Abstract
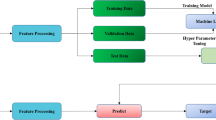
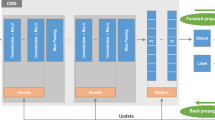
Recognizing early cardiac sarcoidosis (CS) imaging phenotypes can help identify opportunities for effective treatment before irreversible myocardial pathology occurs. We aimed to characterize regional CS myocardial remodeling features correlating with future adverse cardiac events by coupling automated image processing and data analysis on cardiac magnetic resonance (CMR) imaging datasets. A deep convolutional neural network (DCNN) was used to process a CMR database of a 10-year cohort of 117 consecutive biopsy-proven sarcoidosis patients. The maximum relevance − minimum redundancy method was used to select the best subset of all the features—24 (from manual processing) and 232 (from automated processing) left ventricular (LV) structural/functional features. Three machine learning (ML) algorithms, logistic regression (LogR), support vector machine (SVM) and multi-layer neural networks (MLP), were used to build classifiers to categorize endpoints. Over a median follow-up of 41.8 (inter-quartile range 20.4–60.5) months, 35 sarcoidosis patients experienced a total of 43 cardiac events. After manual processing, LV ejection fraction (LVEF), late gadolinium enhancement, abnormal segmental wall motion, LV mass (LVM), LVMI index (LVMI), septal wall thickness, lateral wall thickness, relative wall thickness, and wall thickness of 9 (out of 17) individual LV segments were significantly different between patients with and without endpoints. After automated processing, LVEF, end-diastolic volume, end-systolic volume, LV mass and wall thickness of 92 (out of 216) individual LV segments were significantly different between patients with and without endpoints. To achieve the best predictive performance, ML algorithms selected lateral wall thickness, abnormal segmental wall motion, septal wall thickness, and increased wall thickness of 3 individual segments after manual image processing, and selected end-diastolic volume and 7 individual segments after automated image processing. LogR, SVM and MLP based on automated image processing consistently showed better predictive accuracies than those based on manual image processing. Automated image processing with a DCNN improves data resolution and regional CS myocardial remodeling pattern recognition, suggesting that a framework coupling automated image processing with data analysis can help clinical risk stratification.





Similar content being viewed by others
References
Iannuzzi MC, Rybicki BA, Teirstein AS (2007) Sarcoidosis. N Eng J Med 357:2153–2165. https://doi.org/10.1056/NEJMra071714
Hamzeh N, Steckman DA, Sauer WH, Judson MA (2015) Pathophysiology and clinical management of cardiac sarcoidosis. Nat Rev Cardiol 12:278–288. https://doi.org/10.1038/nrcardio.2015.22
Patel MR, Cawley PJ, Heitner JF et al (2009) Detection of myocardial damage in patients with sarcoidosis. Circulation 120:1969–1977. https://doi.org/10.1161/circulationaha.109.851352
Kouranos V, Tzelepis GE, Rapti A et al (2017) Complementary role of CMR to conventional screening in the diagnosis and prognosis of cardiac sarcoidosis. JACC Cardiovasc Imaging 10:1437–1447. https://doi.org/10.1016/j.jcmg.2016.11.019
de Roos A, van den Berg-Huysmans A, Schoones JW (2017) Prognostic value of CMR-verified myocardial scarring in cardiac sarcoidosis: what to learn from a systematic review and meta-analysis? JACC Cardiovasc Imaging 10:421–423. https://doi.org/10.1016/j.jcmg.2016.07.018
Coleman GC, Shaw PW, Balfour PC Jr et al (2017) Prognostic value of myocardial scarring on CMR in patients with cardiac sarcoidosis. JACC Cardiovasc Imaging 10:411–420. https://doi.org/10.1016/j.jcmg.2016.05.009
Youssef G, Beanlands RS, Birnie DH, Nery PB (2011) Cardiac sarcoidosis: applications of imaging in diagnosis and directing treatment. Heart 97:2078–2087. https://doi.org/10.1136/hrt.2011.226076
Smedema JP, Snoep G, van Kroonenburgh MP et al (2005) Evaluation of the accuracy of gadolinium-enhanced cardiovascular magnetic resonance in the diagnosis of cardiac sarcoidosis. J Am Coll Cardiol 45:1683–1690. https://doi.org/10.1016/j.jacc.2005.01.047
Nagai T, Kohsaka S, Okuda S, Anzai T, Asano K, Fukuda K (2014) Incidence and prognostic significance of myocardial late gadolinium enhancement in patients with sarcoidosis without cardiac manifestation. Chest 146:1064–1072. https://doi.org/10.1378/chest.14-0139
Puntmann VO, Isted A, Hinojar R, Foote L, Carr-White G, Nagel E (2017) T1 and T2 mapping in recognition of early cardiac involvement in systemic sarcoidosis. Radiology 285:63–72. https://doi.org/10.1148/radiol.2017162732
Jeudy J, Burke AP, White CS, Kramer GB, Frazier AA (2015) Cardiac sarcoidosis: the challenge of radiologic-pathologic correlation—erratum. Radiographics 35:1316. https://doi.org/10.1148/rg.2015154010
Ise T, Hasegawa T, Morita Y et al (2014) Extensive late gadolinium enhancement on cardiovascular magnetic resonance predicts adverse outcomes and lack of improvement in LV function after steroid therapy in cardiac sarcoidosis. Heart 100:1165–1172. https://doi.org/10.1136/heartjnl-2013-305187
Nadel J, Lancefield T, Voskoboinik A, Taylor AJ (2015) Late gadolinium enhancement identified with cardiac magnetic resonance imaging in sarcoidosis patients is associated with long-term ventricular arrhythmia and sudden cardiac death. Eur Heart J Cardiovas Imaging 16:634–641. https://doi.org/10.1093/ehjci/jeu294
Chan K (2020) Extending the role of cardiac magnetic resonance in sarcoidosis risk stratification: editorial for “regional myocardial remodeling characteristics correlates with cardiac events in sarcoidosis.” J Magn Reson Imaging 52:510–511. https://doi.org/10.1002/jCMR.27096
Roberts WC, Becker TM, Hall SA (2018) Usefulness of total 12-lead QRS voltage as a clue to diagnosis of patients with cardiac sarcoidosis severe enough to warrant orthotopic heart transplant. JAMA Cardiol 3:64–68. https://doi.org/10.1001/jamacardio.2017.4172
Roberts WC, Chung MS, Ko JM, Capehart JE, Hall SA (2014) Morphologic features of cardiac sarcoidosis in native hearts of patients having cardiac transplantation. Am J Cardiol 113:706–712. https://doi.org/10.1016/j.amjcard.2013.11.015
Roberts WC, McAllister HA Jr, Ferrans VJ (1977) Sarcoidosis of the heart. A clinicopathologic study of 35 necropsy patients (group 1) and review of 78 previously described necropsy patients (group 11). Am J Med 63:86–108. https://doi.org/10.1016/0002-343(77)90121-8
Uemura A, Morimoto S, Hiramitsu S, Kato Y, Ito T, Hishida H (1999) Histologic diagnostic rate of cardiac sarcoidosis: evaluation of endomyocardial biopsies. Am heart J 138(2 Pt 1):299–302. https://doi.org/10.1016/s0002-8703(99)70115-8
Liu K, Ahmed J, Feiglin D (2017) A 54-year-old man with new-onset ventricular fibrillation. Heart 103:1922–1924. https://doi.org/10.1136/heartjnl-2017-312052
Lu C, Chen J, Suksaranjit P et al (2020) Regional myocardial remodeling characteristics correlates with cardiac events in sarcoidosis. J Magn Reson Imaging 52:499–509. https://doi.org/10.1002/jCMR.27057
Chen J, Lei J, Scalzetti E et al (2018) Myocardial contractile patterns predict future cardiac events in sarcoidosis. Int J Cardiovasc Imaging 34:251–262. https://doi.org/10.1007/s10554-017-1233-9
Pozo E, Kanwar A, Deochand R et al (2014) Cardiac magnetic resonance evaluation of left ventricular remodelling distribution in cardiac amyloidosis. Heart 100:1688–1695. https://doi.org/10.1136/heartjnl-2014-305710
Florian A, Masci PG, De Buck S et al (2012) Geometric assessment of asymmetric septal hypertrophic cardiomyopathy by CMR. JACC Cardiovasc imaging 5:702–711. https://doi.org/10.1016/j.jcmg.2012.03.011
Kawaji K, Codella NC, Prince MR et al (2009) Automated segmentation of routine clinical cardiac magnetic resonance imaging for assessment of left ventricular diastolic dysfunction. Circ Cardiovasc Imaging 2:476–484. https://doi.org/10.1161/circimaging.109.879304
Diller GP, Orwat S, Vahle J et al (2020) Prediction of prognosis in patients with tetralogy of Fallot based on deep learning imaging analysis. Heart 106:1007–1014. https://doi.org/10.1136/heartjnl-2019-315962
Bello GA, Dawes TJW, Duan J et al (2019) Deep learning cardiac motion analysis for human survival prediction. Nat Mach Intell 1:95–104. https://doi.org/10.1038/s42256-019-0019-2
Bai W, Sinclair M, Tarroni G et al (2018) Automated cardiovascular magnetic resonance image analysis with fully convolutional networks. J Cardiovasc Magn Reson 20:65. https://doi.org/10.1186/s12968-018-0471-x
Captur G, Radenkovic D, Li C et al (2017) Community delivery of semiautomated fractal analysis tool in cardiac mr for trabecular phenotyping. J Magn Reson Imaging 46:1082–1088. https://doi.org/10.1002/jCMR.25644
Lei J, Wang YG, Bhatta L et al (2019) Ventricular geometry-regularized QRSd predicts cardiac resynchronization therapy response: machine learning from crosstalk between electrocardiography and echocardiography. Int J Cardiovasc Imaging 35:1221–1229. https://doi.org/10.1007/s10554-019-01545-5
Biton Y, Goldenberg I, Kutyifa V et al (2016) Relative wall thickness and the risk for ventricular tachyarrhythmias in patients with left ventricular dysfunction. J Am Coll Cardiol 67:303–312. https://doi.org/10.1016/j.jacc.2015.10.076
Pedregosa F, Varoquaux G, Gramfort A, Michel V, Thirion B, Grisel O et al (2011) Sckikit-learn: machine learning in Python. J Mach Learn Res 12:2825–2830
Radovic M, Ghalwash M, Filipovic N, Obradovic Z (2017) Minimum redundancy maximum relevance feature selection approach for temporal gene expression data. BMC Bioinform 18:9. https://doi.org/10.1186/s12859-016-1423-9
Singh D, Sisodia DS, Singh P (2020) Multi-objective evolutionary approach for the performance improvement of learners using ensembling feature selection and discretization technique on medical data. Cur Med Imaging 16:355–370. https://doi.org/10.2174/1573405614666180903114534
Sabovčik F, Cauwenberghs N, Kouznetsov D et al (2020) Applying machine learning to detect early stages of cardiac remodelling and dysfunction. Eur Heart J Cardiovasc Imaging. https://doi.org/10.1093/ehjci/jeaa135
Goldstein BA, Navar AM, Carter RE (2017) Moving beyond regression techniques in cardiovascular risk prediction: applying machine learning to address analytic challenges. Eur Heart J 38:1805–1814. https://doi.org/10.1093/eurheartj/ehw302
Shah RV, Yeri AS, Murthy VL et al (2017) Association of multiorgan computed tomographic phenomap with adverse cardiovascular health outcomes: the framingham heart study. JAMA Cardiol 2:1236–1246. https://doi.org/10.1001/jamacardio.2017.3145
Johnson KW, Torres Soto J, Glicksberg BS et al (2018) Artificial intelligence in cardiology. J Am Coll Cardiol 71:2668–2679. https://doi.org/10.1016/j.jacc.2018.03.521
Beam AL, Kohane IS (2018) Big data and machine learning in health care. JAMA 319:1317–1318. https://doi.org/10.1001/jama.2017.18391
Obermeyer Z, Emanuel EJ (2016) Predicting the future—big data, machine learning, and clinical medicine. N Eng J Med 375:1216–1219. https://doi.org/10.1056/NEJMp1606181
Paetsch I, Jahnke C, Ferrari VA et al (2006) Determination of interobserver variability for identifying inducible left ventricular wall motion abnormalities during dobutamine stress magnetic resonance imaging. Eur Heart J 27:1459–1464. https://doi.org/10.1093/eurheartj/ehi883
Al’Aref SJ, Anchouche K, Singh G et al (2019) Clinical applications of machine learning in cardiovascular disease and its relevance to cardiac imaging. Eur Heart J 40:1975–1986. https://doi.org/10.1093/eurheartj/ehy404
Leiner T, Rueckert D, Suinesiaputra A et al (2019) Machine learning in cardiovascular magnetic resonance: basic concepts and applications. J Cardiovasc Magn Reson 21:61. https://doi.org/10.1186/s12968-019-0575-y
Singh A, Voss WB, Lentz RW, Thomas JD, Akhter N (2019) The diagnostic and prognostic value of echocardiographic strain. JAMA Cardiol 4:580–588. https://doi.org/10.1001/jamacardio.2019.1152
Velangi PS, Chen KA, Kazmirczak F et al (2020) Right ventricular abnormalities on cardiovascular magnetic resonance imaging in patients with sarcoidosis. JACC Cardiovasc Imaging 13:1395–1405. https://doi.org/10.1016/j.jcmg.2019.12.011
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lu, C., Wang, Y.G., Zaman, F. et al. Predicting adverse cardiac events in sarcoidosis: deep learning from automated characterization of regional myocardial remodeling. Int J Cardiovasc Imaging 38, 1825–1836 (2022). https://doi.org/10.1007/s10554-022-02564-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-022-02564-5