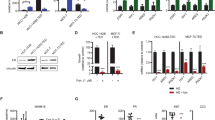
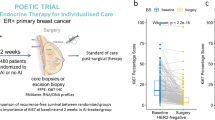
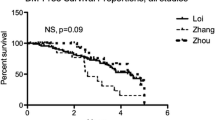
Abstract
Estrogens (E) and estrogen receptors (ER) are implicated in breast cancer growth and are targets of hormonal therapies. Such therapies commonly use aromatase inhibitors (AI) to block E production, or antiestrogens like tamoxifen (TAM), which targets ER. Here we compare genes in pre-and post-treatment tumor pairs of patients with ER+ tumors, that were treated preoperatively with the AI exemestane alone, or with exemestane plus TAM. The accompanying manuscript shows that tumors from patients treated with AI + TAM responded less well than tumors treated with AI alone. The present manuscript defines the E-signaling mechanisms underlying these differences, and describes genetic differences between hormone responsive versus intrinsically resistant ER+ tumors. Gene expression profiling was performed on paired tumor biopsies of individual patients before treatment, and after 4 months of treatment with AI or AI + TAM. Separately, E and TAM regulated genes were defined using a human breast cancer xenograft model. We demonstrate: (1) that AI alone alters global gene expression ∼5 times more than AI + TAM, and is 11 times more effective in modifying expression of E regulated genes; (2) among E regulated genes, there is little overlap between AI and AI + TAM treatment groups. AI + TAM preferentially induce genes, like androgen receptors, expressing TAM “E-like” agonist activity, or genes uniquely regulated by TAM. (3) A pre-treatment 25 gene signature of ER+ tumors may predict response or intrinsic resistance to endocrine therapies. We conclude that in the presence of exemestane, the agonist properties of TAM are paradoxically exposed, diminishing the effectiveness of combination therapy.







Similar content being viewed by others
Abbreviations
- ER:
-
Estrogen receptors
- PR:
-
Progesterone receptors
- AR:
-
Androgen receptors
- E:
-
Estrogens or estradiol
- ovx:
-
Ovariectomized
- TAM:
-
Tamoxifen
- AI:
-
Aromatase inhibitors
References
Chalbos D, Vignon F, Keydar I, Rochefort H (1982) Estrogens stimulate cell proliferation and induce secretory proteins in a human breast cancer cell line (T47D). J Clin Endocrinol Metab 55:276–283
Russo J, Russo IH (2006) The role of estrogen in the initiation of breast cancer. J Steroid Biochem Mol Biol 102:89–96
Beatson G (1896) On the treatment of inoperable cases of carcinoma of the mamma: suggestions for a new method of treatment, with illustrative cases. Lancet 147:101–107
Smith IE, Dowsett M (2003) Aromatase inhibitors in breast cancer. N Engl J Med 348:2431–2442
Osborne CK (1998) Tamoxifen in the treatment of breast cancer. N Engl J Med 339:1609–1618
Shek LL, Godolphin W (1989) Survival with breast cancer: the importance of estrogen receptor quantity. Eur J Cancer Clin Oncol 25:243–250
Normanno N, Di Maio M, De Maio E et al (2005) Mechanisms of endocrine resistance and novel therapeutic strategies in breast cancer. Endocr Relat Cancer 12:721–747
Osborne CK, Zhao H, Fuqua SA (2000) Selective estrogen receptor modulators: structure, function, and clinical use. J Clin Oncol 18:3172–3186
Fisher B, Costantino JP, Wickerham DL et al (2005) Tamoxifen for the prevention of breast cancer: current status of the National Surgical Adjuvant Breast and Bowel Project P-1 study. J Natl Cancer Inst 97:1652–1662
Frasor J, Stossi F, Danes JM et al (2004) Selective estrogen receptor modulators: discrimination of agonistic versus antagonistic activities by gene expression profiling in breast cancer cells. Cancer Res 64:1522–1533
Horwitz KB, Koseki Y, McGuire WL (1978) Estrogen control of progesterone receptor in human breast cancer: role of estradiol and antiestrogen. Endocrinology 103:1742–1751
Sutherland RL, Hall RE, Taylor IW (1983) Cell proliferation kinetics of MCF-7 human mammary carcinoma cells in culture and effects of tamoxifen on exponentially growing and plateau-phase cells. Cancer Res 43:3998–4006
Thiantanawat A, Long BJ, Brodie AM (2003) Signaling pathways of apoptosis activated by aromatase inhibitors and antiestrogens. Cancer Res 63:8037–8050
Bianco NR, Perry G, Smith MA et al (2003) Functional implications of antiestrogen induction of quinone reductase: inhibition of estrogen-induced deoxyribonucleic acid damage. Mol Endocrinol 17:1344–1355
Jelovac D, Macedo L, Handratta V et al (2004) Effects of exemestane and tamoxifen in a postmenopausal breast cancer model. Clin Cancer Res 10:7375–7381
Baum M, Budzar AU, Cuzick J et al (2002) Anastrozole alone or in combination with tamoxifen versus tamoxifen alone for adjuvant treatment of postmenopausal women with early breast cancer: first results of the ATAC randomised trial. Lancet 359:2131–2139
Smith IE, Dowsett M, Ebbs SR et al (2005) Neoadjuvant treatment of postmenopausal breast cancer with anastrozole, tamoxifen, or both in combination: the Immediate Preoperative Anastrozole, Tamoxifen, or Combined with Tamoxifen (IMPACT) multicenter double-blind randomized trial. J Clin Oncol 23:5108–5116
Harvell DM, Spoelstra NS, Singh M et al (2007) Molecular signatures of response and intrinsic resistance to neoadjuvant endocrine therapy for breast cancer. Breast Cancer Res Treat. doi:10.1007/s10549-008-9897-4
Harvell DM, Richer JK, Allred DC et al (2006) Estradiol regulates different genes in human breast tumor xenografts compared with the identical cells in culture. Endocrinology 147:700–713
Frasor J, Danes JM, Komm B et al (2003) Profiling of estrogen up- and down-regulated gene expression in human breast cancer cells: insights into gene networks and pathways underlying estrogenic control of proliferation and cell phenotype. Endocrinology 144:4562–4574
Creighton CJ, Cordero KE, Larios JM et al (2006) Genes regulated by estrogen in breast tumor cells in vitro are similarly regulated in vivo in tumor xenografts and human breast tumors. Genome Biol 7:R28
Katoh Y, Katoh M (2006) Canonical WNT signaling pathway and human AREG. Int J Mol Med 17:1163–1166
Jonsson M, Dejmek J, Bendahl PO et al (2002) Loss of Wnt-5a protein is associated with early relapse in invasive ductal breast carcinomas. Cancer Res 62:409–416
Salomon DS, Normanno N, Ciardiello F et al (1995) The role of amphiregulin in breast cancer. Breast Cancer Res Treat 33:103–114
Frasor J, Chang EC, Komm B et al (2006) Gene expression preferentially regulated by tamoxifen in breast cancer cells and correlations with clinical outcome. Cancer Res 66:7334–7340
Chambers RC, Leoni P, Kaminski N et al (2003) Global expression profiling of fibroblast responses to transforming growth factor-beta1 reveals the induction of inhibitor of differentiation-1 and provides evidence of smooth muscle cell phenotypic switching. Am J Pathol 162:533–546
Morgan TE, Laping NJ, Rozovsky I et al (1995) Clusterin expression by astrocytes is influenced by transforming growth factor beta 1 and heterotypic cell interactions. J Neuroimmunol 58:101–110
Alaoui-Jamali MA, Song DJ, Benlimame N et al (2003) Regulation of multiple tumor microenvironment markers by overexpression of single or paired combinations of ErbB receptors. Cancer Res 63:3764–3774
Landis MD, Seachrist DD, Montanez-Wiscovich ME et al (2005) Gene expression profiling of cancer progression reveals intrinsic regulation of transforming growth factor-beta signaling in ErbB2/Neu-induced tumors from transgenic mice. Oncogene 24:5173–5190
Mackay A, Jones C, Dexter T et al (2003) cDNA microarray analysis of genes associated with ERBB2 (HER2/neu) overexpression in human mammary luminal epithelial cells. Oncogene 22:2680–2688
Detre S, Riddler S, Salter J et al (2003) Comparison of the selective estrogen receptor modulator arzoxifene (LY353381) with tamoxifen on tumor growth and biomarker expression in an MCF-7 human breast cancer xenograft model. Cancer Res 63:6516–6522
Itoh T, Karlsberg K, Kijima I et al (2005) Letrozole-, anastrozole-, and tamoxifen-responsive genes in MCF-7aro cells: a microarray approach. Mol Cancer Res 3:203–218
Kristensen VN, Sorlie T, Geisler J et al (2005) Effects of anastrozole on the intratumoral gene expression in locally advanced breast cancer. J Steroid Biochem Mol Biol 95:105–111
Scafoglio C, Ambrosino C, Cicatiello L et al (2006) Comparative gene expression profiling reveals partially overlapping but distinct genomic actions of different antiestrogens in human breast cancer cells. J Cell Biochem 98:1163–1184
Sorlie T, Perou CM, Tibshirani R et al (2001) Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA 98:10869–10874
Mackay A, Urruticoechea A, Dixon JM et al (2007) Molecular response to aromatase inhibitor treatment in primary breast cancer. Breast Cancer Res 9:R37
Chen S, Masri S, Wang X et al (2006) What do we know about the mechanisms of aromatase inhibitor resistance? J Steroid Biochem Mol Biol 102:232–240
Becker M, Sommer A, Kratzschmar JR et al (2005) Distinct gene expression patterns in a tamoxifen-sensitive human mammary carcinoma xenograft and its tamoxifen-resistant subline MaCa 3366/TAM. Mol Cancer Ther 4:151–168
Meijer D, van Agthoven T, Bosma PT et al (2006) Functional screen for genes responsible for tamoxifen resistance in human breast cancer cells. Mol Cancer Res 4:379–386
Shioda T, Chesnes J, Coser KR et al (2006) Importance of dosage standardization for interpreting transcriptomal signature profiles: evidence from studies of xenoestrogens. Proc Natl Acad Sci USA 103:12033–12038
Whitfield ML, George LK, Grant GD et al (2006) Common markers of proliferation. Nat Rev Cancer 6:99–106
Jansen MP, Foekens JA, van Staveren IL et al (2005) Molecular classification of tamoxifen-resistant breast carcinomas by gene expression profiling. J Clin Oncol 23:732–740
Tozlu-Kara S, Roux V, Andrieu C et al (2007) Oligonucleotide microarray analysis of estrogen receptor alpha-positive postmenopausal breast carcinomas: identification of HRPAP20 and TIMELESS as outstanding candidate markers to predict the response to tamoxifen. J Mol Endocrinol 39:305–318
Brys M, Wojcik M, Romanowicz-Makowska H et al (2002) Androgen receptor status in female breast cancer: RT-PCR and Western blot studies. J Cancer Res Clin Oncol 128:85–90
Moinfar F, Okcu M, Tsybrovskyy O et al (2003) Androgen receptors frequently are expressed in breast carcinomas: potential relevance to new therapeutic strategies. Cancer 98:703–711
Ayyanan A, Civenni G, Ciarloni L et al (2006) Increased Wnt signaling triggers oncogenic conversion of human breast epithelial cells by a Notch-dependent mechanism. Proc Natl Acad Sci USA 103:3799–3804
Hodges LC, Cook JD, Lobenhofer EK et al (2003) Tamoxifen functions as a molecular agonist inducing cell cycle-associated genes in breast cancer cells. Mol Cancer Res 1:300–311
Bedirian A, Baldwin C, Abe J et al (2004) Pleckstrin homology and phosphotyrosine-binding domain-dependent membrane association and tyrosine phosphorylation of Dok-4, an inhibitory adapter molecule expressed in epithelial cells. J Biol Chem 279:19335–19349
Franco M, Furstoss O, Simon V et al (2006) The adaptor protein Tom1L1 is a negative regulator of Src mitogenic signaling induced by growth factors. Mol Cell Biol 26:1932–1947
Dooley TP, Haldeman-Cahill R, Joiner J et al (2000) Expression profiling of human sulfotransferase and sulfatase gene superfamilies in epithelial tissues and cultured cells. Biochem Biophys Res Commun 277:236–245
Tang B, Vu M, Booker T et al (2003) TGF-beta switches from tumor suppressor to prometastatic factor in a model of breast cancer progression. J Clin Invest 112:1116–1124
Slamon DJ, Leyland-Jones B, Shak S et al (2001) Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med 344:783–792
Todorovic-Rakovic N (2005) TGF-beta 1 could be a missing link in the interplay between ER and HER-2 in breast cancer. Med Hypotheses 65:546–551
Oh DS, Troester MA, Usary J et al (2006) Estrogen-regulated genes predict survival in hormone receptor-positive breast cancers. J Clin Oncol 24:1656–1664
Acknowledgements
We thank Ted Shade, Tzu Phang, Susan Trapp and Larry Hunter for help with the microarray data analysis and acknowledge use of the Microarray Core and Computational Bioscience Program of the University of Colorado Cancer Center. This work was funded by the National Institutes of Health (National Research Service Award F32 CA103511); United Negro College Fund/Pfizer grant (to D.M.E.H.); DOD Clinical Bridge Award DAMD17-02-1-0353 (to J.K.R.); and the National Institutes of Health Grant (RO1 CA26869), the National Foundation of Cancer Research, the Avon Foundation, and the Breast Cancer Research Foundation (to K.B.H.).
Author information
Authors and Affiliations
Corresponding author
Additional information
This is a commentary to 10.1007/s10549-008-9897-4.
Djuana M. E. Harvell, Jennifer K. Richer—Co-first authors.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Harvell, D.M.E., Richer, J.K., Singh, M. et al. Estrogen regulated gene expression in response to neoadjuvant endocrine therapy of breast cancers: tamoxifen agonist effects dominate in the presence of an aromatase inhibitor. Breast Cancer Res Treat 112, 489–501 (2008). https://doi.org/10.1007/s10549-008-9923-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-008-9923-6