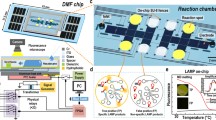
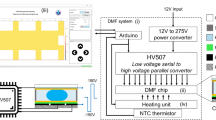
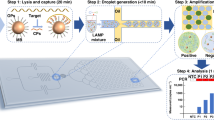
Abstract
A major goal in the development of point-of-care (POC) devices is to build them as portable to provide a rapid and effective determination for disease pathogens. In nucleic acid testing, an optical detection system used to monitor the product of nucleic acid amplification has always been a bulky accessory. In this work, we developed a handheld, automatic and detection system-free thermal digital microfluidic (DMF) device for DNA detection by loop-mediated isothermal amplification (LAMP). Droplet manipulation and real-time temperature control systems were integrated into a handheld device. The control software could be installed into any tablet and communicate with the device via Bluetooth. In the experimentation, we loaded 2-μl samples with an electrowetting force into sandwich-structured DMF chips, thereby considerably reducing reagent consumptions. After an on-chip LAMP reaction, we added a highly concentrated SYBR Green I droplet and mixed it with a reaction droplet to enable product detection with the naked eye. This step prevented aerosol contamination by avoiding the exposure of the reaction droplet to the air. Using a blood parasite Trypanosoma brucei as a model system, this system showed similar results as a commercial thermal cycler and could detect 40 copies per reaction of the DNA target. This low-cost, compact device removed the bulky optical system for DNA detection, thus enabling it to be well suited for POC testing.




Similar content being viewed by others
References
M. Berriman et al., The genome of the African trypanosome Trypanosoma brucei. Science 309, 416–422 (2005). https://doi.org/10.1126/science.1112642
B. Coelho, B. Veigas, E. Fortunato, R. Martins, H. Aguas, R. Igreja, P.V. Baptista, Digital microfluidics for nucleic acid amplification. Sensors (Basel) 17 (2017a). https://doi.org/10.3390/s17071495
B.J. Coelho, B. Veigas, H. Aguas, E. Fortunato, R. Martins, P.V. Baptista, R. Igreja, A digital microfluidics platform for loop-mediated isothermal amplification detection. Sensors (Basel) 17 (2017b). https://doi.org/10.3390/s17112616
P. Craw, R.E. Mackay, A. Naveenathayalan, C. Hudson, M. Branavan, S.T. Sadiq, W. Balachandran, A simple, low-cost platform for real-time isothermal nucleic acid amplification. Sensors (Basel) 15, 23418–23430 (2015). https://doi.org/10.3390/s150923418
C. Dong, Y. Jia, J. Gao, T. Chen, P.I. Mak, M.I. Vai, R.P. Martins, A 3D microblade structure for precise and parallel droplet splitting on digital microfluidic chips. Lab Chip 17, 896–904 (2017). https://doi.org/10.1039/c6lc01539e
P.K. Drain, E.P. Hyle, F. Noubary, K.A. Freedberg, D. Wilson, W.R. Bishai, W. Rodriguez, I.V. Bassett, Diagnostic point-of-care tests in resource-limited settings. Lancet Infect. Dis. 14, 239–249 (2014). https://doi.org/10.1016/S1473-3099(13)70250-0
J. Gao, X. Liu, T. Chen, P.I. Mak, Y. Du, M.I. Vai, B. Lin, R.P. Martins, An intelligent digital microfluidic system with fuzzy-enhanced feedback for multi-droplet manipulation. Lab Chip 13, 443–451 (2013). https://doi.org/10.1039/c2lc41156c
M. Goto, E. Honda, A. Ogura, A. Nomoto, K. Hanaki, Colorimetric detection of loop-mediated isothermal amplification reaction by using hydroxy naphthol blue. Biotechniques 46, 167–172 (2009). https://doi.org/10.2144/000113072
T. Iwamoto, T. Sonobe, K. Hayashi, Loop-mediated isothermal amplification for direct detection of Mycobacterium tuberculosis complex, M. avium, and M. intracellulare in sputum samples. J. Clin. Microbiol. 41, 2616–2622 (2003)
A.P. Jackson, M. Sanders, A. Berry, J. McQuillan, M.A. Aslett, M.A. Quail, B. Chukualim, P. Capewell, A. MacLeod, S.E. Melville, W. Gibson, J.D. Barry, M. Berriman, C. Hertz-Fowler, The genome sequence of Trypanosoma brucei gambiense, causative agent of chronic human african trypanosomiasis. PLoS Negl. Trop. Dis. 4, e658 (2010). https://doi.org/10.1371/journal.pntd.0000658
S. Kalsi, M. Valiadi, M.N. Tsaloglou, L. Parry-Jones, A. Jacobs, R. Watson, C. Turner, R. Amos, B. Hadwen, J. Buse, C. Brown, M. Sutton, H. Morgan, Rapid and sensitive detection of antibiotic resistance on a programmable digital microfluidic platform. Lab Chip 15, 3065–3075 (2015). https://doi.org/10.1039/c5lc00462d
K. Karthik, R. Rathore, P. Thomas, T.R. Arun, K.N. Viswas, K. Dhama, R.K. Agarwal, New closed tube loop mediated isothermal amplification assay for prevention of product cross-contamination. MethodsX 1, 137–143 (2014). https://doi.org/10.1016/j.mex.2014.08.009
M. Kuhnemund, D. Witters, M. Nilsson, J. Lammertyn, Circle-to-circle amplification on a digital microfluidic chip for amplified single molecule detection. Lab Chip 14, 2983–2992 (2014). https://doi.org/10.1039/c4lc00348a
S.M. Lanham, D.G. Godfrey, Isolation of salivarian trypanosomes from man and other mammals using DEAE-cellulose. Exp. Parasitol. 28, 521–534 (1970)
C. Liang, S. Cheng, Y. Chu, H. Wu, B. Zou, H. Huang, T. Xi, G. Zhou, A closed-tube detection of loop-mediated isothermal amplification (LAMP) products using a wax-sealed fluorescent intercalator. J. Nanosci. Nanotechnol. 13, 3999–4005 (2013)
M.G. Mauk, J. Song, C. Liu, H.H. Bau, Simple approaches to minimally-instrumented, microfluidic-based point-of-care nucleic acid amplification tests. Biosensors (Basel) 8 (2018). https://doi.org/10.3390/bios8010017
Y. Mori, K. Nagamine, N. Tomita, T. Notomi, Detection of loop-mediated isothermal amplification reaction by turbidity derived from magnesium pyrophosphate formation. Biochem. Biophys. Res. Commun. 289, 150–154 (2001). https://doi.org/10.1006/bbrc.2001.5921
F.B. Myers, R.H. Henrikson, J.M. Bone, L.P. Lee, A handheld point-of-care genomic diagnostic system. PLoS One 8, e70266 (2013). https://doi.org/10.1371/journal.pone.0070266
K. Nagamine, T. Hase, T. Notomi, Accelerated reaction by loop-mediated isothermal amplification using loop primers. Mol. Cell. Probes 16, 223–229 (2002)
A. Niemz, T.M. Ferguson, D.S. Boyle, Point-of-care nucleic acid testing for infectious diseases. Trends Biotechnol. 29, 240–250 (2011). https://doi.org/10.1016/j.tibtech.2011.01.007
Z.K. Njiru, A.S. Mikosza, T. Armstrong, J.C. Enyaru, J.M. Ndung'u, A.R. Thompson, Loop-mediated isothermal amplification (LAMP) method for rapid detection of Trypanosoma brucei rhodesiense. PLoS Negl. Trop. Dis. 2, e147 (2008a). https://doi.org/10.1371/journal.pntd.0000147
Z.K. Njiru, A.S. Mikosza, E. Matovu, J.C. Enyaru, J.O. Ouma, S.N. Kibona, R.C. Thompson, J.M. Ndung'u, African trypanosomiasis: Sensitive and rapid detection of the sub-genus Trypanozoon by loop-mediated isothermal amplification (LAMP) of parasite DNA. Int. J. Parasitol. 38, 589–599 (2008b). https://doi.org/10.1016/j.ijpara.2007.09.006
Notomi T, Okayama H, Masubuchi H, Yonekawa T, Watanabe K, Amino N, Hase T (2000) Loop-mediated isothermal amplification of DNA. Nucleic Acids Res. 28:E63, 63e
I.P. Oscorbin, E.A. Belousova, A.I. Zakabunin, U.A. Boyarskikh, M.L. Filipenko, Comparison of fluorescent intercalating dyes for quantitative loop-mediated isothermal amplification (qLAMP). Biotechniques 61, 20–25 (2016). https://doi.org/10.2144/000114432
C.M. Pandey, S. Augustine, S. Kumar, S. Kumar, S. Nara, S. Srivastava, B.D. Malhotra, Microfluidics based point-of-care diagnostics. Biotechnol. J. 13 (2018). https://doi.org/10.1002/biot.201700047
M.M. Parida, S.R. Santhosh, P.K. Dash, N.K. Tripathi, P. Saxena, S. Ambuj, A.K. Sahni, P.V. Lakshmana Rao, K. Morita, Development and evaluation of reverse transcription-loop-mediated isothermal amplification assay for rapid and real-time detection of Japanese encephalitis virus. J. Clin. Microbiol. 44, 4172–4178 (2006). https://doi.org/10.1128/JCM.01487-06
S. Sharma, J. Zapatero-Rodriguez, P. Estrela, R. O'Kennedy, Point-of-care diagnostics in low resource settings: Present status and future role of microfluidics. Biosensors (Basel) 5, 577–601 (2015). https://doi.org/10.3390/bios5030577
J. Song, M.G. Mauk, B.A. Hackett, S. Cherry, H.H. Bau, C. Liu, Instrument-free point-of-care molecular detection of Zika virus. Anal. Chem. 88, 7289–7294 (2016). https://doi.org/10.1021/acs.analchem.6b01632
J. Sun, M.J. Najafzadeh, V. Vicente, L. Xi, G.S. de Hoog, Rapid detection of pathogenic fungi using loop-mediated isothermal amplification, exemplified by Fonsecaea agents of chromoblastomycosis. J. Microbiol. Methods 80, 19–24 (2010). https://doi.org/10.1016/j.mimet.2009.10.002
N. Tomita, Y. Mori, H. Kanda, T. Notomi, Loop-mediated isothermal amplification (LAMP) of gene sequences and simple visual detection of products. Nat. Protoc. 3, 877–882 (2008). https://doi.org/10.1038/nprot.2008.57
A.H. Velders, C. Schoen, V. Saggiomo, Loop-mediated isothermal amplification (LAMP) shield for Arduino DNA detection. BMC Res Notes 11, 93 (2018). https://doi.org/10.1186/s13104-018-3197-9
L. Wan, T. Chen, J. Gao, C. Dong, A.H. Wong, Y. Jia, P.I. Mak, C.X. Deng, R.P. Martins, A digital microfluidic system for loop-mediated isothermal amplification and sequence specific pathogen detection. Sci. Rep. 7, 14586 (2017). https://doi.org/10.1038/s41598-017-14698-x
Acknowledgements
This work was supported by FDCT110/2016/A3 and AMSV SKL Fund from the Macao Science and Technology Development Fund (FDCT), MYRG2017-00022-AMSV and SRG2016-00072-AMSV from the University of Macau, and #31720103918 from the National Natural Science Foundation of China.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wan, L., Gao, J., Chen, T. et al. LampPort: a handheld digital microfluidic device for loop-mediated isothermal amplification (LAMP). Biomed Microdevices 21, 9 (2019). https://doi.org/10.1007/s10544-018-0354-9
Published:
DOI: https://doi.org/10.1007/s10544-018-0354-9