Abstract
Monoplane hemodynamic TEE (hTEE) monitoring (ImaCor® ClariTEE®) might be a useful alternative to continuously evaluate cardiovascular function and we aimed to investigate the feasibility and influence of hTEE monitoring on postoperative management in cardiac surgery patients. After IRB approval we reviewed the electronic data of cardiac surgery patients admitted to our intensive care between 01/01/2012 and 30/06/2013 in a case-controlled matched-pairs design. Patients were eligible for the study when they presented a sustained hemodynamic instability postoperatively with the clinical need of an extended hemodynamic monitoring: (a) hTEE (hTEE group, n = 18), or (b) transpulmonary thermodilution (control group, n = 18). hTEE was performed by ICU residents after receiving an approximately 6-h hTEE training session. For hTEE guided hemodynamic optimization an institutional algorithm was used. The hTEE probe was blindly inserted at the first attempt in all patients and image quality was at least judged to be adequate. The frequency of hemodynamic examinations was higher (ten complete hTEE examinations every 2.6 h) in contrast to the control group (one examination every 8 h). hTEE findings, including five unexpected right heart failure and one pericardial tamponade, led to a change of current therapy in 89 % of patients. The cumulative dose of epinephrine was significantly reduced (p = 0.034) and levosimendan administration was significantly increased (p = 0.047) in the hTEE group. hTEE was non-inferior to the control group in guiding norepinephrine treatment (p = 0.038). hTEE monitoring performed by ICU residents was feasible and beneficially influenced the postoperative management of cardiac surgery patients.
Similar content being viewed by others
Introduction
Several studies in the last decade found benefit in “goal directed therapy” (GDT) [1, 2]. Goal directed fluid administration reduced the length of hospital stay [3] and improved patient outcome after surgery [4, 5]. Excessive fluid restriction consistently increased the level of hypovolemia and the incidence of postoperative complications [6]. The fundamental principle behind GDT is optimization of oxygen delivery to tissues (DO2) by optimizing cardiovascular function using fluids, catecholamines, red blood cells and supplementary oxygen [7].
Cardiac output (CO) is one of the major determinates of oxygen delivery [8]. CO depends on: (a) preload, (b) afterload, (c) myocardial contractility, (d) valve function and (e) heart rate/rhythm, all of which can be examined using echocardiography. Postoperative low cardiac output syndrome (LCOS), especially in cardiac surgery, thus results in deficient oxygen delivery. Therefore, in cases of unexplained life-threatening hemodynamic instability—possibly persisting despite corrective therapy—a multiplane transesophageal echocardiographic examination is recommended (Class I indication) to identify the underlying pathophysiological cause, e.g. hypovolemia, reduced myocardial contractility, pericardial tamponade [9], and to guide goal-directed therapy [10, 11].
However, in practice conventional transesophgeal echocardiography can only be performed intermittently [12]. Hence, the new ImaCor® ClariTEE® might be a useful alternative in daily clinical practice. Because of its small size it can be introduced orally in the patient´s esophagus and can remain up to 72 h in situ. With its flexible probe tip the three most important two-dimensional views of the heart can be obtained on demand to identify and manage the pathophysiological causes of LCOS, thus the name “hemodynamic TEE” (hTEE).
Up to now, there have only been a few clinical trials investigating the possible benefit of using the ImaCor® ClariTEE® to guide hemodynamic therapy in high-risk surgical and/or critically ill patients [13–19]. Monoplane TEE in the hands of intensivists has been shown to provide clinically useful estimates of LV function, RV dilation, hypovolemia and occurrence of pericardial effusion, as shown evaluated by between experienced echocardiographers [16]. Furthermore, hTEE frequently led to changes in clinical management of hemodynamic unstable patients on ventilator support [15] and also of patients on ventricular assist devices [14]. We were therefore interested in the feasibility (=ease of insertion and technical operation) and influence (=utility as hemodynamic monitor) of hTEE monitoring on postoperative management in cardiac surgery patients during its clinical implementation.
Methods
After approval of the Charité Ethics Committee, Berlin, Germany (Study ID number: EA1-249-13; ClinGov. registration number: NCT02046954), we reviewed charts and data derived from two electronic patient data management systems (PDMS: COPRA GmbH, Sasbachwalden, Germany and SAP AG, Walldorf, Germany) of cardiac surgery patients admitted to our intensive care unit (ICU) of the department of anesthesiology and intensive care medicine at the Campus Charité Mitte, Berlin, between 01/01/2012 and 30/06/2013. Written informed consent was waived by the ethics committee.
Patients were eligible for the study when they presented with a sustained hemodynamic instability (i.e. lasting longer than 1 h after ICU admission) despite corrective therapy, and needed extended hemodynamic monitoring according to current recommendations [20, 21]. Hemodynamic instability was clinically defined as (two or more criteria): persistent tachycardia (heart rate >100/min), arterial hypotension (mean arterial pressure <60 mmHg), need for catechlominergic support (norepinephrine ≥0.1 µg/kg/min in combination with epinephrine ≥0.1 µg/kg/min and/or dobutamine ≥5 µg/kg/min and/or enoximone ≥1 µg/kg/min), lactate acidosis (pH < 7.2) and/or negative base excess (BE < −2 mmol/l), declining diuresis (<0.5 ml/kg/h), and/or suspicion of pericardial tamponade. Those criteria were used to define “hemodynamic instability” for the purpose of increasing assay sensitivity by (a) detecting the most hemodynamic compromised patients by means of (b) the most frequently documented values in the local PDMS. In order to estimate the influence of hTEE monitoring, we retrospectively investigated the following time periods where (a) hTEE (01/10/2012–30/06/2013), or (b) transpulmonary thermodilution (PICCO®, bolus administration technique) (01/01/2012–30/09/2012, e.g. before implementation of hTEE) was used. Exclusion criteria were: (a) age less than 18 years, (b) use of pulmonary artery catheter and (c) incomplete medical records. In addition to comparing morphometric, demographic and operative data, we evaluated (a) the feasibility (=ease of insertion and technical operation) of hTEE, (b) its utility as a hemodynamic monitor: amount and frequency of hemodynamic evaluations per patient, use and cumulative doses of catecholamines in the first week after extending hemodynamic monitoring, postoperative lactate time course, net fluid balance of the subsequent three postoperative days (POD), and (c) overall in-hospital mortality.
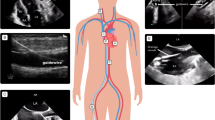
The ClariTEE® probe (Imacor, New York, USA) is a single use, miniaturized, single-plane (angle = 0°), two-dimensional transesophageal probe, which was orally inserted in the patient´s esophagus and remained up to 72 h in situ for continuously available (“on demand”) use (as approved by the Federal Drug Agency) (Fig. 1). The probe has a flexible tip providing ante- and retroflexion, and was connected to a standard echocardiographic system (Zura, Imacor, New York, USA) including standard recording capabilities (one loop consist of approximately five ECG cycles), provided by the company (http://imacorinc.com/htee/products/zura-evo-1-imaging-system.html). Two-dimensional measurements, e.g. left ventricular end-diastolic dimension (LVEDD) and pericardial effusion, and color Doppler mapping were possible.
During implementation of this transesophageal monoplane probe into clinical routine, residents of our ICU received a training session of approximately 6 h on this device in vivo and on a hTEE simulator under supervision of a team of certified echocardiographers (EACVI/EACTA Adult TEE accreditation and/or national certification by the German Society of Anesthesiology and Intensive Care Medicine)—available for 24 h 7 days a week. In addition, hTEE loops presenting specific hemodynamic conditions were shown on computer. Residents were trained to obtain the three most important two-dimensional hTEE views to determine hemodynamics: (a) transgastric short axis view (TG SAX) to evaluate global systolic left ventricular (LV) function, paradoxical septal motion, and pericardial effusion; (b) midesophageal four chamber view (4C) to evaluate global systolic biventricular morphology/function, paradoxical septal motion, mitral valve morphology, and pericardial effusion; and (c) midesophageal superior vena cava (SVC) view to evaluate volume responsiveness and aortic valve morphology/function [17]. A complete examination was defined to consist of these three views. After each examination residents had to complete a report with their qualitative results: (1) global systolic function of the left ventricle (LV): normal, slightly/moderately/severely reduced; (2) systolic function of the right ventricle: normal, reduced; (3) RV/LV-Index: normal, moderately/severely dilated [22]; (4) position and mobility of the interatrial septum (IAS): (hyper-)mobile, centered, right- or leftward shift [23]; (5) position/motion of the interventricular septum: normal, paradoxical motion; (6) signs of hypovolemia: kissing papillary muscles, small cardiac chamber areas, (hyper-)mobile IAS, collapse of SVC under mechanical ventilation [24]; (7) presence of pericardial effusion with potential signs of cardiac tamponade. Certified echocardiographers then reviewed findings and interpretations of ICU residents. Additionally, overall image quality was evaluated by the ICU residents as optimal, adequate (not optimal but sufficient for hemodynamic guidance), or inadequate and was further reviewed by certified echocardiographers. ICU residents then had to propose a therapy based on their findings according to an institutional algorithm (Fig. 2). In case of unexpected findings and/or difficulties in interpretation of the gained images a cardiologist and/or a certified echocardiographer had to be called to perform a multiplane TEE (“seek expert help”). Patients who were monitored with transpulmonary thermodilution were treated in a similar approach: therapy was guided by the measured parameters (cardiac index, systemic vascular resistance, pulmonary artery pressure, etc.) according to institutional standards guided by the German S3 guidelines [20].
For informational purposes, cardiopulmonary bypass (CPB) and anesthesia management were performed according to our standard operating procedures [25]. Normothermic CPB was established with a non-pulsatile flow of 2.5 l min−1 m2 and a mean arterial pressure >60 mmHg. Cardioplegic arrest was induced and maintained by intermittent administration of antegrade warm potassium enriched blood [26]. After chest closure, the patient was transferred intubated and mechanically ventilated (pressure-controlled mode) to ICU. Patients were kept sedated with propofol (1–3 mg kg−1 h−1) and opioid bolus administration until cardiopulmonary stability was achieved, chest tube drainage was insignificant (<100 ml/h), and the patient was judged to be extubated. If mechanical ventilation was required for ≥12 h, analgesic sedation was switched to midazolam 0.01–0.2 mg kg−1 h−1 combined with sufentanil 0.15–0.7 µg kg−1 h−1, and weaning from mechanical ventilation was performed according to the standard operating procedures at our hospital [27]. Further therapy was based on actual medical standards.
Results are expressed as mean ± standard deviation (SD), median ± interquartile range (IQR) or percentage, as appropriate. Normality of continuous variables was checked with the Kolmogorov–Smirnov test. The exact Mann–Whitney U-test was used to analyze differences between groups. The relative frequencies of variables were analyzed by the exact Chi square test. Non-inferiority of guiding norepinephrine therapy was based on the on the used cumulative doses of norepinephrine in the first week after extending hemodynamic monitoring using hTEE or transpulmonary thermodilution and was tested by means of the one-sided Schuirmann-test with an inferiority margin of “0.3 × mean (control)”. p < 0.05 was considered statistically significant. All tests should be understood as constituting exploratory data analysis, such that no adjustments for multiple tests have been made. Statistics were performed using SPSS 20.0 software (IBM Corporation, Armonk, New York, USA) and EquivTest 2.0 Copyright© 2001 Statistical Solutions Ltd. Cork Ireland.
Results
18 patients were eligible according to the aforementioned criteria during the study period and were thus monitored with hTEE (hTEE group). These patients were retrospectively case-controlled matched based on their SAPS-II admission score on ICU (score ± 5) to 18 patients monitored with transpulmonary thermodilution before hTEE implementation (control group).
Basic patient characteristics and surgical data are presented in Table 1. There were no significant differences in preoperative functional status, e.g. NYHA classification, surgical procedures, or other comorbidities between groups. Concerning the nature of valve surgery (VR) patients with reconstructive, replacement or redo of the aortic valve due to aortic stenosis and/or insufficiency and/or endocarditis were included.
ICU residents at the first attempt in all patients blindly inserted the hTEE probe. Image quality was judged to be adequate to guide hemodynamic therapy or better in all patients (adequate: n = 11, 61 %; optimal: n = 7, 39 %). Image quality was never inadequate and we never had to proceed to a conventional multiplane TEE based on hTEE findings. In 4 patients (22 %) only 2 of the 3 views could be obtained (no TG SAX: n = 3, no SVC: n = 1), however, these missing views were not assessed to have any further influence on clinical decision-making. Therefore all studies were rated to be complete. In addition, supervisors were also not able to obtain these missing views and their interpretations were equal with those of the residents. 30 ± 20 hTEE loops were recorded per patient with a mean duration of probe insertion of 26.2 ± 17.6 h. This resulted in approximately ten complete hTEE examinations per patient, one every 2.6 h. In the control group, extravascular lung water index (EVLWI) as a surrogate parameter for the frequency of transpulmonary thermodilutions was determined an average of 9 times per patients in a 3 day period resulting in approximately one complete examination every 8 h.
Results of the hTEE examination changed the current therapy in nearly all cases (n = 16, 89 %): (1) administration of levosimendan due to a severely reduced left ventricular function (n = 10, 56 %), (2) supporting right ventricular function, i.e. administration of iloprost and/or PDE-III-inhibitors and/or levosimendan, due to new/unexpected postoperative right heart failure (n = 5, 28 %) (Fig. 2) (supplementary videos), and (3) re-operation due to pericardial tamponade (n = 1, 6 %). The first documented transpulmonary thermodilution led to a change in current therapy in 44.4 % (n = 8): (a) volume administration (n = 6) and (b) adding PDE-III-inhibitor (n = 2). In addition, interpretation of the first transpulmonary thermodilution parameters never triggered administration of levosimendan (Fig. 3).
7 patients in the control group and only 3 patients in the hTEE group died in hospital, but the difference did not reach statistical significance (p = 0.264). Data on the postoperative management in survivors (hTEE: n = 15; control: n = 11) are presented in Table 2. Maximum arterial lactate level and its decline in the observed postoperative course were comparable in both groups. Net fluid balance in the first three postoperative days was not significantly different between groups. However, the cumulative dose of epinephrine in the first week after extending hemodynamic monitoring was significantly reduced (p = 0.034) along with a significantly increased frequency of levosimendan administration (p = 0.047) in the hTEE group. There was a non-significant trend towards a lower daily cumulative dose of enoximone in the hTEE group (p = 0.051). Finally, hTEE was non-inferior to the control group in guiding norepinephrine treatment (p = 0.038).
Discussion
The main findings of this retrospective, single center analysis were that postoperative hTEE monitoring after cardiac surgery is feasible and immediately influences hemodynamic therapy and patient management.
After a short and intensive training programme, residents of our ICU were able to achieve adequate or optimal quality of obtainable images in all cases comparable to a previous study [17]. In 22 % of the patients only two of the three hTEE views could be acquired. However, in face of hemodynamic guidance the missing views were not rated to have any further influence on clinical decision-making. This is in line with a previous prospective trial in 94 ventilated critically ill patients where image quality allowed hemodynamic monitoring in 85 % [16]. Furthermore, the technical limitations of the small, monoplane hTEE probe were the main cause for an overall lower image quality image quality compared to a conventional multiplane probe in this retrospective analysis [16]. This may be caused by (a) the small, more flexible probe establishing too less surface contact area and (b) the inability to rotate the probe resulting in suboptimal cross-sections. In particular, at times the lateral wall of the left ventricle was poorly visualized in the midesophageal four chamber view, but always optimal in the transgastric short axis view. However, image quality was never inadequate for guiding hemodynamic optimization. Indeed, residents of our department felt quickly familiar with the probe focusing on only three views and the possibility to frequently repeat the examination. Nevertheless, owing to these technical limitations, a comprehensive echocardiographic evaluation cannot be conducted with the ClariTEE® probe. Therefore, in case of unexpected findings a multiplane TEE has to be performed according to our institutional algorithm (Fig. 2: “Seek expert help”). This was never the case in this study. In addition, we did not observe any harmful event related to probe insertion in this retrospective analysis [28]. Especially, we saw no thermal injury since by design, imaging is “frozen” (the system is off) and pressure is removed (no flexion) between two examinations (mean time interval 2.6 h).
As early as 1998 Benjamin et al. [29] demonstrated that an additional multiplane transesophageal echocardiography performed by intensivists changed pre-exam hemodynamic therapy based on pulmonary artery catheter findings in >50 % of patients. This was especially true in the case of reduced left ventricular contractility. In the present study, interpretation of hTEE findings after probe insertion provoked changes of the current therapy in 89 % of patients. Observers mostly reported echocardiographic findings consistent with refractory left and/or right ventricular failure due to chronic heart insufficiency, leading them to administer levosimendan as ultima ratio add-on medication [30]. This resulted in a significant reduction in the cumulative dose of epinephrine. In addition, the cumulative dose of enoximone was non-significantly reduced; however, this may be explained in part because we do not administer levosimendan and enoximone simultaneously in our ICU, based on their pharmacodynamic and -kinetic profiles [31]. Furthermore, we did not observe an increase in the frequency and dose of norepinephrine in the hTEE group despite vasodilating properties of levosimendan [32]. This is consistent with a previous study demonstrating that levosimendan treatment did not lead to an increase in norepinephrine when goal-directed volume therapy is applied at the same time [30].
Interestingly, hTEE monitoring detected new and/or clinically unexpected right ventricular (RV) dysfunction [19, 33, 34] in approximately one-third of the patients, comparable to a previous study [14]. Fletcher et al. [19] also detected even more new right ventricular failure (67.6 %) in ventilated patients with severe cardiogenic shock requiring hemodynamic support using hTEE. Also, one pericardial tamponade by monoplane hTEE was detected in a very early postoperative time frame with consequent re-operation [35–39]. In accord with the mean duration of hTEE probe insertion [17], our results emphasize the role of cardiac visualization early in the postoperative course in order to detect (new) cardiac dysfunction, i.e. especially RV failure, optimize hemodynamics and improve patients management [11, 16]. In this context, it is worth noticing that we observed a non-significant trend towards improved in-hospital mortality in the hTEE group. We therefore conclude that the implementation of hTEE monitoring into clinical practice following an approximately 6-h teaching session demonstrates that a focused echocardiographic approach is feasible and sufficient to enable physicians optimizing hemodynamics [40–43].
We did not detect a difference in the amount of volume that has been administered between both groups. It may be that hTEE monitoring only helps in detecting the right time point when to administer volume, especially when atrial fibrillation [8, 44–46] and/or right heart dysfunction are present. In this context it has been shown that concomitant tricuspid regurgitation is associated with underestimation of cardiac output measured by thermodilution [47]. In addition, clinically used parameters to estimate volume responsiveness seem to be unreliable in patients with right heart dysfunction [48, 49]. Finally, hTEE monitoring was non-inferior to standard patient care in guiding norepinephrine treatment.
This study stands out for its clinical approach, and as such it has some limitations. As with all retrospective studies, our analysis was limited by the variables available in routine patient care. First, the number of patients analyzed in this study may also be regarded as relatively small. In addition, EVLWI as a surrogate for the frequency of transpulmonary thermodilutions might be inadequate in patients with sinus rhythm and continuous pulse contour analysis. However, there may exist some clinical scenarios in which testing for validity of continuous pulse contour analysis is still missing up to date. In this context it is important to mention that ≥50 % of the patients of both groups presented with atrial fibrillation (AF) preoperatively or developed AF postoperatively. These findings represent our first clinical experience with a new interesting device during its implementation into daily routine. In contrast to conventional hemodynamic monitors, the advantage of hTEE might be the frequent “on demand” visualization of the heart in combination with the here presented hemodynamic optimization algorithm. Secondly, one might assume that patients in the hTEE group were sicker and therefore received significantly more often levosimendan. However, patients´ functional status, all other comorbidities and their clinical status upon arrival on ICU were comparable, and all patients suffered sustained, life-threating hemodynamic instability after cardiac surgery. Here, hTEE findings in postoperative cardiac surgery patients lead to a change in therapy in nearly all cases. Therefore, despite limitations of its case-controlled matched-pairs approach, this study clearly demonstrated the benefit of a focused transesophageal echocardiography early in the postoperative course if hemodynamic instability is present. Thirdly, because assessment of volume responsiveness, the clearance of lactate and the decrease in catecholaminergic support is a compound of several factors and physician expertise, the here presented impact of hTEE monitoring has to be interpreted on that condition. However, there is a need for individualized therapeutic strategies and algorithms based on physiology [50, 51].
In conclusion, this retrospective analysis revealed a beneficial influence of hTEE monitoring on the postoperative management of cardiac surgery patients. Despite a short training session, ICU residents achieved at least adequate image quality to guide hemodynamic therapy. hTEE monitoring is clinically feasible and can simply be implemented into daily routine. It is not inferior to standard patient care in guiding norepinephrine therapy. Besides one urgent re-operation due to pericardial tamponade, hTEE monitoring seemed to influence hemodynamic management in patients with postoperative low cardiac output syndrome. Finally, hTEE monitoring was useful in the diagnosis and management of unexpected postoperative right heart failure. Further prospective, randomized trials are warranted to investigate the capabilities of this new device.
Abbreviations
- GDT:
-
Goal-directed therapy
- DO2 :
-
Delivery of oxygen
- CO:
-
Cardiac output
- LCOS:
-
Low cardiac output syndrome
- hTEE:
-
Hemodynamic monoplane transesophageal echocardiography
- ICU:
-
Intensive care unit
- PICCO® :
-
Pulse contour cardiac output (monitor)
- ESC:
-
European Society of Cardiology
- EACVI:
-
European Association of Cardiovascular Imaging
- EACTA:
-
European Association of Cardiothoracic Anaesthesiologists
- TG SAX:
-
Transgastric short axis (view)
- 4C:
-
Midesophageal four chamber (view)
- SVC:
-
Superior vena cava (view)
- LV:
-
Left ventricle
- RV:
-
Right ventricle
- IAS:
-
Interatriale septum
- TEE:
-
Multiplane transesophageal echocardiography
- SD:
-
Standard deviation
- IQR:
-
Interquartiles
- SAPS-II:
-
Simplified acute physiology score II
- NYHA:
-
New York Heart Association
- CPB:
-
Cardiopulmonary bypass
- CABG:
-
Coronary bypass grafting
- VR:
-
Valve reconstruction and/or replacement
- EVLWI:
-
Extravascular lung water index
References
Pearse R, Dawson D, Fawcett J, Rhodes A, Grounds RM et al (2005) Early goal-directed therapy after major surgery reduces complications and duration of hospital stay. A randomised, controlled trial [ISRCTN38797445]. Crit Care 9:R687–R693
Hamilton MA, Cecconi M, Rhodes A (2011) A systematic review and meta-analysis on the use of preemptive hemodynamic intervention to improve postoperative outcomes in moderate and high-risk surgical patients. Anesth Analg 112:1392–1402
Gan TJ, Soppitt A, Maroof M, el-Moalem H, Robertson KM et al (2002) Goal-directed intraoperative fluid administration reduces length of hospital stay after major surgery. Anesthesiology 97:820–826
Grocott MPW, Mythen MG, Gan TJ (2005) Perioperative fluid management and clinical outcomes in adults. Anesth Analg 100:1093–1106
Rivers E, Nguyen B, Havstad S, Ressler J, Muzzin A et al (2001) Early goal directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med 345:1368–1377
Futier E, Constantin JM, Petit A, Chanques G, Kwiatkowski F et al (2010) Conservative vs restrictive individualized goal-directed fluid replacement strategy in major abdominal surgery: a prospective randomized trial. Arch Surg 145:1193–1200
Della Rocca G, Pompei L (2011) Goal-directed therapy in anesthesia: any clinical impact or just a fashion? Minerva Anestesiol 77:545–553
McGee WT, Raghunathan K (2013) Physiologic goal-directed therapy in the perioperative period: the volume prescription for high-risk patients. J Cardiothorac Vasc Anesth 27:1079–1086
Practice guidelines for perioperative transesophageal echocardiography (2010) An updated report by the American Society of Anesthesiologists and the Society of Cardiovascular Anesthesiologists Task Force on Transesophageal Echocardiograph. Anesthesiology 112:1084–1096
Giglio M, Dalfino L, Puntillo F, Rubino G, Marucci M et al (2012) Haemodynamic goal-directed therapy in cardiac and vascular surgery. A systematic review and meta-analysis. Interact CardioVasc Thorac Surg 15:878–887
Aya HD, Cecconi M, Hamilton M, Rhodes A (2013) Goal-directed therapy in cardiac surgery: a systematic review and meta-analysis. Br J Anaesth 110:510–517
Geisen M, Spray D, Fletcher N (2014) Echocardiography-based hemodynamic management in the cardiac surgical intensive care unit. J Cardiothorac Vasc Anesth 28:733–744
Wagner CE, Bick JS, Webster BH, Selby JH, Byrne JG (2012) Use of a miniaturized transesophageal echocardiographic probe in the intensive care unit for diagnosis and treatment of a hemodynamically unstable patient after aortic valve replacement. J Cardiothorac Vasc Anesth 26:95–97
Maltais S, Costello WT, Billings FTt, Bick JS, Byrne JG et al (2013) Episodic monoplane transesophageal echocardiography impacts postoperative management of the cardiac surgery patient. J Cardiothorac Vasc Anesth 27:665–669
Haglund NA, Maltais S, Bick JS, Costello W, Keebler M et al (2014) Hemodynamic transesophageal echocardiography after left ventricular assist device implantation. J Cardiothorac Vasc Anesth 28:1184–1190
Vieillard-Baron A, Slama M, Mayo P, Charron C, Amiel JB et al (2013) A pilot study on safety and clinical utility of a single-use 72-hour indwelling transesophageal echocardiography probe. Intensive Care Med 39:629–635
Cioccari L, Baur HR, Berger D, Wiegand J, Takala J et al (2013) Hemodynamic assessment of critically ill patients using a miniaturized transesophageal echocardiography probe. Crit Care 17:R121
Cavarocchi NC, Pitcher HT, Yang Q, Karbowski P, Miessau J et al (2013) Weaning of extracorporeal membrane oxygenation using continuous hemodynamic transesophageal echocardiography. J Thorac Cardiovasc Surg 146:1474–1479
Fletcher N, Geisen M, Meeran H, Spray D, Cecconi M (2015) Initial clinical experience with a miniaturized transesophageal echocardiography probe in a cardiac intensive care unit. J Cardiothorac Vasc Anesth. doi:10.1053/j.jvca.2014.09.016
Carl M, Alms A, Braun J, Dongas A, Erb J et al (2010) S3 guidelines for intensive care in cardiac surgery patients: hemodynamic monitoring and cardiocirculary system. Ger Med Sci 8. doi:10.3205/000101
Cheitlin MD, Armstrong WF, Aurigemma GP, Beller GA, Bierman FZ et al (2003) ACC/AHA/ASE 2003 Guideline Update for the Clinical Application of Echocardiography: summary article. A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (ACC/AHA/ASE Committee to Update the 1997 Guidelines for the Clinical Application of Echocardiography). J Am Soc Echocardiogr 16:1091–1110
Kukucka M, Stepanenko A, Potapov E, Krabatsch T, Redlin M et al (2011) Right-to-left ventricular end-diastolic diameter ratio and prediction of right ventricular failure with continuous-flow left ventricular assist devices. J Heart Lung Transplant 30:64–69
Kusumoto FM, Muhiudeen IA, Kuecherer HF, Cahalan MK, Schiller NB (1993) Response of the interatrial septum to transatrial pressure gradients and its potential for predicting pulmonary capillary wedge pressure: an intraoperative study using transesophageal echocardiography in patients during mechanical ventilation. J Am Coll Cardiol 21:721–728
Vieillard-Baron A, Chergui K, Rabiller A, Peyrouset O, Page B et al (2004) Superior vena caval collapsibility as a gauge of volume status in ventilated septic patients. Intensive Care Med 30:1734–1739
Spies C, Kastrup M, Kerner T, Melzer-Gartzke C, Zielke H, Kox W (2013) SOPs in Anästhesiologie und Schmerztherapie: Alle relevanten Standards und Techniken für die Klinik. 1 ed. Thieme
Calafiore AM, Teodori G, Di Giammarco G, Bosco G, Mezzetti A et al (1994) Intermittent antegrade cardioplegia: warm blood vs cold crystalloid. A clinical study. J Cardiovasc Surg 35:179–184
Spies C, Kastrup M, Kerner T, Melzer-Gartzke C, Zielke H, Kox W (2013) SOPs in Intensivmedizin und Notfallmedizin: Alle relevanten Standards und Techniken für die KLinik, 1 ed. Thieme
Hilberath JN, Oakes DA, Shernan SK, Bulwer BE, D’Ambra MN et al (2010) Safety of transesophageal echocardiography. J Am Soc Echocardiogr 23:1115–1127
Benjamin E, Griffin K, Leibowitz AB, Manasia A, Oropello JM et al (1998) Goal-directed transesophageal echocardiography performed by intensivists to assess left ventricular function: comparison with pulmonary artery catheterization. J Cardiothorac Vasc Anesth 12:10–15
Erb J, Beutlhauser T, Feldheiser A, Schuster B, Treskatsch S et al (2014) Influence of levosimendan on organ dysfunction in patients with severely reduced left ventricular function undergoing cardiac surgery. J Int Med Res 42:750–764
Nieminen MS, Fruhwald S, Heunks LM, Suominen PK, Gordon AC et al (2013) Levosimendan: current data, clinical use and future development. Heart Lung Vessel 5:227–245
Toller WG, Stranz C (2006) Levosimendan, a new inotropic and vasodilator agent. Anesthesiology 104:556–569
Haddad F, Couture P, Tousignant C, Denault AY (2009) The right ventricle in cardiac surgery, a perioperative perspective: II. Pathophysiology, clinical importance, and management. Anesth Analg 108:422–433
Vlahakes GJ (2005) Right ventricular failure following cardiac surgery. Coron Artery Dis 16:27–30
Imren Y, Tasoglu I, Oktar GL, Benson A, Naseem T et al (2008) The importance of transesophageal echocardiography in diagnosis of pericardial tamponade after cardiac surgery. J Card Surg 23:450–453
Kuvin JT, Harati NA, Pandian NG, Bojar RM, Khabbaz KR (2002) Postoperative cardiac tamponade in the modern surgical era. Ann Thorac Surg 74:1148–1153
Bommer WJ, Follette D, Pollock M, Arena F, Bognar M et al (1995) Tamponade in patients undergoing cardiac surgery: a clinical-echocardiographic diagnosis. Am Heart J 130:1216–1223
Weitzman LB, Tinker WP, Kronzon I, Cohen ML, Glassman E et al (1984) The incidence and natural history of pericardial effusion after cardiac surgery—an echocardiographic study. Circulation 69:506–511
Hirose H, GuptaS Pitcher H, Miessau J, Yang Q et al (2014) Feasibility of diagnosis of postcardiotomy tamponade by miniaturized transesophageal echocardiography. J Surg Res 190:27276–27279
Cholley BP, Vieillard-Baron A, Mebazaa nA (2006) Echocardiography in the ICU: time for widespread use! Intensive Care Med 32:9–10
Manasia AR, Nagaraj HM, Kodali RB, Croft LB, Oropello JM et al (2005) Feasibility and potential clinical utility of goal-directed transthoracic echocardiography performed by noncardiologist intensivists using a small hand-carried device (SonoHeart) in critically ill patients. J Cardiothorac Vasc Anesth 19:155–159
Jensen MB, Sloth E, Larsen KM, Schmidt MB (2004) Transthoracic echocardiography for cardiopulmonary monitoring in intensive care. Eur J Anaesthesiol 21:700–707
Kaul S, Stratienko AA, Pollock SG, Marieb MA, Keller MW et al (1994) Value of two-dimensional echocardiography for determining the basis of hemodynamic compromise in critically ill patients: a prospective study. J Am Soc Echocardiogr 7:598–606
Wittkowski U, Spies C, Sander M, Erb J, Feldheiser A et al (2009) Haemodynamic monitoring in the perioperative phase. Available systems, practical application and clinical data. Der Anaesthesist 58(764–778):780–786
Habicher M, Perrino A Jr, Spies CD, von Heymann C, Wittkowski U et al (2011) Contemporary fluid management in cardiac anesthesia. J Cardiothorac Vasc Anesth 25:1141–1153
Ostergaard M, Nilsson LB, Nilsson JC, Rasmussen JP, Berthelsen PG (2005) Precision of bolus thermodilution cardiac output measurements in patients with atrial fibrillation. Acta Anaesthesiol Scand 49:366–372
Balik M, Pachl J, Hendl J (2002) Effect of the degree of tricuspid regurgitation on cardiac output measurements by thermodilution. Intensive Care Med 28:1117–1121
Wyler von Ballmoos M, Takala J, Roeck M, Porta F, Tueller D et al (2010) Pulse-pressure variation and hemodynamic response in patients with elevated pulmonary artery pressure: a clinical study. Crit Care 14:R111
Ranucci M, Pazzaglia A, Tritapepe L, Guarracino F, Lupo M et al (2009) Fluid responsiveness and right ventricular function in cardiac surgical patients. A multicenter study. HSR Proc Intensive Care Cardiovasc Anesth 1:21–29
Zhang Z, Ni H, Qian Z (2015) Effectiveness of treatment based on PiCCO parameters in critically ill patients with septic shock and/or acute respiratory distress syndrome: a randomized controlled trial. Intensive Care Med 41:444–451
Boulain T, Cecconi M (2015) Can one size fit all? The fine line between fluid overload and hypovolemia. Intensive Care Med 41:544–546
Acknowledgments
This study was performed within an institutional Grant from Charité – Universitätsmedizin Berlin. ImaCor provided the echocardiographic system (Zura, Imacor, New York, USA) and the ClariTEE® probes without charge for this study, the company provided no further financial support.
Conflict of interest
S.T. received funding for experimental research from B. Braun and honoraria for lectures from Edwards and Carinopharm. C.S. received (a) funding for research from the following companies and societies: Abbott, Aspect, Baxter, Care Fusion, Deltex, Fresenius, Grünenthal, Hutchinson, Köhler Chemie, MSD, MCN, Novartis, Pajunk, Pulsion, Roche, Sysmex, University Hospital Stavanger, Argus, BDA, BMBF, DKH, DLR, German Research Society, GIZ, Charité, Stifterverband, and (b) honoraria for lectures and travel/accommodations/meeting expenses unrelated to activities listed from Abbott, Essex Pharma, GSK, Bispebjerg Hospital, Aspect, and (c) expert testimony from the ethical committee Vienna Faculty of Medicine. J.P.B. received honoraria for lectures from Edwards, Carinopharm, Abbott, Orion and Pfizer. M.S. received funding for research from Edwards Life Sciences, The Medicines Company and Pulsion, honoraria for lectures from Edwards and Pulsion. For the remaining authors none conflict of interests were declared.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Treskatsch, S., Balzer, F., Knebel, F. et al. Feasibility and influence of hTEE monitoring on postoperative management in cardiac surgery patients. Int J Cardiovasc Imaging 31, 1327–1335 (2015). https://doi.org/10.1007/s10554-015-0689-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-015-0689-8