylate for treatment of advanced gastrointestinal stromal tumor: CALGB 150105 study by cancer and leukemia group B and southwest oncology gr. Journal of Clinical Oncology.
Van Glabbeke, M. (2010). Comparison of two doses of imatinib for the treatment of unresectable or metastatic gastrointestinal stromal tumors: A meta-analysis of 1, 640 patients. Journal of Clinical Oncology.
Heinrich, M. C., Maki, R. G., Corless, C. L., et al. (2008). Primary and secondary kinase genotypes correlate with the biological and clinical activity of sunitinib in imatinib-resistant gastrointestinal stromal tumor. Journal of Clinical Oncology.
Antonescu, C. R., Besmer, P., Guo, T., et al. (2005). Acquired resistance to imatinib in gastrointestinal stromal tumor occurs through secondary gene mutation. Clinical Cancer Research.
Desai, J., Shankar, S., Heinrich, M. C., et al. (2007). Clonal evolution of resistance to imatinib in patients with metastatic gastrointestinal stromal tumors. Clinical Cancer Research.
Wardelmann, E., Merkelbach-Bruse, S., Pauls, K., et al. (2006). Polyclonal evolution of multiple secondary KIT mutations in gastrointestinal stromal tumors under treatment with imatinib mesylate. Clinical Cancer Research.
George, S., Blay, J. Y., Casali, P. G., et al. (2009). Clinical evaluation of continuous daily dosing of sunitinib malate in patients with advanced gastrointestinal stromal tumour after imatinib failure. European Journal of Cancer.
Demetri, G. D., van Oosterom, A. T., Garrett, C. R., et al. (2006). Efficacy and safety of sunitinib in patients with advanced gastrointestinal stromal tumour after failure of imatinib: A randomised controlled trial. Lancet.
George, S., Wang, Q., Heinrich, M. C., et al. (2012). Efficacy and safety of regorafenib in patients with metastatic and/or unresectable GI stromal tumor after failure of imatinib and sunitinib: A multicenter phase II trial. Journal of Clinical Oncology.
Demetri, G. D., Reichardt, P., Kang, Y. K., et al. (2013). Effi cacy and safety of regorafenib for advanced gastrointestinal stromal tumours after failure of imatinib and sunitinib (GRID): An international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet.
Ben-Ami, E., Barysauskas, C. M., von Mehren, M., et al. (2016). Long-term follow-up results of the multicenter phase II trial of regorafenib in patients with metastatic and/or unresectable GI stromal tumor after failure of standard tyrosine kinase inhibitor therapy. Annals of Oncology.
Yeh, C. N., Chen, M. H., Chen, Y. Y., et al. (2017). A phase II trial of regorafenib in patients with metastatic and/ or a unresectable gastrointestinal stromal tumor harboring secondary mutations of exon 17. Oncotarget.
Heinrich, M. C., Jones, R. L., von Mehren, M., et al. (2020). Avapritinib in advanced PDGFRA D842V-mutant gastrointestinal stromal tumour (NAVIGATOR): A multicentre, open-label, phase 1 trial. Lancet Oncol [Internet], 21(7), 935–946. https://doi.org/10.1016/S1470-2045(20)30269-2.
Smith, B. D., Kaufman, M. D., Lu, W. P., et al. (2019). Ripretinib (DCC-2618) is a switch control kinase inhibitor of a broad spectrum of oncogenic and drug-resistant KIT and PDGFRA variants. Cancer Cell.
George, S., Heinrich, M., Chi, P., et al. (2018). Initial results of phase I study of DCC-2618, a broad-spectrum KIT and PDGFRa inhibitor, in patients (pts) with gastrointestinal stromal tumor (GIST) by number of prior regimens. Annals Oncology Official: Journal Euro Society Medicine Oncology.
von Mehren, M., Serrano, C., Bauer, S., et al. (2019). INVICTUS: A phase III, interventional, double-blind, placebo-controlled study to assess the safety and efficacy of ripretinib as ≥ 4th-line therapy in patients with advanced gastrointestinal stromal tumors (GIST) who have received treatment with prior ant. Annals of Oncology.
Nemunaitis, J., Bauer, S., Blay, J. Y., et al. (2019). Intrigue: Phase III study of ripretinib versus sunitinib in advanced gastrointestinal stromal tumor after imatinib. Future Oncology.
Gebreyohannes, Y. K., Schöffski, P., Van Looy, T., et al. (2016). Cabozantinib is active against human gastrointestinal stromal tumor xenografts carrying different KIT mutations. Molecular Cancer Therapeutics.
Schoffski, P., Mir, O., Kasper, B., et al. (2019). Activity and safety of cabozantinib in patients with gastrointestinal stromal tumor after failure of imatinib and sunitinib: EORTC phase II trial 1317 CaboGIST. Journal of Clinical Oncology.
Manley, P. W., Stiefl, N., Cowan-Jacob, S. W., et al. (2010). Structural resemblances and comparisons of the relative pharmacological properties of imatinib and nilotinib. Bioorganic and Medicinal Chemistry.
Ozaslan, E., Ozkan, M., Bozkurt, O., et al. (2015). Are Rogerofenib and Nilotinib effective for advanced gastrointestinal stromal tumor (GIST) patients who have already been given main treatments? Asian Pacific Journal of Cancer Prevention.
Demetri, G. D., Casali, P. G., Blay, J. Y., et al. (2009). A phase I study of single-agent nilotinib or in combination with imatinib in patients with imatinib-resistant gastrointestinal stromal tumors. Clinical Cancer Research.
Cauchi, C., Somaiah, N., Engstrom, P. F., et al. (2012). Evaluation of nilotinib in advanced GIST previously treated with imatinib and sunitinib. Cancer Chemotherapy and Pharmacology.
Reichardt, P., Blay, J. Y., Gelderblom, H., et al. (2012). Phase III study of nilotinib versus best supportive care with or without a TKI in patients with gastrointestinal stromal tumors resistant to or intolerant of imatinib and sunitinib. Annals of Oncology.
Sawaki, A., Nishida, T., Doi, T., et al. (2011). Phase 2 study of nilotinib as third-line therapy for patients with gastrointestinal stromal tumor. Cancer.
Blay, J. Y., Shen, L., Kang, Y. K., et al. (2015). Nilotinib versus imatinib as first-line therapy for patients with unresectable or metastatic gastrointestinal stromal tumours (ENESTg1): A randomised phase 3 trial. The Lancet Oncology.
del Muro, X. G. (2015). Nilotinib, imatinib, and GIST therapy. The Lancet Oncology.
Garner, A. P., Gozgit, J. M., Anjum, R., et al. (2014). Ponatinib inhibits polyclonal drug-resistant KIT oncoproteins and shows therapeutic potential in heavily pretreated gastrointestinal stromal tumor (GIST) patients. Clinical Cancer Research.
Heinrich, M. C., von Mehren, M., & Demetri, G. D. (2014). et al., A phase 2 study of ponatinib in patients (pts) with advanced gastrointestinal stromal tumors (GIST) after failure of tyrosine kinase inhibitor (TKI) therapy: Initial report. Journal of Clinical Oncology.
Mir, O., Cropet, C., Toulmonde, M., et al. (2016). Pazopanib plus best supportive care versus best supportive care alone in advanced gastrointestinal stromal tumours resistant to imatinib and sunitinib (PAZOGIST): A randomised, multicentre, open-label phase 2 trial. The Lancet Oncology.
Nishida, T., & Doi, T. (2016). Pazopanib for both GIST and soft-tissue sarcoma. The Lancet Oncology.
Ganjoo, K. N., Villalobos, V. M., Kamaya, A., et al. (2014). A multicenter phase II study of pazopanib in patients with advanced gastrointestinal stromal tumors (GIST) following failure of at least imatinib and sunitinib. Annals of Oncology.
Dubreuil, P., Letard, S., Ciufolini, M., et al. (2009). Masitinib (AB1010), a potent and selective tyrosine kinase inhibitor targeting KIT. PLoS One.
Soria, J. C., Massard, C., Magné, N., et al. (2009). Phase 1 dose-escalation study of oral tyrosine kinase inhibitor masitinib in advanced and/or metastatic solid cancers. European Journal of Cancer.
Le Cesne, A., Blay, J., Bui, N. B., et al. (2009). Masatinib mesylate in imatinib-naive locally advanced or metastatic gastrointestinal stromal tumor (GIST): Results of the French Sarcoma Group phase II trial. Journal of Clinical Oncology.
Blay, J.-Y., Heinrich, M. C., Hohenberger, P., et al. (2017). A randomized, double-blind, placebo-controlled, phase III study of crenolanib in advanced or metastatic GIST patients bearing a D842V mutation in PDGFRA: The CrenoGIST study. Journal of Clinical Oncology.
Heinrich, M. C., Griffith, D., McKinley, A., et al. (2012). Crenolanib inhibits the drug-resistant PDGFRA D842V mutation associated with imatinib-resistant gastrointestinal stromal tumors. Clinical Cancer Research.
Huynh, H., Lee, J. W. J., Chow, P. K. H., et al. (2009). Sorafenib induces growth suppression in mouse models of gastrointestinal stromal tumor. Molecular Cancer Therapeutics.
Wiebe, L., Kasza, K. E., Maki, R. G., et al. (2008). Activity of sorafenib (SOR) in patients (pts) with imatinib (IM) and sunitinib (SU)-resistant (RES) gastrointestinal stromal tumors (GIST): A phase II trial of the University of Chicago Phase II Consortium. Journal of Clinical Oncology.
Park, S. H., Ryu, M. H., Ryoo, B. Y., et al. (2012). Sorafenib in patients with metastatic gastrointestinal stromal tumors who failed two or more prior tyrosine kinase inhibitors: A phase II study of Korean gastrointestinal stromal tumors study group. Investigational New Drugs.
Campbell, N. P., Wroblewski, K., Maki, R. G., et al. (2011). Final results of a University of Chicago phase II consortium trial of sorafenib (SOR) in patients (pts) with imatinib (IM)- and sunitinib (SU)-resistant (RES) gastrointestinal stromal tumors (GIST). Journal of Clinical Oncology.
Brzozowska, M., Wierzba, W., Szafraniec-Buryło, S., et al. (2019). Real-world evidence of patient outcome following treatment of advanced gastrointestinal stromal tumor (GIST) with imatinib, sunitinib, and sorafenib in publicly funded health care in Poland. Medical Science Monitor.
Rutkowski, P., Jagielska, B., Andrzejuk, J., et al. (2017). The analysis of the long-term outcomes of sorafenib therapy in routine practice in imatinib and sunitinib resistant gastrointestinal stromal tumors (GIST). Wspolczesna Onkology.
Singeltary, B., Ghose, A., Sussman, J., Choe, K., & Olowokure, O. (2014). Durable response with a combination of imatinib and sorafenib in KIT exon 17 mutant gastrointestinal stromal tumor. Journal of Gastrointest Oncology.
Schöffski, P., Reichardt, P., Blay, J. Y., et al. (2010). A phase I-II study of everolimus (RAD001) in combination with imatinib in patients with imatinib-resistant gastrointestinal stromal tumors. Annals of Oncology.
Hohenberger, P., Bauer, S., Grunwald, V., et al. (2010). Multicenter, single-arm, two-stage phase II trial of everolimus (RAD001) with imatinib in imatinib-resistant patients (pts) with advanced GIST. Journal of Clinical Oncology.
Piovesan, C., Fumagalli, E., Coco, P., et al. (2009). Response to sirolimus in combination to tirosine kinase inhibitors (TKI) in three cases of PDGFRA-D842V metastatic gastrointestinal stromal tumor (GIST). Journal of Clinical Oncology.
Palassini, E., Fumagalli, E., Coco, P., et al. (2008). Combination of PKC412 and sirolimus in a metastatic patient with PDGFRA-D842V gastrointestinal stromal tumor (GIST). Journal of Clinical Oncology.
Tan, Y., Trent, J. C., Wilky, B. A., Kerr, D. A., & Rosenberg, A. E. (2017). Current status of immunotherapy for gastrointestinal stromal tumor. Cancer Gene Therapy.
Schroeder, B. A., Kohli, K., O’Malley, R. B., et al. Durable tumor regression in highly refractory metastatic KIT/PDGFRA wild-type GIST following treatment with nivolumab. Oncoimmunology [Internet] 2020;9(1). Available from https://doi.org/10.1080/2162402X.2019.1710064.
Rosenbaum, E., Kelly, C., D’Angelo, S. P., et al. (2019). A phase I study of binimetinib (MEK162) combined with pexidartinib (PLX3397) in patients with advanced gastrointestinal stromal tumor. Oncologist.
Chi, P., Qin, L.-X., D’Angelo, S. P., et al. (2015). A phase Ib/II study of MEK162 (binimetinib [BINI]) in combination with imatinib in patients with advanced gastrointestinal stromal tumor (GIST). Journal of Clinical Oncology.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Informed consent Specific informed consent is not required. Images and radiography are de-identified. Patient in the case included in the article has signed an informed consent preoperatively for possible use of images and pictures for research.
Conflict of interest
Asfar Azmi—funding from Janssen and Karyopharm Therapeutics Inc. Consultant for Guidepoint and GLG. Speaker bureau at Karyopharm Therapeutics Inc. All other authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Al-Share, B., Alloghbi, A., Al Hallak, M.N. et al. Gastrointestinal stromal tumor: a review of current and emerging therapies. Cancer Metastasis Rev 40, 625–641 (2021). https://doi.org/10.1007/s10555-021-09961-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10555-021-09961-7