Abstract
Purpose
Cutoffs of the 21-gene recurrence score (RS), a commonly used genomic assay for hormone receptor-positive breast cancer, have been updated. Little is known about racial/ethnic differences in RS results, RS-guided chemotherapy use, and outcomes on updated cutoff (RS ≥ 31 defined as high-risk) in the real-world setting.
Methods
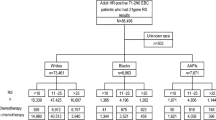
A total of 81,937 women [75.0% whites, 7.7% blacks, 8.3% Asian American/Pacific Islanders (AAPIs), and 9.0% Hispanics] diagnosed with hormone receptor-positive breast cancer between 2004 and 2015, who received the 21-gene assay, were identified from the Surveillance, Epidemiology, and End Results. Logistic regressions estimated the race-associated odds ratios (ORs) of RS and chemotherapy use. Cox regressions estimated the race-associated hazard ratios (HRs) of breast cancer-specific and all-cause mortality.
Results
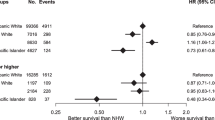
Compared with white women, black women were more likely to have RS-defined high-risk tumors (adjusted OR [aOR] 1.29; 95% CI 1.16–1.42). In high RS, blacks had lower odds of chemotherapy use (aOR 0.76; 95% CI 0.62–0.94) than whites, particularly among women ≥ 65 years (aOR 0.51; 95% CI 0.35–0.76), while AAPI and Hispanic women had no variation in chemotherapy use compared with whites in high RS. Black women had a higher risk of breast cancer-specific mortality (HR 1.37; 95% CI 1.12–1.67) and all-cause mortality compared with white women after adjusting for demographic and pathological factors, county-level socioeconomic deprivation, treatments and RS; AAPIs had lower mortality and Hispanics had similar mortality.
Conclusions
Black women were more likely to have a high-risk RS tumor and less likely to receive chemotherapy in the group of high RS, especially those ≥ 65 years. Further studies are needed to identify barriers to chemotherapy in black patients with high RS scores.
Similar content being viewed by others
Data availability
The datasets analyzed during the current study are available Dr. Han on reasonable request.
Abbreviations
- AAPI:
-
Asian Americans/Pacific Islander
- CI:
-
Confidence interval
- HER2:
-
Human epidermal growth factor receptor 2
- HR:
-
Hazard ratio
- NCCN:
-
National Comprehensive Cancer Network
- OR:
-
Odds ratio
- RS:
-
Recurrence score
- RUCC:
-
Rural–urban continuum codes
- SEER:
-
Surveillance, epidemiology, and end results
- TAILORx:
-
Trial assigning individualized options for treatment
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6):394–424. https://doi.org/10.3322/caac.21492
American Cancer Society (2017) Breast cancer facts & figures 2017–2018. American Cancer Society Inc, Atlanta
Howlader N, Altekruse SF, Li CI, Chen VW, Clarke CA, Ries LA, Cronin KA (2014) US incidence of breast cancer subtypes defined by joint hormone receptor and HER2 status. J Natl Cancer Inst. https://doi.org/10.1093/jnci/dju055
Warner ET, Tamimi RM, Hughes ME, Ottesen RA, Wong YN, Edge SB, Theriault RL, Blayney DW, Niland JC, Winer EP, Weeks JC, Partridge AH (2015) Racial and ethnic differences in breast cancer survival: mediating effect of tumor characteristics and sociodemographic and treatment factors. J Clin Oncol 33(20):2254–2261. https://doi.org/10.1200/jco.2014.57.1349
O'Brien KM, Cole SR, Tse CK, Perou CM, Carey LA, Foulkes WD, Dressler LG, Geradts J, Millikan RC (2010) Intrinsic breast tumor subtypes, race, and long-term survival in the Carolina Breast Cancer Study. Clin Cancer Res 16(24):6100–6110. https://doi.org/10.1158/1078-0432.ccr-10-1533
Anderson WF, Rosenberg PS, Menashe I, Mitani A, Pfeiffer RM (2008) Age-related crossover in breast cancer incidence rates between black and white ethnic groups. J Natl Cancer Inst 100(24):1804–1814. https://doi.org/10.1093/jnci/djn411
Hershman DL, Unger JM, Barlow WE, Hutchins LF, Martino S, Osborne CK, Livingston RB, Albain KS (2009) Treatment quality and outcomes of African American versus white breast cancer patients: retrospective analysis of Southwest Oncology studies S8814/S8897. J Clin Oncol 27(13):2157–2162. https://doi.org/10.1200/jco.2008.19.1163
Bickell NA, Wang JJ, Oluwole S, Schrag D, Godfrey H, Hiotis K, Mendez J, Guth AA (2006) Missed opportunities: racial disparities in adjuvant breast cancer treatment. J Clin Oncol 24(9):1357–1362. https://doi.org/10.1200/jco.2005.04.5799
Menashe I, Anderson WF, Jatoi I, Rosenberg PS (2009) Underlying causes of the black-white racial disparity in breast cancer mortality: a population-based analysis. J Natl Cancer Inst 101(14):993–1000. https://doi.org/10.1093/jnci/djp176
Troester MA, Sun X, Allott EH, Geradts J, Cohen SM, Tse CK, Kirk EL, Thorne LB, Mathews M, Li Y, Hu Z, Robinson WR, Hoadley KA, Olopade OI, Reeder-Hayes KE, Earp HS, Olshan AF, Carey LA, Perou CM (2018) Racial differences in PAM50 subtypes in the Carolina Breast Cancer Study. J Natl Cancer Inst. https://doi.org/10.1093/jnci/djx135
Petkov VI, Miller DP, Howlader N, Gliner N, Howe W, Schussler N, Cronin K, Baehner FL, Cress R, Deapen D, Glaser SL, Hernandez BY, Lynch CF, Mueller L, Schwartz AG, Schwartz SM, Stroup A, Sweeney C, Tucker TC, Ward KC, Wiggins C, Wu XC, Penberthy L, Shak S (2016) Breast-cancer-specific mortality in patients treated based on the 21-gene assay: a SEER population-based study. NPJ Breast Cancer 2:16017. https://doi.org/10.1038/npjbcancer.2016.17
Paik S, Shak S, Tang G, Kim C, Baker J, Cronin M, Baehner FL, Walker MG, Watson D, Park T, Hiller W, Fisher ER, Wickerham DL, Bryant J, Wolmark N (2004) A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N Engl J Med 351(27):2817–2826. https://doi.org/10.1056/NEJMoa041588
Kwa M, Makris A, Esteva FJ (2017) Clinical utility of gene-expression signatures in early stage breast cancer. Nat Rev Clin Oncol 14(10):595–610. https://doi.org/10.1038/nrclinonc.2017.74
Geyer CE Jr, Tang G, Mamounas EP, Rastogi P, Paik S, Shak S, Baehner FL, Crager M, Wickerham DL, Costantino JP, Wolmark N (2018) 21-Gene assay as predictor of chemotherapy benefit in HER2-negative breast cancer. NPJ Breast Cancer 4:37. https://doi.org/10.1038/s41523-018-0090-6
Mamounas EP, Russell CA, Lau A, Turner MP, Albain KS (2018) Clinical relevance of the 21-gene recurrence score((R)) assay in treatment decisions for patients with node-positive breast cancer in the genomic era. NPJ Breast Cancer 4:27. https://doi.org/10.1038/s41523-018-0082-6
Paik S, Tang G, Shak S, Kim C, Baker J, Kim W, Cronin M, Baehner FL, Watson D, Bryant J, Costantino JP, Geyer CE Jr, Wickerham DL, Wolmark N (2006) Gene expression and benefit of chemotherapy in women with node-negative, estrogen receptor-positive breast cancer. J Clin Oncol 24(23):3726–3734. https://doi.org/10.1200/jco.2005.04.7985
Dowsett M, Cuzick J, Wale C, Forbes J, Mallon EA, Salter J, Quinn E, Dunbier A, Baum M, Buzdar A, Howell A, Bugarini R, Baehner FL, Shak S (2010) Prediction of risk of distant recurrence using the 21-gene recurrence score in node-negative and node-positive postmenopausal patients with breast cancer treated with anastrozole or tamoxifen: a TransATAC study. J Clin Oncol 28(11):1829–1834. https://doi.org/10.1200/jco.2009.24.4798
Albain KS, Barlow WE, Shak S, Hortobagyi GN, Livingston RB, Yeh IT, Ravdin P, Bugarini R, Baehner FL, Davidson NE, Sledge GW, Winer EP, Hudis C, Ingle JN, Perez EA, Pritchard KI, Shepherd L, Gralow JR, Yoshizawa C, Allred DC, Osborne CK, Hayes DF (2010) Prognostic and predictive value of the 21-gene recurrence score assay in postmenopausal women with node-positive, oestrogen-receptor-positive breast cancer on chemotherapy: a retrospective analysis of a randomised trial. Lancet Oncol 11(1):55–65. https://doi.org/10.1016/s1470-2045(09)70314-6
Paik S (2007) Development and clinical utility of a 21-gene recurrence score prognostic assay in patients with early breast cancer treated with tamoxifen. Oncologist 12(6):631–635. https://doi.org/10.1634/theoncologist.12-6-631
Massarweh SA, Sledge GW, Miller DP, McCullough D, Petkov VI, Shak S (2018) Molecular characterization and mortality from breast cancer in men. J Clin Oncol 36(14):1396–1404. https://doi.org/10.1200/jco.2017.76.8861
National Comprehensive Cancer Network (2019) Breast cancer (version 1. 2019). National Comprehensive Cancer Network, Plymouth Meeting
Sparano JA, Gray RJ, Makower DF, Pritchard KI, Albain KS, Hayes DF, Geyer CE Jr, Dees EC, Goetz MP, Olson JA Jr, Lively T, Badve SS, Saphner TJ, Wagner LI, Whelan TJ, Ellis MJ, Paik S, Wood WC, Ravdin PM, Keane MM, Gomez Moreno HL, Reddy PS, Goggins TF, Mayer IA, Brufsky AM, Toppmeyer DL, Kaklamani VG, Berenberg JL, Abrams J, Sledge GW Jr (2018) Adjuvant chemotherapy guided by a 21-gene expression assay in breast cancer. N Engl J Med 379(2):111–121. https://doi.org/10.1056/NEJMoa1804710
Albain K, Gray R, Sparano J, Makower D, Pritchard K, Hayes D, Geyer C, Dees E, Goetz M, Olson J, Lively T, Badve S, Saphner T, Wagner L, Whelan T, Ellis M, Paik S, Wood W, Ravdin P, Keane M, Gomez H, Reddy P, Goggins T, Mayer I, Brufsky A, Toppmeyer D, Kaklamani V, Berenberg J, Abrams J, Sledge G (2019) Abstract GS4–07: Race, ethnicity and clinical outcomes in hormone receptor-positive, HER2-negative, node-negative breast cancer: results from the TAILORx trial. Cancer Res. https://doi.org/10.1158/1538-7445.sabcs18-gs4-07
National Cancer Institute (2019) Surveillance, epidemiology, and end results program. SEER incidence data, 1975–2016. National Cancer Institute, Bethesda
National Cancer Institute. Surveillance, Epidemiology, and End Results program.Oncotype DX Database (2004–2015).
National Cancer Institute (2019) Surveillance, epidemiology, and end results program. Rural-urban continuum codes. https://seer.cancer.gov/seerstat/variables/countyattribs/ruralurban.html. Accessed 12 June 2019
Lian M, Struthers J, Liu Y (2016) Statistical assessment of neighborhood socioeconomic deprivation environment in spatial epidemiologic studies. Open J Stat 6:436–442
Lian M, Perez M, Liu Y et al (2014) Neighborhood socioeconomic deprivation, tumor subtypes, and causes of death after non-metastatic invasive breast cancer diagnosis: a multilevel competing-risk analysis. Breast Cancer Res Treat 147:661–670
Zhang S, Liu Y, Yun S et al (2018) Impacts of neighborhood characteristics on treatment and outcomes in women with ductal carcinoma in situ of the breast. Cancer Epidemiol Biomark Prev 27:1298–1306
Lin DY, Wei LJ, Ying Z (1993) Checking the Cox model with cumulative sums of martingale-based residuals. Biometrika 80(3):557–572. https://doi.org/10.1093/biomet/80.3.557
Holowatyj AN, Cote ML, Ruterbusch JJ, Ghanem K, Schwartz AG, Vigneau FD, Gorski DH, Purrington KS (2018) Racial differences in 21-gene recurrence scores among patients with hormone receptor-positive, node-negative breast cancer. J Clin Oncol 36(7):652–658. https://doi.org/10.1200/jco.2017.74.5448
Lund MJ, Mosunjac M, Davis KM, Gabram-Mendola S, Rizzo M, Bumpers HL, Hearn S, Zelnak A, Styblo T, O'Regan RM (2012) 21-Gene recurrence scores: racial differences in testing, scores, treatment, and outcome. Cancer 118(3):788–796. https://doi.org/10.1002/cncr.26180
Jasem J, Amini A, Rabinovitch R, Borges VF, Elias A, Fisher CM, Kabos P (2016) 21-gene recurrence score assay as a predictor of adjuvant chemotherapy administration for early-stage breast cancer: an analysis of use, therapeutic implications, and disparity profile. J Clin Oncol 34(17):1995–2002. https://doi.org/10.1200/jco.2015.65.0887
Osborne C, Ostir GV, Du X, Peek MK, Goodwin JS (2005) The influence of marital status on the stage at diagnosis, treatment, and survival of older women with breast cancer. Breast Cancer Res Treat 93(1):41–47. https://doi.org/10.1007/s10549-005-3702-4
Hinyard L, Wirth LS, Clancy JM, Schwartz T (2017) The effect of marital status on breast cancer-related outcomes in women under 65: a SEER database analysis. Breast (Edinburgh, Scotland) 32:13–17. https://doi.org/10.1016/j.breast.2016.12.008
Hsu CD, Wang X, Habif DV Jr, Ma CX, Johnson KJ (2017) Breast cancer stage variation and survival in association with insurance status and sociodemographic factors in US women 18 to 64 years old. Cancer 123(16):3125–3131. https://doi.org/10.1002/cncr.30722
Williams F, Thompson E (2016) Disparity in breast cancer late stage at diagnosis in missouri: does rural versus urban residence matter? J Racial Ethnic Health Disparities 3(2):233–239. https://doi.org/10.1007/s40615-015-0132-9
Nguyen-Pham S, Leung J, McLaughlin D (2014) Disparities in breast cancer stage at diagnosis in urban and rural adult women: a systematic review and meta-analysis. Ann Epidemiol 24(3):228–235. https://doi.org/10.1016/j.annepidem.2013.12.002
Sparano JA, Gray RJ, Makower DF, Pritchard KI, Albain KS, Hayes DF, Geyer CE Jr, Dees EC, Perez EA, Olson JA Jr, Zujewski J, Lively T, Badve SS, Saphner TJ, Wagner LI, Whelan TJ, Ellis MJ, Paik S, Wood WC, Ravdin P, Keane MM, Gomez Moreno HL, Reddy PS, Goggins TF, Mayer IA, Brufsky AM, Toppmeyer DL, Kaklamani VG, Atkins JN, Berenberg JL, Sledge GW (2015) Prospective validation of a 21-gene expression assay in breast cancer. N Engl J Med 373(21):2005–2014. https://doi.org/10.1056/NEJMoa1510764
Mamounas EP, Liu Q, Paik S, Baehner FL, Tang G, Jeong JH, Kim SR, Butler SM, Jamshidian F, Cherbavaz DB, Sing AP, Shak S, Julian TB, Lembersky BC, Wickerham DL, Costantino JP, Wolmark N (2017) 21-gene recurrence score and locoregional recurrence in node-positive/ER-positive breast cancer treated with chemo-endocrine therapy. J Natl Cancer Inst. https://doi.org/10.1093/jnci/djw259
Roberts MC, Weinberger M, Dusetzina SB, Dinan MA, Reeder-Hayes KE, Troester MA, Carey LA, Wheeler SB (2015) Racial variation in adjuvant chemotherapy initiation among breast cancer patients receiving oncotype DX testing. Breast Cancer Res Treat 153(1):191–200. https://doi.org/10.1007/s10549-015-3518-9
Press DJ, Ibraheem A, Dolan ME, Goss KH, Conzen S, Huo D (2018) Racial disparities in omission of oncotype DX but no racial disparities in chemotherapy receipt following completed oncotype DX test results. Breast Cancer Res Treat 168(1):207–220. https://doi.org/10.1007/s10549-017-4587-8
Tammemagi CM, Nerenz D, Neslund-Dudas C, Feldkamp C, Nathanson D (2005) Comorbidity and survival disparities among black and white patients with breast cancer. JAMA 294(14):1765–1772. https://doi.org/10.1001/jama.294.14.1765
Adler NE, Newman K (2002) Socioeconomic disparities in health: pathways and policies. Health Affairs (Project Hope) 21(2):60–76. https://doi.org/10.1377/hlthaff.21.2.60
Buist DS, Chubak J, Prout M, Yood MU, Bosco JL, Thwin SS, Gold HT, Owusu C, Field TS, Quinn VP, Wei F, Silliman RA (2009) Referral, receipt, and completion of chemotherapy in patients with early-stage breast cancer older than 65 years and at high risk of breast cancer recurrence. J Clin Oncol 27(27):4508–4514. https://doi.org/10.1200/jco.2008.18.3459
Sparano JA, Wang M, Zhao F, Stearns V, Martino S, Ligibel JA, Perez EA, Saphner T, Wolff AC, Sledge GW Jr, Wood WC, Davidson NE (2012) Race and hormone receptor-positive breast cancer outcomes in a randomized chemotherapy trial. J Natl Cancer Inst 104(5):406–414. https://doi.org/10.1093/jnci/djr543
Enewold L, Geiger AM, Zujewski J, Harlan LC (2015) Oncotype Dx assay and breast cancer in the United States: usage and concordance with chemotherapy. Breast Cancer Res Treat 151(1):149–156. https://doi.org/10.1007/s10549-015-3366-7
Cress RD, Chen YS, Morris CR, Chew H, Kizer KW (2016) Underutilization of gene expression profiling for early-stage breast cancer in California. Cancer Causes Control CCC 27(6):721–727. https://doi.org/10.1007/s10552-016-0743-4
Farias AJ, Du XL (2017) Racial differences in adjuvant endocrine therapy use and discontinuation in association with mortality among medicare breast cancer patients by receptor status. Cancer Epidemiol Biomark Prev 26(8):1266–1275. https://doi.org/10.1158/1055-9965.epi-17-0280
Farias AJ, Du XL (2017) Association between out-of-pocket costs, race/ethnicity, and adjuvant endocrine therapy adherence among medicare patients with breast cancer. J Clin Oncol 35(1):86–95. https://doi.org/10.1200/jco.2016.68.2807
Funding
Dr. Han is supported by the foundations from Barnes-Jewish Hospital and Breast Cancer Research Foundation. Dr. Peterson is supported by the American Cancer Society. Dr. Colditz is supported by the Breast Cancer Research Foundation and the Siteman Biostatistics Shared Resource P30 CA091842. Drs. Liu and Lian are supported by an American Cancer Society-Denim Days Research Scholar Grant (RSG-18-116-01-CPHPS) and the National Cancer Institute (R01CA215418). The funders had no role in design and conduct of the study, collection, management, analysis and interpretation of the data; preparation, review or approval of the manuscript or decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional research committees.
Informed consent
Informed consent was not required by the institutional research committees.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Han, Y., Miao, ZF., Lian, M. et al. Racial and ethnic disparities in 21-gene recurrence scores, chemotherapy, and survival among women with hormone receptor-positive, node-negative breast cancer. Breast Cancer Res Treat 184, 915–925 (2020). https://doi.org/10.1007/s10549-020-05902-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-020-05902-0