Abstract
According to updated Lake-Louise Criteria, impaired regional myocardial function serves as a supportive criterion in diagnosing myocarditis. This study aimed to assess visual regional wall motional abnormalities (RWMA) and novel quantitative regional longitudinal peak strain (RLS) for risk stratification in the clinical setting of myocarditis. In patients undergoing CMR and meeting clinical criteria for suspected myocarditis global longitudinal strain (GLS), late gadolinium enhancement (LGE), RWMA and RLS were assessed in the anterior, septal, inferior, and lateral regions and correlated to the occurrence of major adverse cardiac events (MACE), including heart failure hospitalization, sustained ventricular tachycardia, recurrent myocarditis, and all-cause death. In 690 consecutive patients (age: 48.0 ± 16.0 years; 37.7% female) with suspected myocarditis impaired RLS was correlated with RWMA and LV-GLS but not with the presence of LGE. At median follow up of 3.8 years, MACE occurred in 116 (16.8%) patients. Both, RWMA and RLS in anterior-, septal-, inferior-, and lateral- locations were univariately associated with outcomes (all p < 0.001), but not after adjusting for clinical characteristics and LV-GLS. In the subgroup of patients with normal LV function, RWMA were not predictive of outcomes, whereas septal RLS had incremental and independent prognostic value over clinical characteristics (HRadjusted = 1.132, 95% CI 1.020–1.256; p = 0.020). RWMA and RLS can be used to assess regional impairment of myocardial function in myocarditis but are of limited prognostic value in the overall population. However, in the subgroup of patients with normal LV function, septal RLS represents a distinctive marker of regional LV dysfunction, offering potential for risk-stratification.
Graphical abstract
CI: confidence interval, CMR: cardiac magnetic resonance imaging, HR: hazard ratio, MACE major adverse cardiovascular events

Similar content being viewed by others
Introduction
Diagnosing myocarditis remains a challenge, due to its heterogeneous clinical presentation with a wide range of symptoms from asymptomatic cases to heart failure and sudden cardiac death [1, 2]. In the clinical setting of suspected myocarditis, cardiac magnetic resonance imaging (CMR) provides the ability to evaluate various functional and tissue characterization parameters in one comprehensive exam. The pivotal role of CMR to non-invasively diagnose myocarditis is underscored in the Lake Louise Criteria (LLC) [3]. In 2018 updated LLC encompass major criteria including non-ischemic myocardial injury evaluated by diffuse fibrosis and late gadolinium enhancement (LGE), and myocardial edema, while signs of pericarditis and global- as well as regional left ventricular (LV) dysfunction serve as supportive criteria [3]. For global LV function, the assessment of quantitative global longitudinal strain (GLS) by CMR feature tracking has proven to be of higher prognostic value over traditional functional assessments such as LV ejection fraction (EF) [4]. Currently, regional left ventricular (LV) dysfunction is mostly evaluated by qualitative visual assessment (i.e. visual regional wall motion abnormalities, RWMA). It is unclear whether RWMA or novel quantitative regional longitudinal peak strain (RLS) can help to risk stratify patients with suspected myocarditis. In this retrospective observational study, we sought to investigate the respective prognostic values of measurements of regional myocardial dysfunction from CMR (RWMA and RLS) for major adverse cardiovascular events (MACE) in suspected myocarditis. As myocarditis frequently affects localized regions, and patients present initially with normal global LV function, we also seek to determine if localized myocardial injury reflected by either RWMA or RLS is associated with adverse cardiac outcomes in patients with preserved global LV function.
Methods
Study design
Consecutive patients with clinical suspicion for myocarditis, who were referred for CMR, were included in two registries at tertiary centers (i.e. CMRMyo, CMR Features in Patients With Suspected Myocarditis registry, NCT03470571, and the FlamBer, Inflammatory Cardiomyopathy Bern Registry, NCT04774549). Patients were enrolled at the Brigham and Women’s Hospital, Harvard Medical School Boston, MA, USA between 2002 and 2015 and at Inselspital, University Hospital Bern, Switzerland between 2005 and 2019. Detailed inclusion and exclusion criteria for this cohort can be found elsewhere [4,5,6,7,8]. Patients who fulfilled clinical criteria for suspected myocarditis, as suggested by European Society of Cardiology (ESC) [1] were included in the final analysis. In short, ≥ 1 clinical criteria (e.g. acute chest pain, dyspnea) and ≥ 1 diagnostic criteria (e.g. abnormal ECG, elevated troponin) or presence of edema or LGE in a typical myocarditis pattern in CMR or ≥ 2 diagnostic criteria, were considered as clinically suspected myocarditis [1]. Follow up was performed at both centers by clinically indicated outpatient visits, standardized interviews, documentation from referring physicians and hospital discharge summaries. Endpoint was a composite of first MACE including all-cause death, hospitalization for congestive heart failure, documented sustained ventricular tachycardia for ≥ 30 s or recurrent myocarditis according to the ESC criteria [1]. The study was approved by the local ethics committees and was conducted in accordance with the Declaration of Helsinki.
Image acquisition and analysis
CMR imaging was either performed on a 1.5 or 3.0 Tesla scanner (Magnetom Trio and Area, Siemens Healthineers, Erlangen, Germany) as described previously [4, 7, 9]. The application cvi41 (Circle Cardiovascular Imaging, Calgary, Canada) was used for postprocessing. Endo- and epicardial contours were automatically generated in end diastole and manually checked for plausibility in a short axis stack and three long axis (2-chamber, 3-chamber and 4-chamber view) cine sequences. Tracking these contours over the full cardiac cycle allowed to derive biventricular volumes, EF and feature tracking based peak global and RLS based on 16-segment American Heart Association (AHA) nomenclature [10]. RLS of each segment was individually checked for adequate time to peak and plausibility (e.g. only tracking during a part of the cardiac cycle). If required, values were either corrected for the peak strain at correct time to peak or excluded. Since reliability of segment based RLS is limited [9], segments were also summarized to different regions by calculating the mean RLS of summarized segments. Regions were defined as anterior (AHA-segments 1, 7, and 13), septal (segments 2, 3, 8, 9, 14), inferior (segments 4, 10, 15) and lateral (segments 5, 6, 11, 12, 16). RWMA were visually evaluated in a short-axis stack, according to regions depicted above. Patients with diffuse hypokinesis (LVEF < 30%) were categorized as having RWMA in each region. The LGE extent was semi-quantitatively determined by the sum of all AHA segments exhibiting LGE. For the sub-analysis of patients with normal LV function, LV-GLS below − 14.2% for women and below − 12.7% for men based on the publication by Kawel-Boehm et al. [11] was considered normal.
Statistical analysis
Continuous variables were expressed as mean ± standard deviation or median and interquartile range based on normality. Categorical variables were presented as frequency and percent of the population. Patient characteristics and global findings are reported for the entire cohort, as well as for patients with and without normal global LV function, based on LV-GLS (see above). Groups were compared by a chi-square test or an independent t-test, for categorical or continuous variables, respectively. Pearson correlation was used to describe the association between RLS and GLS. Univariate and multivariate regression for the association with MACE was performed using Cox proportional hazards ratio (HR) and reported with 95% confidence intervals (CI). A multivariate Cox regression model for MACE was applied, including manually selected variables, which were not included in the diagnostic work up based on ESC criteria for suspected myocarditis [1]. Variables with > 10% missing values (e.g. results of parametric mapping) or those that could introduce multicollinearity (e.g. LV volumes in addition to LVEF) were not considered. The goodness of fit for each model was evaluated by Chi-Square from a likelihood ratio test and compared to the subsequent model for statistically significant differences. Kaplan Meier survival curves were plotted for data dichotomized by median septal RLS (< − 14.6% vs. ≥ 14.6%). Images of 25 randomly selected patients were replicated by the same reader and a second independent reader and inter‑ and intra-observer reliability of RWMA and RLS was assessed using a two-way intra-class correlation (ICC) test for absolute measures. ICC was interpreted according to Koo et al. (ICC ≥ 0.9–excellent, ICC = 0.75–0.89 – good, ICC = 0.5–0.74 – moderate, ICC < 0.5 – poor) [12]. Statistical significance was defined with a 2-sided p-value of < 0.05. Statistical analysis was performed with R software version 4.1.2. (R Foundation for Statistical Computing, Vienna Austria).
Results
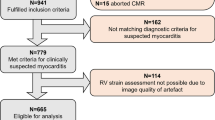
Out of the 1,125 consecutive patients initially referred to CMR for suspected myocarditis, 75 (6.7%) were excluded due to evidence of coronary artery disease, 50 (4.4%) refused study participation, 44 (3.9%) suffered from other cardiomyopathies (e.g. cardiac amyloidosis, stress cardiomyopathy, hypertrophic cardiomyopathy, severe valvular heart disease, LV non-compaction) and 15 (1.3%) did not complete the CMR exam. Among the remaining 941 patients, in 82 (8.7%) cases regional strain analysis was not possible due to arrhythmia, missing or foreshortened 2-, 3-, or 4-chamber view or artefact. Another 161 (16.0%) patients did not meet ESC criteria for clinically suspected myocarditis (1) and 8 (1.1%) patients were lost to follow-up, leaving 690 patients for the current analysis (Fig. 1). Mean age was 48.0 ± 16.0 years and 260 (37.7%) were women (Table 1). Most common symptoms at admission were chest pain and dyspnea which were present in 259 (37.5%) and 300 (44.7%) patients, respectively. Common ECG alterations comprised T-wave inversions in 193 patients (30.4%), left bundle branch block in 55 (8.6%) and ST-segment elevation in 37 (5.4%) patients. Mean LVEF was 49.1 ± 15.0% and mean LV-GLS was -13.1 ± 4.4%. Global LV function, as defined by sex-specific cutoffs for LV-GLS mentioned above, was impaired in 303 (43.9%) patients (Table 2). LGE was present in 442 (64.1%) patients and edema in T2 weighted imaging or T2 mapping was observed in 211 (37.9%) of 557 patients without missing T2 based image sequences (Fig. 2). Regional analysis demonstrated high prevalence of LGE in the inferior and lateral region (i.e., AHA-segments 4, 5, 10 and 11) (Fig. 3). RWMA by visual assessment was most common (25%) in inferior and lateral regions and less common in the anterior segments. RLS across regions was associated with the presence of RWMA (p < 0.001) but not with the presence of LGE (p = 0.456) within that same region (Fig. 4). Mean RLS for each region with and without LGE or RWMA is displayed in Supplemental Fig. 1. RLS of all regions was correlated to LV-GLS with correlation coefficients ranging from 0.758 to 0.836 (Supplemental Fig. 2). Reproducibility of RLS was good to moderate and ICC for intra-reader reliability was between 0.84 and 0.93 and ranged from 0.70 to 0.89 for inter-reader reliability (Supplemental Table 1). Intra- and inter-reader agreement for assessment of RWMA was good and good to moderate, respectively (Supplemental Table 2).
At median follow up of 3.8 years, MACE occurred in 116 (16.8%) patients, including heart failure hospitalization (40; 5.8%), sustained ventricular tachycardia (29; 4.2%), recurrent myocarditis (18; 2.6%), and all-cause death (29; 4.2%). MACE stratified by LVEF is presented in Supplemental Table 3. In addition to parameters of global LV function such as LVEF, LV-GLS and the presence of visually assessed RWMA, LGE and clinical characteristics including body-mass index (BMI), smoking, and a history of diabetes were associated with MACE in the univariate analysis. Anterior, septal, inferior, and lateral RLS and RWMA were also univariately associated with outcomes, while septal LGE was the only regional LGE pattern associated with MACE (Table 3). A basic multivariable predictive model was defined based on the results of univariate cox-regression and included BMI, smoking, history of diabetes mellitus, LVEF, LV-GLS, and LGE extent (Model χ2 = 65.86). None of the regional findings (RLS, RWMA and LGE) sequentially added to this model improved prognostication (Table 4) for the overall cohort. Separate results after removing LV GLS and LVEF from the multivariable models are provided in Supplemental Table 4.
For sub analyses, patients were stratified by LV-GLS into those with normal and impaired global LV function. Those with normal LV function were younger (p = 0.001), more often male (p < 0.001), less commonly suffered from diabetes mellitus (p = 0.006) and had more often chest pain symptoms (p < 0.001) but less dyspnea (p < 0.001) at admission (Table 1). LV function by LVEF, RLS and presence of RWMA correlated strongly to LV-GLS (Table 2). In patients with impaired LV function, several imaging parameters, including LVEF, RLS and RWMA of all regions, and LGE extent were associated with MACE, while in patients with normal LV function only clinical characteristics (BMI and smoking) and septal RLS were associated with outcomes (Table 3). Basic models incorporated LVEF and LGE extent in patients with impaired LV function, while BMI and smoking were included in the basic model for patients with normal LV function. The addition of regional findings demonstrated incremental and independent prognostic value of septal RLS over clinical characteristics in patients with normal LV function (HRadjusted = 1.132, 95% CI 1.020–1.256; p = 0.002), while no independent effect was observed in those with impaired LV function (Fig. 5). Visually assessed RWMA were not associated with outcomes in this subgroup of patients (Graphical Abstract).
Kaplan Meier curves stratified by median septal RLS (-14.6%) and the presence of visually assessed RWMA for the composite endpoint of MACE. *Hazard ratios and log-rank tests for the overall cohort were adjusted for body-mass-index, history of smoking and diabetes, LVEF, LV GLS and LGE extent. Hazard ratios and log rank tests for the subgroup of patients with impaired LV function were adjusted for LVEF and LGE extent. Hazard ratios and log rank tests for the subgroup of patients with normal LV function were adjusted for body-mass index and history of smoking. LGE late gadolinium enhancement, MACE major adverse cardiac events, RLS regional longitudinal peak strain, RWMA wall motion abnormalities
Discussion
This study for the first time investigated the prognostic value of visual and quantitative regional myocardial dysfunction in suspected myocarditis. Both the presence of RWMA and RLS had moderate to high measurement reproducibility and were associated with clinical outcomes. However, in the model adjusted to key clinical risk markers, regional LV dysfunction, indicated by either RLS or RWMA, did not independently predict clinical outcomes, thus is of limited predictive value in the overall cohort of patients with suspected myocarditis. In the subgroup of patients with preserved LV function septal RLS demonstrated incremental and independent association with MACE. Impaired septal RLS might represent a high-risk pattern and incorporation into risk stratification may refine the prediction of MACE in this subgroup.
RLS has been demonstrated to be useful in the detection of RWMA in patients with ischemic heart disease [13,14,15], to be correlated with LGE [14,15,16], and inherits predictive value in patients following myocardial infarction and chronic total occlusion [17, 18]. Our hypothesis was that also in suspected myocarditis impaired RLS and RWMA might indicate areas of regional inflammation or scarring. However, contrasting the findings made in ischemic heart disease, RLS and RWMA were neither associated with regional LGE nor had independent prognostic value over global LV function. Following myocardial infarction, myocardial scarring is more often transmural and limited to a specific area of coronary supply, while in myocarditis less dense subendocardial or patchy LGE pattern are common [7, 19], that are notably less strong associated to impaired regional function. Consequently, RLS and RWMA were strongly associated to LV-GLS in our study, which might explain the lack of incremental value over LV-GLS. Even if of limited prognostic value, RLS is a reproducible tool and correlated well to visually assessed RWMA, suggesting a diagnostic role as supportive criterion outlined in the updated LLC in patients with suspected myocarditis [3].
Consistent with our results, recent studies have shown the prognostic potential of several CMR parameters including LGE, LVEF and GLS in suspected myocarditis [4,5,6,7, 20,21,22,23]. However, in patients with normal global LV function, these parameters are of limited value and do not incrementally and independently predict outcomes. Although patients with normal LV function are at significantly lower risk for adverse events, a certain proportion of these patients still develops heart failure, ventricular tachycardia, or recurrent myocarditis at follow-up, underlying the need for better risk stratification in this subgroup. Impairment of septal RLS was associated with outcomes in this patient population, which is in line with previous studies, that identified septal involvement by LGE as a high-risk feature in myocarditis [7, 19] or ischemic heart disease [24]. Septal involvement might indicate extending of inflammation to the right ventricle or can go along with involvement of the conduction system, both of which are associated with poor outcomes [8, 25,26,27]. In fact, whereas septal LGE was a strong outcome predictor in our sub-cohort with impaired LV function, septal LGE was not associated with outcomes in the subcohort with preserved LV function. Therefore, and in addition to widely established tissue characterization by LGE, septal RLS might represent a marker for septal involvement and may help to refine risk stratification especially in patients with preserved global LV function.
So far, RLS has not yet been implemented in clinical practice and is rather seen as a research tool. The application and interpretation of RLS is challenging and several limitations of the technique should be considered. First, RLS shows limited inter-reader reproducibility when assessed on basis of AHA-segmentation [28, 29], emphasizing the need to cluster segments to regions, as done by this study and to standardize the post-processing. However, a definition of which AHA-segments should be included to a region is rather arbitrary, especially in diseases like myocarditis without a coronary-anatomy-based distribution of lesions. Additionally, through a tethering of strain, regions with normal myocardium neighboring impaired segments can also exhibit low RLS [14, 30, 31]. Finally, no reference values exist for different RLS pattern, which is further complicated by inter-vendor variabilities and limited comparability to echocardiography. Addressing these aspects would allow for a more widespread use of RLS not only in suspected myocarditis, but also in other cardiac disease where regional differences in myocardial function are expected.
Limitations
Several limitations require attention. First, this was a retrospective study, and the heterogeneous nature of myocarditis presentations makes the patient selection prone to selection bias. We tried to mitigate this concern by using standardized inclusion criteria for clinically suspected myocarditis according to the published ESC recommendations and by excluding all other cardiomyopathies and ischemic heart disease. Second, parametric mapping sequences (T1, T2 and extracellular volume fraction) were only available in more recently scanned patients and were therefore not included to outcome analysis or correlated to RLS. Third, since no reference values are available for regional peak strain assessed by CMR, cut-offs for Kaplan Meier curves were selected based on median values in our cohort. Fifths, follow-up imaging, such as repeat CMR to detect dilative cardiomyopathy as a consequence of acute myocarditis was not available for the majority of study participants. Sixths, although based on our experience we believe that the learning curve for the assessment of regional strain peaks early if a standardized protocol is followed, we did not analyze reproducibility over time. Finally, we did not confirm the diagnosis of myocarditis by a diagnostic gold-standard such as endomyocardial biopsy, which reflect current practice. Further, endomyocardial biopsy is prone to sampling error, associated with a non-negligible risk, and is currently reserved for more severe cases.
Conclusion
RLS is a feasible tool to assess regional impairment of myocardial function and correlates to visually assessed RWMA. Both are associated with outcomes but are of lower prognostic value when compared to LV GLS. Nevertheless, in the subgroup of patients with normal LV function, septal RLS represents a marker of regional LV dysfunction with higher prognostic value than RWMA and can be used to refine risk-stratification in suspected myocarditis.
Data availability
Data available upon reasonable request from the corresponding author.
Abbreviations
- AHA:
-
American Heart Association
- CAD:
-
Coronary artery disease
- CI:
-
Confidence Interval
- CMR:
-
Cardiovascular Magnetic Resonance Imaging
- ECV:
-
Extracellular volume
- EMB:
-
Endomyocardial biopsy
- ESC:
-
European society of cardiology
- FWHM:
-
Full width half maximum
- GLS:
-
Global longitudinal peak strain
- HR:
-
Hazard ratio
- ICC:
-
Intra-class correlation coefficient
- LGE:
-
Late gadolinium enhancement
- LLC:
-
Lake Louise criteria
- LVEF:
-
Left ventricular ejection fraction
- MACE:
-
Major adverse cardiac events
- RLS:
-
Regional longitudinal peak strain
- RWMA:
-
Visual regional wall motional abnormalities
References
Caforio AL, Pankuweit S, Arbustini E, Basso C, Gimeno-Blanes J, Felix SB, Fu M, Helio T, Heymans S, Jahns R, Klingel K, Linhart A, Maisch B, McKenna W, Mogensen J, Pinto YM, Ristic A, Schultheiss HP, Seggewiss H, Tavazzi L, Thiene G, Yilmaz A, Charron P, Elliott PM, European Society of Cardiology Working Group on Myocardial and Pericardial Diseases (2013) Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: a position statement of the European society of cardiology working group on myocardial and pericardial diseases. Eur Heart J 34(2636–48):2648a–2648d. https://doi.org/10.1093/eurheartj/eht210
Cooper LT Jr (2009) Myocarditis. N Engl J Med 360:1526–1538. https://doi.org/10.1056/NEJMra0800028
Ferreira VM, Schulz-Menger J, Holmvang G, Kramer CM, Carbone I, Sechtem U, Kindermann I, Gutberlet M, Cooper LT, Liu P, Friedrich MG (2018) Cardiovascular magnetic resonance in nonischemic myocardial inflammation: expert recommendations. J Am Coll Cardiol 72:3158–3176. https://doi.org/10.1016/j.jacc.2018.09.072
Fischer K, Obrist SJ, Erne SA, Stark AW, Marggraf M, Kaneko K, Guensch DP, Huber AT, Greulich S, Aghayev A, Steigner M, Blankstein R, Kwong RY, Grani C (2020) Feature tracking myocardial strain incrementally improves prognostication in myocarditis beyond traditional CMR imaging features. JACC Cardiovasc Imaging 13:1891–1901. https://doi.org/10.1016/j.jcmg.2020.04.025
Fischer K, Marggraf M, Stark AW, Kaneko K, Aghayev A, Guensch DP, Huber AT, Steigner M, Blankstein R, Reichlin T, Windecker S, Kwong RY, Grani C (2020) Association of ECG parameters with late gadolinium enhancement and outcome in patients with clinical suspicion of acute or subacute myocarditis referred for CMR imaging. PLoS ONE 15:e0227134. https://doi.org/10.1371/journal.pone.0227134
Grani C, Biere L, Eichhorn C, Kaneko K, Agarwal V, Aghayev A, Steigner M, Blankstein R, Jerosch-Herold M, Kwong RY (2019) Incremental value of extracellular volume assessment by cardiovascular magnetic resonance imaging in risk stratifying patients with suspected myocarditis. Int J Cardiovasc Imaging 35:1067–1078. https://doi.org/10.1007/s10554-019-01552-6
Grani C, Eichhorn C, Biere L, Murthy VL, Agarwal V, Kaneko K, Cuddy S, Aghayev A, Steigner M, Blankstein R, Jerosch-Herold M, Kwong RY (2017) Prognostic value of cardiac magnetic resonance tissue characterization in risk stratifying patients with suspected myocarditis. J Am Coll Cardiol 70:1964–1976. https://doi.org/10.1016/j.jacc.2017.08.050
Bernhard B, Schnyder A, Garachemani D, Fischer K, Tanner G, Safarkhanlo Y, Stark AW, Schütze J, Pavlicek-Bahlo M, Greulich S, Johner C, Wahl A, Benz DC, Kwong RY, Gräni C (2023) Prognostic value of right ventricular function in patients with suspected myocarditis undergoing cardiac magnetic resonance. JACC Cardiovasc Imaging 16:28–41. https://doi.org/10.1016/j.jcmg.2022.08.011
Grani C, Stark AW, Fischer K, Furholz M, Wahl A, Erne SA, Huber AT, Guensch DP, Vollenbroich R, Ruberti A, Dobner S, Heg D, Windecker S, Lanz J, Pilgrim T (2022) Diagnostic performance of cardiac magnetic resonance segmental myocardial strain for detecting microvascular obstruction and late gadolinium enhancement in patients presenting after a ST-elevation myocardial infarction. Front Cardiovasc Med 9:909204. https://doi.org/10.3389/fcvm.2022.909204
Cerqueira MD, Weissman NJ, Dilsizian V, Jacobs AK, Kaul S, Laskey WK, Pennell DJ, Rumberger JA, Ryan T, Verani MS (2002) Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart. A statement for healthcare professionals from the cardiac imaging committee of the council on clinical cardiology of the American heart association. Circulation 105:539–542. https://doi.org/10.1161/hc0402.102975
Kawel-Boehm N, Hetzel SJ, Ambale-Venkatesh B, Captur G, Francois CJ, Jerosch-Herold M, Salerno M, Teague SD, Valsangiacomo-Buechel E, van der Geest RJ, Bluemke DA (2020) Reference ranges (“normal values”) for cardiovascular magnetic resonance (CMR) in adults and children: 2020 update. J Cardiovasc Magn Reson 22:87. https://doi.org/10.1186/s12968-020-00683-3
Koo TK, Li MY (2016) A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J Chiropr Med 15:155–163. https://doi.org/10.1016/j.jcm.2016.02.012
Stathogiannis K, Mor-Avi V, Rashedi N, Lang RM, Patel AR (2020) Regional myocardial strain by cardiac magnetic resonance feature tracking for detection of scar in ischemic heart disease. Magn Reson Imaging 68:190–196. https://doi.org/10.1016/j.mri.2020.02.009
Yu S, Zhou J, Yang K, Chen X, Zheng Y, Zhao K, Song J, Ji K, Zhou P, Yan H, Zhao S (2021) Correlation of myocardial strain and late gadolinium enhancement by cardiac magnetic resonance after a first anterior ST-segment elevation myocardial infarction. Front Cardiovasc Med 8:705487. https://doi.org/10.3389/fcvm.2021.705487
Polacin M, Karolyi M, Eberhard M, Matziris I, Alkadhi H, Kozerke S, Manka R (2022) Segmental strain for scar detection in acute myocardial infarcts and in follow-up exams using non-contrast CMR cine sequences. BMC Cardiovasc Disord 22:226. https://doi.org/10.1186/s12872-022-02664-z
Erley J, Genovese D, Tapaskar N, Alvi N, Rashedi N, Besser SA, Kawaji K, Goyal N, Kelle S, Lang RM, Mor-Avi V, Patel AR (2019) Echocardiography and cardiovascular magnetic resonance based evaluation of myocardial strain and relationship with late gadolinium enhancement. J Cardiovasc Magn Reson 21:46. https://doi.org/10.1186/s12968-019-0559-y
Gavara J, Rodriguez-Palomares JF, Rios-Navarro C, Valente F, Monmeneu JV, Lopez-Lereu MP, Ferreira-Gonzalez I, Garcia Del Blanco B, Otaegui I, Canoves J, de Dios E, Perez N, Racugno P, Bonanad C, Minana G, Marcos V, Barrabes JA, Evangelista A, Moratal D, Bayes-Genis A, Nunez J, Chorro FJ, Bodi V (2021) Longitudinal strain in remote non-infarcted myocardium by tissue tracking CMR: characterization, dynamics, structural and prognostic implications. Int J Cardiovasc Imaging 37:241–253. https://doi.org/10.1007/s10554-020-01890-w
Elias J, van Dongen IM, Hoebers LP, Ouweneel DM, Claessen B, Ramunddal T, Laanmets P, Eriksen E, Piek JJ, van der Schaaf RJ, Ioanes D, Nijveldt R, Tijssen JG, Henriques JPS, Hirsch A, EXPLORE investigators (2020) Recovery and prognostic value of myocardial strain in ST-segment elevation myocardial infarction patients with a concurrent chronic total occlusion. Eur Radiol 30:600–608. https://doi.org/10.1007/s00330-019-06338-x
Aquaro GD, Ghebru Habtemicael Y, Camastra G, Monti L, Dellegrottaglie S, Moro C, Lanzillo C, Scatteia A, Di Roma M, Pontone G, Perazzolo Marra M, Barison A, Di Bella G (2019) Cardiac magnetic resonance" working group of the Italian society of C. Prognostic value of repeating cardiac magnetic resonance in patients with acute myocarditis. J Am Coll Cardiol 74:2439–2448. https://doi.org/10.1016/j.jacc.2019.08.1061
Greulich S, Seitz A, Müller KAL, Grün S, Ong P, Ebadi N, Kreisselmeier KP, Seizer P, Bekeredjian R, Zwadlo C, Gräni C, Klingel K, Gawaz M, Sechtem U, Mahrholdt H (2020) Predictors of mortality in patients with biopsy-proven viral myocarditis: 10-year outcome data. J Am Heart Assoc 9:e015351. https://doi.org/10.1161/jaha.119.015351
Eitel I, Stiermaier T, Lange T, Rommel KP, Koschalka A, Kowallick JT, Lotz J, Kutty S, Gutberlet M, Hasenfuss G, Thiele H, Schuster A (2018) Cardiac magnetic resonance myocardial feature tracking for optimized prediction of cardiovascular events following myocardial infarction. JACC Cardiovasc Imaging 11:1433–1444. https://doi.org/10.1016/j.jcmg.2017.11.034
Gavara J, Rodriguez-Palomares JF, Valente F, Monmeneu JV, Lopez-Lereu MP, Bonanad C, Ferreira-Gonzalez I, Garcia Del Blanco B, Rodriguez-Garcia J, Mutuberria M, de Dios E, Rios-Navarro C, Perez-Sole N, Racugno P, Paya A, Minana G, Canoves J, Pellicer M, Lopez-Fornas FJ, Barrabes J, Evangelista A, Nunez J, Chorro FJ, Garcia-Dorado D, Bodi V (2018) Prognostic value of strain by tissue tracking cardiac magnetic resonance after ST-segment elevation myocardial infarction. JACC Cardiovasc Imaging 11:1448–1457. https://doi.org/10.1016/j.jcmg.2017.09.017
Sardana M, Konda P, Hashmath Z, Oldland G, Gaddam S, Miller R, Satija V, Ansari B, Lee J, Mustafa A, Akers SR, Chirinos JA (2019) Usefulness of left ventricular strain by cardiac magnetic resonance feature-tracking to predict cardiovascular events in patients with and without heart failure. Am J Cardiol 123:1301–1308. https://doi.org/10.1016/j.amjcard.2019.01.025
Tat E, Ball C, Camren GP, Wroblewski I, Dajani KA, Goldberg A, Kinno M, Sanagala T, Syed MA, Wilber DJ, Rabbat M (2022) Impact of late gadolinium enhancement extent, location, and pattern on ventricular tachycardia and major adverse cardiac events in patients with ischemic vs. non-ischemic cardiomyopathy. Front Cardiovasc Med 9:1026215. https://doi.org/10.3389/fcvm.2022.1026215
Bernhard B, Tanner G, Garachemani D, Schnyder A, Fischer K, Huber AT, Safarkhanlo Y, Stark AW, Guensch DP, Schütze J, Greulich S, Bastiaansen JAM, Pavlicek-Bahlo M, Benz DC, Kwong RY, Gräni C (2023) Predictive value of cardiac magnetic resonance right ventricular longitudinal strain in patients with suspected myocarditis. J Cardiovasc Magn Reson 25:49. https://doi.org/10.1186/s12968-023-00957-6
Buttà C, Zappia L, Laterra G, Roberto M (2020) Diagnostic and prognostic role of electrocardiogram in acute myocarditis: a comprehensive review. Ann Noninvasive Electrocardiol. https://doi.org/10.1111/anec.12726
Chen J, Chen S, Li Z, Zhou P, Huang W, Wang H, Shi J, Ni Y, Lin L, Lei Y (2020) Role of electrocardiograms in assessment of severity and analysis of the characteristics of ST elevation in acute myocarditis: a two-centre study. Exp Ther Med 20:20. https://doi.org/10.3892/etm.2020.9148
Feisst A, Kuetting DLR, Dabir D, Luetkens J, Homsi R, Schild HH, Thomas D (2018) Influence of observer experience on cardiac magnetic resonance strain measurements using feature tracking and conventional tagging. Int J Cardiol Heart Vasc 18:46–51. https://doi.org/10.1016/j.ijcha.2018.02.007
Mangion K, Burke NMM, McComb C, Carrick D, Woodward R, Berry C (2019) Feature-tracking myocardial strain in healthy adults- a magnetic resonance study at 3.0 tesla. Sci Rep 9:3239. https://doi.org/10.1038/s41598-019-39807-w
Morgan AE, Zhang Y, Tartibi M, Goldburg S, Kim JJ, Nguyen TD, Guccione J, Ge L, Weinsaft JW, Ratcliffe MB (2018) Ischemic mitral regurgitation: abnormal strain overestimates nonviable myocardium. Ann Thorac Surg 105:1754–1761. https://doi.org/10.1016/j.athoracsur.2018.01.005
Wamil M, Borlotti A, Liu D, Briosa EGA, Bracco A, Alkhalil M, De Maria GL, Piechnik SK, Ferreira VM, Banning AP, Kharbanda RK, Neubauer S, Choudhury RP, Channon KM, Dall’Armellina E (2019) Combined T1-mapping and tissue tracking analysis predicts severity of ischemic injury following acute STEMI-an Oxford Acute Myocardial Infarction (OxAMI) study. Int J Cardiovasc Imaging 35:1297–1308. https://doi.org/10.1007/s10554-019-01542-8
Acknowledgements
We thank Lukas Lüthi, Karini Ampalam and Laura Morf from the research study team for their excellent support.
Funding
Open access funding provided by University of Bern. Dr Bernhard receives funding from the Swiss National Science Foundation (P500PM_210908).
Author information
Authors and Affiliations
Contributions
All authors read and approved the final manuscript and significantly contributed. Authors BB, RYK, and CG designed the study. Authors PJ, NG, AWS, JS, DPG, and DCB collected data for the study and analyzed CMR images or provided infrastructure. JAMB, and MPB were responsible for clinical management and follow-up of study participants. Authors IS, YS and KF were responsible for data cleaning and/or statistical analysis.
Corresponding author
Ethics declarations
Conflict of interest
Dr Gräni received research funding from the Swiss National Science Foundation and Innosuisse and the Center for Artificial Intelligence in Medicine Research Project Fund University Bern, GAMBIT foundation, Novartis for Medical-Biological Research foundation, outside of the submitted work. Dr Bernhard received a career development grant from the Swiss National Science Foundation. Dr Benz received consultancy fees from Pfizer and travel support from Philips Healthcare, outside of the submitted work. All other authors report no relevant conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bernhard, B., Joss, P., Greisser, N. et al. Prognostic value of visual and quantitative CMR regional myocardial function in patients with suspected myocarditis. Int J Cardiovasc Imaging 40, 907–920 (2024). https://doi.org/10.1007/s10554-024-03059-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-024-03059-1