Abstract
Accurate standardization of left atrium volume (LAV) in patients with obesity is challenging. The aim of this study was to investigate and to examine the relation between LAV indexed to height2 and left atrial function in patients with moderate to severe obesity. Echocardiograms of patients with moderate to severe obesity (body mass index (BMI) ≥ 35 kg/m2) without known cardiac disease were analyzed. LAV was indexed to body surface area (BSA) and height2, and patients were divided into those with or without left atrial enlargement (LAE) based on normalization using either BSA (LAEbsa) or height2 (LAEh2). Using speckle tracking echocardiography, LA reservoir strain (LASr), LA conduit strain (LAScd), and LA contractile strain (LASct) were assessed as a measure of LA function. LA dysfunction was defined as LASct < 14%. A total of 142 patients were included in the analysis of whom 54.2% had LAEh2 and 18.3% LAEBSA. The LAEh2 group had significantly lower LASct (12.2% ± 3.2% vs. 13.6% ± 4.5%, p = 0.019) as compared to the patients without LAEh2. Significantly more patients with LA dysfunction would be correctly identified by LAEh2 than by LAEBSA (41.5% vs. 15.0%, p < 0.001). In patients with moderate to severe obesity, the use of LAEh2 identified significantly more patients with decreased LA function. LAVh2 should be preferred over LAVBSA in patients with moderate to severe obesity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Left atrial enlargement (LAE) is well established as a prognostic marker in heart failure with preserved ejection fraction (HFpEF) and is used as one of the morphologic diagnostic criteria to diagnose HFpEF [1]. Current ESC guidelines recommend indexing LAV to body surface area (LAVBSA) to determine LAE because of the widely available data [2]. However, since BSA is mainly driven by an increase in fat mass, indexing LAV to BSA can lead to overcorrection of LAV among patients with obesity and thereby has the potential of normalizing LA dilatation. Moreover, LAV indexed to BSA is an isometric measure that assumes a linear relationship between LAV and BSA, which is incorrect since heart and body size do not grow proportionally [3]. This is especially relevant since the majority of heart failure patients are either overweight or have obesity [4, 5].
It has been suggested that a more appropriate measure to define LAE in patients with obesity could be to use allometric scaling by indexing LAV to height2 (LAVh2) [6]. Recent studies have demonstrated that indexing LAV to height2 better predicts mortality in patients with severe obesity, whereas indexing to BSA has limited predictive value in these patients [7, 8].
Another emerging parameter of the LA in obesity patients is LA strain [9]. A previous study by our group demonstrated that patients with obesity have impairment in LA function before alterations in conventional echocardiographic parameters occur [10]. The potential value of LAVh2 may be underscored if this parameter would be related to LA function, which has not been investigated before. Therefore, the purpose of our study was to investigate the relation between LAVh2 and LAS, and to further establish the added value of LAVh2 as a parameter for LAE in patients with moderate to severe obesity.
Methods
For this study, echocardiograms of the CARDIOBESE study and AF OBESE study were used. The CARDIOBESE and AF OBESE study are both multicenter prospective cross-sectional studies in which 192 patients with obesity without known cardiac disease were enrolled, who were referred for bariatric surgery in the Franciscus Gasthuis & Vlietland and Maasstad Ziekenhuis, both in Rotterdam, the Netherlands [11, 12]. Patients were enrolled if they were between 35 and 65 years old. All patients had a BMI of ≥ 35 kg/m2. Height (in meters) and weight (in kilograms) were measured at the time of the echocardiogram. BMI was calculated as weight/height2. BSA was calculated by using the Du Bois formula (BSA [m2]) = 0.007184 x height [cm]0.725 x weight [kg]0.425. Study protocols were approved by the local ethics committee and participants provided written informed consent.
Transthoracic echocardiography
Two-dimensional greyscale harmonic images were obtained in the left lateral decubitus position using a commercially available ultrasound system (EPIQ 7, Philips, the Netherlands), equipped with a broadband (1–5 MHz) X5-1 transducer. All acquisitions and measurements were performed according to the current guidelines [2, 13]. LAV was measured on the 4-chamber and 2-chamber view. LAV was then indexed to height2 (LAVh2) and BSA (LAVBSA). LAEh2 was defined according to the ESC/ESH hypertension guidelines (LAVh2 >18.5 ml/m2 in males and LAVh2 >16.5 ml/m2 in females) [14]. When BSA was used, LAEBSA was defined as LAVBSA > 34 ml/m2 [2]. For a sub-analysis, the study population was split by obesity class according to the World Health Organization definition to check the difference in prevalence of LAE when using LAVh2 and LAVBSA [15].
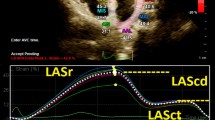
LA strain was measured with speckle tracking and analyzed offline with dedicated software (TomTec-Arena, integrated in Sectra IDS7). The apical 4-chamber view was used preferably for the analysis. LA endocardial borders were automatically traced using end-diastole as reference. When tracking was suboptimal, fine-tuning was performed manually. If the 4-chamber view was of poor image quality, the 2-chamber view was used. Patients with images of insufficient quality to perform LA strain analysis were excluded. LA function was described according to the three phases of the LA cycle: LA reservoir strain (LASr) which starts at the end of ventricular diastole (mitral valve closure) and continues until mitral valve opening, LA conduit strain (LAScd) which occurs from the time of mitral valve opening through diastasis until the onset of LA contraction, and LA contractile strain (LASct) which occurs from the onset of LA contraction until the end of ventricular diastole (mitral valve closure). LASr, LAScd, and LASct were computed in all patients. An example of LAS measurement in a patient with obesity is shown in Fig. 1.All strain values are reported as absolute values for improved readability and data interpretation [16]. LA dysfunction was defined as LASct < 14% [17].
Statistical analysis
Normally distributed data are presented as means and standard deviation, skewed data as medians and inter-quartile range, and categorical variables as percentages and frequencies. Continuous variables were compared using the independent student T-test in case of normally distributed data and the Mann-Whitney U test for non-normally distributed data. Categorical data were analyzed with the Chi-square test and the McNemar’s test for respectively normally and non-normally distributed data. Statistical significance was defined as a p value less than 0.05. Univariable binary logistic regression (with odds ratio (OR) as main analysis) was used to assess whether abnormal LASct was associated with parameters of diastolic function. Parameters of diastolic function were dichotomized according to defined normal values [2]. Analyses were performed using SPSS Statistical Package version 28.0.
Results
Image quality was insufficient to quantify LA strain in 50 patients, leaving 142 patients for the analysis. Clinical characteristics of the study population are shown in Table 1. 79.6% of the patients were female. Mean age and mean BMI were respectively 52.3 ± 7.3 years and 42.4 ± 4.4 kg/m2. As shown in Table 2, in the total study population LAVBSA was 25.6 ± 7.5 ml/m2 and LAVh2 was 18.4 ± 5.3 ml/m2, resulting in a total of 26 (18.3%) patients having LAEBSA, and 77 (54.2%) patients having LAEh2. In Fig. 2, LAVBSA and LAVh2 were plotted against BMI. As can be seen, LAVBSA decreased with increasing BMI, whereas LAVh2 increased with increasing BMI. The prevalence of LAEh2 was significantly higher than LAEBSA in both obesity class groups (obesity class 2: p < 0.001; obesity class 3 p < 0.001) (Fig. 3). As for LA function, LASr was 30.0 ± 7.8%, LAScd 17.1 ± 6.4%, and LASct 12.8 ± 3.9% in the total study population.
Comparison between patients with and without LAEh2
As presented in Table 1, there was a small but significant difference in age (51.1 ± 7.9 years vs. 53.7 ± 6.2 years, p = 0.033) between the groups. Patients in the LAEh2 group more often had a history of hypertension and more often used diuretics (42.9% vs. 24.6%, p = 0.023, and 26.0% vs. 10.8%, p = 0.021, respectively).
Echocardiographic parameters are shown in Table 2. Apart from an expected significant difference in LAVBSA (30.8 ± 6.0 ml/m2 vs. 19.6 ± 3.1 ml/m2, p < 0.001), there were no differences in other conventional diastolic echocardiographic parameters between the groups. As for LA function, the LAEh2 group had significantly lower LASct (12.2 ± 3.2% vs. 13.6 ± 4.5%, p = 0.019). There was no difference in LASr and LAScd between groups.
LAE in relation to LASct
In Fig. 4 the correlations between LAVh2, LAVBSA, and LASct are depicted. There was a significant, but weak, negative correlation for both LAVh2 and LASct (r=-0.22, p = 0.009) and LAVBSA and LASct (r=-0.21, p = 0.015). Significantly more patients with LA dysfunction as defined by LASct < 14% would have been correctly classified by LAEh2 as compared to LAEBSA (41.5% vs. 15.0%, p < 0.001) (Figs. 4 and 5). Table 3 shows the association of various LV diastolic parameters with LASct. In binary logistic regression LAEh2 was significantly associated with an abnormal LASct (OR 2.64, CI 1.29–5.42, p = 0.008).
A: Relation between left atrial volume indexed to body surface area (LAVBSA) and left atrial contractile strain (LASct). Horizontal red dashed line represents left atrial enlargement indexed to body surface area threshold. Vertical red dashed line represents left atrial strain contractile dysfunction. B: Relation between left atrial volume indexed to height2 (LAVh2) and LASct. Horizontal red dashed lines represent left atrial enlargement indexed to height2 thresholds (female and male respectively). Vertical red dashed line represents left atrial strain contractile dysfunction
Discussion
We demonstrated that, in subjects with moderate and severe obesity without known cardiac disease, LAEh2 was associated with an increased risk for LA dysfunction, in contrast to LAEBSA and other traditional parameters of LV diastolic function. Furthermore, we confirmed findings of previous studies, showing that indexation of LAV to height2 resulted in a higher prevalence of LAE compared to indexation of LAV to BSA in these subjects. Considering the limitations of indexation to BSA in obesity, LAEh2 may be of added value in determining increased risk of cardiac dysfunction in patients with obesity.
LAE in obesity
Obesity is an important risk factor for developing LAE [18], which is an essential parameter in identifying diastolic dysfunction and HFpEF [1, 2]. In addition, both obesity and LAE are associated with an increased risk for developing atrial fibrillation (AF) [19,20,21,22]. There are several mechanisms by which obesity can lead to LAE. For example, obesity can induce hemodynamic changes that can alter cardiac structures, it can cause atrial myopathy related to systemic inflammation, and promote paracrine effects from epicardial adipose tissue [23,24,25].
Normalization of heart chamber sizes is common and necessary, as it reduces the effect of dissimilarities in patients’ proportions. Additionally, normalization allows inter- and intragroup comparisons of cardiac dimensions [26]. Normal values enable the possibility to define normal ranges, that can be used to predict, diagnose, and monitor disease. The use of BSA as indexation method in LA scaling dates back to the 1980s [27], and is still recommended in the current guidelines [2]. However, indexation of LAV to BSA is inaccurate for patients with obesity [6]. The reasons for this are several fold. First of all, indexing LAV to BSA assumes a linear relationship. However, data on the growth patterns of the human heart indicate that the growth relationship is exponential rather than linear [26, 28, 29]. This can be overcome by choosing allometric scaling instead, as allometric scaling assumes an exponential relationship [6]. A few previous studies have assessed different indexation methods in patients with obesity. First, Zong et al. found that allometric scaling was superior to conventional isometric indexation in a population of 717 patients with obesity with a mean BMI of 42.2 kg/m2 [30]. Second, in a paper by Carnavelini et al., a similar conclusion was drawn in 63 patients with mild, and 26 patients with moderate obesity [31]. Although both studies demonstrated that allometric scaling was superior to isometric scaling, potential supportive data regarding the relation of alternative indexing methods with LA function was not available.
The second concern with indexing LAV to BSA in obesity, is that cardiac size is driven by fat free mass (FFM) [26]. In normal weight subjects, BSA is a suitable surrogate for FFM and thus a suitable scaler to index LAV [32]. However, in patients with obesity, BSA is disproportional to FFM and therefore possibly overcorrects LAV [6]. Height appears to be a better estimate for FFM [6]. Our results are consistent with this notion, as can be seen in Fig. 1 where LAV indexed to height2 was related to increasing BMI as expected, in contrast to LAV indexed to BSA. In addition, we found that indexing LAV to height2 resulted in a higher prevalence of LAE compared to BSA. A recent study showed similar results, where as many as 55.4% of the severely obese patients were reclassified as having LAE when height2 was used for indexation instead of BSA [7]. Additionally, recent studies have demonstrated that indexation of LAV to height2 has better predictive value concerning clinical outcomes in patients with obesity [7, 8]. However, both studies did not investigate the relation between LAVh2 and LA function.
Relation between LAVh2 and LA function in obesity
In order to investigate whether LAVh2 may also better identify LA dysfunction in patients with obesity as compared to LAVBSA, this study was the first to relate LAVh2 and LAVBSA to LA strain. Recently, LA strain has emerged as a parameter that has potential added value in identifying diastolic dysfunction. LASr and LASct are both associated with LV filling pressures [33,34,35]. Patients with obesity with LAEh2 had significantly lower LASct compared to patients without LAEh2. Also, more patients with abnormal LASct were identified by LAEh2 as compared to LAEBSA. In addition, LAEh2 was associated with an increased risk (OR 2.64) for an abnormal LASct, in contrast to LAEBSA and other traditional diastolic parameters. Our novel findings underscore the notion that LAVh2 is not only a more sensitive measure of LAE in patients with obesity, but indeed more sensitive for identification of LA dysfunction as well. As LAE and LA dysfunction are important parameters of LV diastolic dysfunction, use of LAVh2 may improve the utility of a diastolic function qualification algorithm. However, we have not investigated that in our study. Further studies confirming improved prognostic value of LAVh2 as compared to LAVBSA are mandatory first.
Study limitations
This study has some limitations that should be noted. First of all, LA strain analysis requires good image quality and not all our subjects (26%) had analyzable LA images, which may have affected the identified proportion of LA dysfunction. Second, a considerable proportion of the subjects had comorbidities, such as hypertension and diabetes, that can also affect LA function. Third, our cohort mostly consisted of females which could have biased the results. Around 80% of patients who undergo bariatric surgery are female, which explains the high percentage of females in our study. Fourth, only LASct and not LASr and LAScd were different between patients with and without LAEh2. Although most of the previous research has focused on LASr, added value of LASct has already been proven as well [16] and is therefore also considered to be an important measure of LA function. Finally, diagnostic value of other echocardiographic parameters in subjects with moderate to severe obesity may also improve when indexed to height2 instead of BSA. However, this fell beyond the scope of the current study.
Conclusion
Relatively easy assessment of LAVh2 could overcome inherent limitations of LAVBSA in patients with obesity and thereby contribute to the detection of cardiac dysfunction in these patients. LAVh2 was more sensitive for detection of LAE and better related to LA dysfunction as compared to the current standard of normalization of LAV for BSA in our population of patients with moderate to severe obesity without known cardiac disease. With the rising prevalence of obesity worldwide, it is pivotal to have an early and accurate assessment of cardiac dysfunction in order to prevent further deterioration to heart failure. Early detection can lead to timely initiation of lifestyle modifications and treatment, and therefore reduce the associated risks and morbidity of obesity.
References
McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Bohm M et al (2021) 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J 42(36):3599–3726
Lang RM, Badano LP, Mor-Avi V, Afilalo J, Armstrong A, Ernande L et al (2015) Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging 16(3):233–270
de Simone G, Galderisi M (2014) Allometric normalization of cardiac measures: producing better, but imperfect, accuracy. J Am Soc Echocardiogr 27(12):1275–1278
Pfeffer MA, Shah AM, Borlaug BA (2019) Heart failure with preserved ejection Fraction in Perspective. Circ Res 124(11):1598–1617
Loai S, Cheng HM (2020) Heart failure with preserved ejection fraction: the missing pieces in diagnostic imaging. Heart Fail Rev 25(2):305–319
Jeyaprakash P, Moussad A, Pathan S, Sivapathan S, Ellenberger K, Madronio C et al (2021) A systematic review of scaling left atrial size: are alternative indexation methods required for an increasingly obese Population? J Am Soc Echocardiogr.
Davis EF, Crousillat DR, He W, Andrews CT, Hung JW, Danik JS (2022) Indexing left Atrial volumes: alternative indexing methods better predict outcomes in overweight and obese populations. JACC Cardiovasc Imaging 15(6):989–997
Olsen FJ, Biering-Sorensen T (2022) Validation of alternative left atrial indexation methods in obesity. J Am Soc Echocardiogr.
Yuda S (2021) Current clinical applications of speckle tracking echocardiography for assessment of left atrial function. J Echocardiogr 19(3):129–140
Aga YS, Kroon D, Snelder SM, Biter LU, de Groot LE, Zijlstra F et al (2023) Decreased left atrial function in obesity patients without known cardiovascular disease. Int J Cardiovasc Imaging 39(3):471–479
Snelder SM, de Groot-de Laat LE, Biter LU, Castro Cabezas M, Pouw N, Birnie E et al (2020) Subclinical cardiac dysfunction in obesity patients is linked to autonomic dysfunction: findings from the CARDIOBESE study. ESC Heart Fail 7(6):3726–3737
Snelder SM, de Groot-de Laat LE, Biter LU, Castro Cabezas M, van de Geijn GJ, Birnie E et al (2018) Cross-sectional and prospective follow-up study to detect early signs of cardiac dysfunction in obesity: protocol of the CARDIOBESE study. BMJ Open 8(12):e025585
Nagueh SF, Smiseth OA, Appleton CP, Byrd BF 3rd, Dokainish H, Edvardsen T et al (2016) Recommendations for the evaluation of left ventricular diastolic function by Echocardiography: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging 17(12):1321–1360
Williams B, Mancia G, Spiering W, Agabiti Rosei E, Azizi M, Burnier M et al (2018) 2018 ESC/ESH guidelines for the management of arterial hypertension. Eur Heart J 39(33):3021–3104
Obesity and overweight fact sheet World Health Organization [Available from: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight
Smiseth OA, Baron T, Marino PN, Marwick TH, Flachskampf FA (2021) Imaging of the left atrium: pathophysiology insights and clinical utility. Eur Heart J Cardiovasc Imaging 23(1):2–13
Pathan F, D’Elia N, Nolan MT, Marwick TH, Negishi K (2017) Normal ranges of left atrial strain by Speckle-Tracking Echocardiography: a systematic review and Meta-analysis. J Am Soc Echocardiogr 30(1):59–70e8
Aiad NN, Hearon C Jr., Hieda M, Dias K, Levine BD, Sarma S (2019) Mechanisms of left atrial enlargement in obesity. Am J Cardiol 124(3):442–447
Lavie CJ, Pandey A, Lau DH, Alpert MA, Sanders P (2017) Obesity and Atrial Fibrillation Prevalence, Pathogenesis, and prognosis: Effects of Weight loss and Exercise. J Am Coll Cardiol 70(16):2022–2035
Nalliah CJ, Sanders P, Kottkamp H, Kalman JM (2016) The role of obesity in atrial fibrillation. Eur Heart J 37(20):1565–1572
Tiwari S, Schirmer H, Jacobsen BK, Hopstock LA, Nyrnes A, Heggelund G et al (2015) Association between diastolic dysfunction and future atrial fibrillation in the Tromso Study from 1994 to 2010. Heart 101(16):1302–1308
Wang TJ, Parise H, Levy D, D’Agostino RB, Sr., Wolf PA, Vasan RS et al (2004) Obesity and the risk of new-onset atrial fibrillation. JAMA 292(20):2471–2477
Boutens L, Hooiveld GJ, Dhingra S, Cramer RA, Netea MG, Stienstra R (2018) Unique metabolic activation of adipose tissue macrophages in obesity promotes inflammatory responses. Diabetologia 61(4):942–953
Packer M (2018) The epicardial adipose inflammatory triad: coronary atherosclerosis, atrial fibrillation, and heart failure with a preserved ejection fraction. Eur J Heart Fail 20(11):1567–1569
Packer M (2020) Do most patients with obesity or type 2 diabetes, and atrial fibrillation, also have undiagnosed heart failure? A critical conceptual framework for understanding mechanisms and improving diagnosis and treatment. Eur J Heart Fail 22(2):214–227
Dewey FE, Rosenthal D, Murphy DJ Jr., Froelicher VF, Ashley EA (2008) Does size matter? Clinical applications of scaling cardiac size and function for body size. Circulation 117(17):2279–2287
Henry WL, Ware J, Gardin JM, Hepner SI, McKay J, Weiner M (1978) Echocardiographic measurements in normal subjects. Growth-related changes that occur between infancy and early adulthood. Circulation 57(2):278–285
Batterham AM, George KP, Whyte G, Sharma S, McKenna W (1999) Scaling cardiac structural data by body dimensions: a review of theory, practice, and problems. Int J Sports Med 20(8):495–502
Gutgesell HP, Rembold CM (1990) Growth of the human heart relative to body surface area. Am J Cardiol 65(9):662–668
Zong P, Zhang L, Shaban NM, Pena J, Jiang L, Taub CC (2014) Left heart chamber quantification in obese patients: how does larger body size affect echocardiographic measurements? J Am Soc Echocardiogr 27(12):1267–1274
Carnevalini M, Deschle H, Amenabar A, Casso N, Gantesti J, Alfie L et al (2020) Evaluation of the size of cardiac structures in patients with high body mass index. Echocardiography 37(2):270–275
George K, Sharma S, Batterham A, Whyte G, McKenna W (2001) Allometric analysis of the association between cardiac dimensions and body size variables in 464 junior athletes. Clin Sci (Lond) 100(1):47–54
Borde D, Joshi S, Jasapara A, Joshi P, Asegaonkar B, Apsingekar P (2021) Left atrial strain as a single parameter to Predict Left ventricular diastolic dysfunction and elevated left ventricular filling pressure in patients undergoing off-pump coronary artery bypass grafting. J Cardiothorac Vasc Anesth 35(6):1618–1625
Lin J, Ma H, Gao L, Wang Y, Wang J, Zhu Z et al (2020) Left atrial reservoir strain combined with E/E’ as a better single measure to predict elevated LV filling pressures in patients with coronary artery disease. Cardiovasc Ultrasound 18(1):11
Singh A, Addetia K, Maffessanti F, Mor-Avi V, Lang RM (2017) LA strain for categorization of LV Diastolic Dysfunction. JACC Cardiovasc Imaging 10(7):735–743
Funding
This work was supported by a grant from Stichting BeterKeten.
Author information
Authors and Affiliations
Contributions
Y.A. and Y.A. were involved in the data analysis and manuscript writing. J.C., D.K., and S.S., participated in the data analysis. L.G.L., L.U.B., F.Z., J.J.B, B.M.D, were involved in manuscript preparation and participated in critical revision of all drafts of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yaar Aga and Yalin Acardag shared first authorship.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Aga, Y., Acardag, Y., Chin, J.F. et al. Improved identification of left atrial enlargement in patients with obesity. Int J Cardiovasc Imaging 40, 65–72 (2024). https://doi.org/10.1007/s10554-023-02981-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-023-02981-0