Abstract
Purpose
Cathepsin S (CTSS) is expressed in a variety of cancers and stimulates tumor progression. However, the regulatory mechanism and role of CTSS in breast cancer progression are poorly understood. The aim of this study was to examine the relationships between CTSS expression and breast cancer grade and stage, and the signaling molecules involved in CTSS expression.
Methods
Immunohistochemical staining was performed in tissue microarray sections of 1451 human invasive breast cancer samples to determine epithelial (E-CTSS) and stromal CTSS (S-CTSS) expression. Gene and protein expression levels in human breast cancer cell lines were measured by polymerase chain reaction and western blotting. Small interfering RNA transfection and a Matrigel transwell invasion assay were used to confirm the signaling pathways regulating CTSS expression.
Results
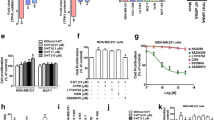
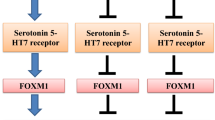
In patient tumor tissue blocks, high grade, late stage, and triple negativity were associated with elevated CTSS protein expression, and expression levels were related to the clinical outcomes of patients with invasive breast cancer. CTSS expression was also higher in triple-negative breast cancer (TNBC) cell lines than in hormone-responsive cells, and CTSS expression patterns matched those of tryptophan hydroxylase 1 (TPH1) and 5-hydroxytryptamine receptor 7 (5-HT7). Treatment of TNBC cells (MDA-MB-231 and HCC-1395) with 5-HT significantly enhanced CTSS protein expression, whereas pharmacological inhibition or knockdown of 5-HT7 significantly inhibited its expression. Correspondingly, cancer cell invasion was increased by 5-HT treatment and suppressed by 5-HT7 knockdown. The expression of CTSS was regulated by PI3K/Akt and Ras/Raf/MAPK signaling pathways, and these signaling pathways were stabilized by HSP90 and enhanced by the 5-HT7 receptor-dependent autocrine effect of 5-HT in TNBC cells.
Conclusion
Our findings suggest CTSS as a candidate target for development of a strategy to inhibit breast cancer invasion, and indicate that HSP90 and 5-HT7 (regulators of CTSS) should be considered as alternative targets for the management of TNBC invasion and metastasis.






Similar content being viewed by others
Abbreviations
- 5-HT:
-
5-Hydroxytryptamine
- 5-HT7 :
-
5-Hydroxytryptamine receptor 7
- AP-1:
-
Activator protein 1
- CTSS:
-
Cathepsin S
- E-CTSS:
-
Epithelial cathepsin S
- ER:
-
Estrogen receptor
- ERK:
-
Extracellular signal-regulated kinase
- FBS:
-
Fetal bovine serum
- GA:
-
Geldanamycin
- GFR:
-
Growth factor receptor
- HSP90:
-
Heat shock protein 90
- IKKβ:
-
Inhibitor kappa-B kinase beta
- MAPK:
-
Mitogen-activated protein kinase
- MMP-9:
-
Matrix metalloproteinase-9
- NF-κB:
-
Nuclear factor-κB
- PI3K:
-
Phosphatidylinositol-3 kinase
- PR:
-
Progesterone receptor
- S-CTSS:
-
Stromal cathepsin S
- Sp-1:
-
Specificity protein-1
- TNBC:
-
Triple-negative breast cancer
- TPH1:
-
Tryptophan hydroxylase 1
References
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144(5):646–674. doi:10.1016/j.cell.2011.02.013
Gocheva V, Joyce JA (2007) Cysteine cathepsins and the cutting edge of cancer invasion. Cell Cycle 6(1):60–64
Yang Y, Lim S-K, Choong L-Y, Lee H, Chen Y, Chong P-K, Ashktorab H, Wang TT, Salto-Tellez M, Yeoh K-G (2010) Cathepsin S mediates gastric cancer cell migration and invasion via a putative network of metastasis-associated proteins. J Proteome Res 9(12):6801
Gormley J, Hegarty S, O’Grady A, Stevenson M, Burden R, Barrett H, Scott C, Johnston J, Wilson R, Kay E (2011) The role of Cathepsin S as a marker of prognosis and predictor of chemotherapy benefit in adjuvant CRC: a pilot study. Br J Cancer 105(10):1487–1494
Sage J, Mallèvre F, Barbarin-Costes F, Samsonov SA, Gehrcke J-P, Pisabarro MT, Perrier E, Schnebert S, Roget A, Livache T (2013) Binding of chondroitin 4-sulfate to cathepsin S regulates its enzymatic activity. Biochemistry 52(37):6487–6498
Shi G-P, Webb AC, Foster KE, Knoll J, Lemere CA, Munger JS, Chapman HA (1994) Human cathepsin S: chromosomal localization, gene structure, and tissue distribution. J Biol Chem 269(15):11530–11536
Gocheva V, Zeng W, Ke D, Klimstra D, Reinheckel T, Peters C, Hanahan D, Joyce JA (2006) Distinct roles for cysteine cathepsin genes in multistage tumorigenesis. Genes Dev 20(5):543–556
Burden RE, Gormley JA, Jaquin TJ, Small DM, Quinn DJ, Hegarty SM, Ward C, Walker B, Johnston JA, Olwill SA (2009) Antibody-mediated inhibition of cathepsin S blocks colorectal tumor invasion and angiogenesis. Clin Cancer Res 15(19):6042–6051
Ryschich E, Lizdenis P, Ittrich C, Benner A, Stahl S, Hamann A, Schmidt J, Knolle P, Arnold B, Hämmerling GJ (2006) Molecular fingerprinting and autocrine growth regulation of endothelial cells in a murine model of hepatocellular carcinoma. Cancer Res 66(1):198–211
Fernández PL, Farré X, Nadal A, Fernández E, Peiró N, Sloane BF, Shi GP, Chapman HA, Campo E, Cardesa A (2001) Expression of cathepsins B and S in the progression of prostate carcinoma. Int J Cancer 95(1):51–55
Fan Q, Wang X, Zhang H, Li C, Fan J, Xu J (2012) Silencing cathepsin S gene expression inhibits growth, invasion and angiogenesis of human hepatocellular carcinoma in vitro. Biochem Biophys Res Commun 425(4):703–710
Wang B, Sun J, Kitamoto S, Yang M, Grubb A, Chapman HA, Kalluri R, Shi G-P (2006) Cathepsin S controls angiogenesis and tumor growth via matrix-derived angiogenic factors. J Biol Chem 281(9):6020–6029
Sevenich L, Bowman RL, Mason SD, Quail DF, Rapaport F, Elie BT, Brogi E, Brastianos PK, Hahn WC, Holsinger LJ (2014) Analysis of tumour-and stroma-supplied proteolytic networks reveals a brain-metastasis-promoting role for cathepsin S. Nat Cell Biol 16(9):876–888
Flannery T, McQuaid S, McGoohan C, McConnell RS, McGregor G, Mirakhur M, Hamilton P, Diamond J, Cran G, Walker B (2006) Cathepsin S expression: an independent prognostic factor in glioblastoma tumours—a pilot study. Int J Cancer 119(4):854–860
Jobs E, Ingelsson E, Risérus U, Nerpin E, Jobs M, Sundström J, Basu S, Larsson A, Lind L, Ärnlöv J (2011) Association between serum cathepsin S and mortality in older adults. JAMA 306(10):1113–1121
Chang W-SW, Wu H-R, Yeh C-T, Wu C-W, Chang J-Y (2007) Lysosomal cysteine proteinase cathepsin S as a potential target for anti-cancer therapy. J Cancer Mol 3(1):5–14
Gautam J, Banskota S, Regmi SC, Ahn S, Jeon YH, Jeong H, Kim SJ, Nam T-G, Jeong B-S, Kim J-A Tryptophan hydroxylase 1 and 5-HT7 receptors preferentially expressed in triple-negative breast cancer promote cancer progression through autocrine serotonin signaling. Mol Cancer (under revision)
Neckers L (2002) Hsp90 inhibitors as novel cancer chemotherapeutic agents. Trends Mol Med 8(4):S55–S61
Pick E, Kluger Y, Giltnane JM, Moeder C, Camp RL, Rimm DL, Kluger HM (2007) High HSP90 expression is associated with decreased survival in breast cancer. Cancer Res 67(7):2932–2937
Subbarao Sreedhar A, Kalmár É, Csermely P, Shen Y-F (2004) Hsp90 isoforms: functions, expression and clinical importance. FEBS Lett 562(1–3):11–15
Bae YK, Kim A, Kim MK, Choi JE, Kang SH, Lee SJ (2013) Fibronectin expression in carcinoma cells correlates with tumor aggressiveness and poor clinical outcome in patients with invasive breast cancer. Hum Pathol 44(10):2028–2037
Choi JE, Kang SH, Lee SJ, Bae YK (2015) Androgen receptor expression predicts decreased survival in early stage triple-negative breast cancer. Ann Surg Oncol 22(1):82–89
Hollestelle A, Elstrodt F, Nagel JH, Kallemeijn WW, Schutte M (2007) Phosphatidylinositol-3-OH kinase or RAS pathway mutations in human breast cancer cell lines. Mol Cancer Res 5(2):195–201
McAuliffe PF, Meric-Bernstam F, Mills GB, Gonzalez-Angulo AM (2010) Deciphering the role of PI3K/Akt/mTOR pathway in breast cancer biology and pathogenesis. Clin Breast Cancer 10:S59–S65
Jang J-Y, Jeon Y-K, Kim C-W (2010) Degradation of HER2/neu by ANT2 shRNA suppresses migration and invasiveness of breast cancer cells. BMC Cancer 10(1):391
Santagata S, Hu R, Lin NU, Mendillo ML, Collins LC, Hankinson SE, Schnitt SJ, Whitesell L, Tamimi RM, Lindquist S (2011) High levels of nuclear heat-shock factor 1 (HSF1) are associated with poor prognosis in breast cancer. Proc Natl Acad Sci 108(45):18378–18383
Geraghty P, Greene CM, O’Mahony M, O’Neill SJ, Taggart CC, McElvaney NG (2007) Secretory leucocyte protease inhibitor inhibits interferon-γ-induced cathepsin s expression. J Biol Chem 282(46):33389–33395
Lee TKW, Cheung VCH, Lu P, Lau EYT, Ma S, Tang KH, Tong M, Lo J, Ng IOL (2014) Blockade of CD47-mediated cathepsin S/protease-activated receptor 2 signaling provides a therapeutic target for hepatocellular carcinoma. Hepatology 60(1):179–191
Graham TR, Odero-Marah VA, Chung LW, Agrawal KC, Davis R, Abdel-Mageed AB (2009) PI3K/Akt-dependent transcriptional regulation and activation of BMP-2-Smad signaling by NF-κB in metastatic prostate cancer cells. Prostate 69(2):168–180
Aggarwal BB, Vijayalekshmi R, Sung B (2009) Targeting inflammatory pathways for prevention and therapy of cancer: short-term friend, long-term foe. Clin Cancer Res 15(2):425–430
Liu P, Wilson MJ (2012) miR-520c and miR-373 upregulate MMP9 expression by targeting mTOR and SIRT1, and activate the Ras/Raf/MEK/Erk signaling pathway and NF-κB factor in human fibrosarcoma cells. J Cell Physiol 227(2):867–876
Cheng XW, Shi G-P, Kuzuya M, Sasaki T, Okumura K, Murohara T (2012) Role for cysteine protease cathepsins in heart disease focus on biology and mechanisms with clinical implication. Circulation 125(12):1551–1562
McCubrey JA, Steelman LS, Chappell WH, Abrams SL, Wong EW, Chang F, Lehmann B, Terrian DM, Milella M, Tafuri A (2007) Roles of the Raf/MEK/ERK pathway in cell growth, malignant transformation and drug resistance. Biochimica et Biophysica Acta (BBA)—Mol Cell Res 1773(8):1263–1284
Lee S-J, Namkoong S, Kim Y-M, Kim C-K, Lee H, Ha K-S, Chung H-T, Kwon Y-G, Kim Y-M (2006) Fractalkine stimulates angiogenesis by activating the Raf-1/MEK/ERK-and PI3K/Akt/eNOS-dependent signal pathways. Am J Physiol Heart Circ Physiol 291(6):H2836–H2846
Yajima I, Kumasaka MY, Thang ND, Goto Y, Takeda K, Yamanoshita O, Iida M, Ohgami N, Tamura H, Kawamoto Y (2011) RAS/RAF/MEK/ERK and PI3K/PTEN/AKT signaling in malignant melanoma progression and therapy. Dermatol Res Pract 2012
Acknowledgments
This work was supported by a Yeungnam University Research Grant (2012).
Author contributions
JAK conceived and designed the study with YKB. YKB performed immunohistochemical staining and analyses on human breast cancer tissue microarray sections. JG performed experiments with cell lines, and analyzed the data with JAK. All authors participated in the manuscript writing, and have approved the final version of the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
The study on CTSS expression in the tissue microarray sections of 1451 human invasive breast cancer samples was approved by the Institutional Review Board of Yeungnam University Hospital (2015-06-007), and the requirement for informed consent was waived.
Informed consent
Not applicable.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. S1 Treatment with GA had no effect on MDA-MB-231 cell viability.
Supplementary Fig. S2 GA (an HSP90 inhibitor) blocked the nuclear translocation of NF-κB in MDA-MB-231 cells. Supplementary material 1 (TIFF 3388 kb)
Rights and permissions
About this article
Cite this article
Gautam, J., Bae, Y.K. & Kim, JA. Up-regulation of cathepsin S expression by HSP90 and 5-HT7 receptor-dependent serotonin signaling correlates with triple negativity of human breast cancer. Breast Cancer Res Treat 161, 29–40 (2017). https://doi.org/10.1007/s10549-016-4027-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-016-4027-1