Abstract
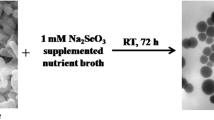
Environmental bacterial isolates play a very important role in bioremediation of metals and toxic metalloids. A bacterial strain with high selenite (SeO32−) tolerance and reducing capability was isolated from electronic waste dump site in Banaras Hindu University, Varanasi, India. Based on 16 S rRNA sequencing and BLAST search, this bacterial isolate was identified as Bacillus paramycoides and designated as strain MF-14. It tolerated Sodium selenite up to 110 mM when grown aerobically in LB broth and reduced selenite into elemental selenium (Se0) significantly within 24 h with concomitant biosynthesis of selenium nanoparticles as clearly revealed by brick red precipitate and specific surface plasmon resonance peak at 210 nm using UV–Visible spectrophotometer. Scanning electron microscopy (SEM) analysis of this bacterial strain exposed to 1mM and 5 mM selenite also demonstrated morphological alterations as cell enlargement due to accumulation and bioprecipitation of elemental selenium (Se0). The FTIR analysis clearly demonstrated that functional groups present on the surface of biogenic selenium nanoparticles (SeNPs) play a significant role in the stabilization and capping of SeNPs. Furthermore, these SeNPs were characterized using spectroscopic analysis involving Dynamic light scattering, zeta potential, XPS, FTIR, XRD and Raman spectroscopy which clearly revealed particle size 10–700 nm, amorphous nature, stability as well as it’s oxidation state. The biochemical studies have demonstrated that membrane bound reductase enzyme may be responsible for significant reduction of selenite into elemental selenium. Therefore, we may employ Bacillus paramycoides strain MF-14 successfully for bioremediation of selenite contaminated environmental sites with concomitant green synthesis of SeNPs.










Similar content being viewed by others
Data Availability
The datasets used/generated in the current study is presented in this manuscript.
References
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215(3):403–410. https://doi.org/10.1016/S0022-2836(05)80360-2
Avendaño R, Chaves N, Fuentes P, Sánchez E, Jiménez JI, Chavarría M (2016) Production of selenium nanoparticles in Pseudomonas putida KT2440. Sci Rep 6(1):1–9. https://doi.org/10.1038/srep37155
Bajaj M, Schmidt S, Winter J (2012) Formation of Se (0) nanoparticles by Duganella sp. and Agrobacterium sp. isolated from Se-laden soil of North-East Punjab, India. Microb Cell Fact 11(1):1–14. https://doi.org/10.1186/1475-2859-11-64
Bhattacharjee A, Basu A, Sen T, Biswas J, Bhattacharya S (2017) Nano-Se as a novel candidate in the management of oxidative stress related disorders and cancer. The Nucleus 60(2):137–145. https://doi.org/10.1007/s13237-016-0183-2
Björnstedt M, Kumar S, Holmgren A (1992) Selenodiglutathione is a highly efficient oxidant of reduced thioredoxin and a substrate for mammalian thioredoxin reductase. J Biol Chem 267(12):8030–8034. https://doi.org/10.1016/S0021-9258(18)42403-9
Borah SN, Goswami L, Sen S, Sachan D, Sarma H, Montes M, Peralta-Videa JR, Pakshirajan K, Narayan M (2021) Selenite bioreduction and biosynthesis of selenium nanoparticles by Bacillus paramycoides SP3 isolated from coal mine over-burden leachate. Environ Pollut 285:117519. https://doi.org/10.1016/j.envpol.2021.117519
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1–2):248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Brown MW, Watkinson JH (1977) An automated fluorimetric method for the determination of nanogram quantities of selenium. Anal Chim Acta 89(1):29–35. https://doi.org/10.1016/S0003-2670(01)83067-1
Che L, Dong Y, Wu M, Zhao Y, Liu L, Zhou H (2017) Characterization of selenite reduction by Lysinibacillus sp. ZYM-1 and photocatalytic performance of biogenic selenium nanospheres. ACS Sustain Chem Eng 5(3):2535–2543. https://doi.org/10.1021/acssuschemeng.6b02889
Coates J (2000) Interpretation of infrared spectra, a practical approach. Encycl anal chem. https://doi.org/10.1002/9780470027318.a5606
Debieux CM, Dridge EJ, Mueller CM, Splatt P, Paszkiewicz K, Knight I, Florance H, Love J, Titball RW, Lewis RJ, Richardson DJ (2011) A bacterial process for selenium nanosphere assembly. PANS 108(33):13480–13485. https://doi.org/10.1073/pnas.1105959108
Dhanjal S, Cameotra SS (2010) Aerobic biogenesis of selenium nanospheres by Bacillus cereus isolated from coalmine soil. Microb cell fact 9(1):1–11. https://doi.org/10.1186/1475-2859-9-52
Dimkpa C (2016) Microbial siderophores: production, detection and application in agriculture and environment. Endocytobiosis & Cell Res 27(2):7–16.
Dubey AK, Jeevaratnam K (2015) Structural characterization and functional evaluation of an exopolysaccharide produced by Weissella confusa AJ53, an isolate from fermented Uttapam batter supplemented with Piper betle L leaves. Food Sci Biotechnol 24(6):2117–2124. https://doi.org/10.1007/s10068-015-0281-y
Eswayah AS, Smith TJ, Gardiner PH (2016) Microbial transformations of selenium species of relevance to bioremediation. Appl Environ Microbiol 82(16):4848–4859. https://doi.org/10.1128/AEM.00877-16
Eszenyi P, Sztrik A, Babka B, Prokisch J (2011) Elemental, nano-sized (100–500 nm) selenium production by probiotic lactic acid bacteria. IJBBB 1(2):148. https://doi.org/10.7763/IJBBB.2011.V1.27
Fernández-Llamosas H, Castro L, Blázquez ML, Díaz E, Carmona M (2016) Biosynthesis of selenium nanoparticles by Azoarcus sp. CIB Microb Cell Fact 15(1):1–10. https://doi.org/10.1186/s12934-016-0510-y
Fernández-Llamosas H, Castro L, Blázquez ML, Díaz E, Carmona M (2017) Speeding up bioproduction of selenium nanoparticles by using Vibrio natriegens as microbial factory. Sci Rep 7(1):1–9. https://doi.org/10.1038/s41598-017-16252-1
Fujita M, Ike M, Kashiwa M, Hashimoto R, Soda S (2002) Laboratory-scale continuous reactor for soluble selenium removal using selenate-reducing bacterium, Bacillus sp. SF-1. Biotechnol Bioeng 80(7):755–761. https://doi.org/10.1002/bit.10425
Ghosh A, Mohod AM, Paknikar KM, Jain RK (2008) Isolation and characterization of selenite-and selenate-tolerant microorganisms from selenium-contaminated sites. World J Microb Biotechnol 24(8):1607–1611. https://doi.org/10.1007/s11274-007-9624-z
Gonzalez-Gil G, Lens PN, Saikaly PE (2016) Selenite reduction by anaerobic microbial aggregates: microbial community structure, and proteins associated to the produced selenium spheres. Front Microbiol 7:571. https://doi.org/10.3389/fmicb.2016.00571
Hatfield DL, Berry MJ, Gladyshev VN (2011) Selenium: its molecular biology and role in human health. SSBM, Geneva
Hatfield DL, Tsuji PA, Carlson BA, Gladyshev VN (2014) Selenium and selenocysteine: roles in cancer, health, and development. TIBS 39(3):112–120. https://doi.org/10.1016/j.tibs.2013.12.007
Hockin SL, Gadd GM (2003) Linked redox precipitation of sulfur and selenium under anaerobic conditions by sulfate-reducing bacterial biofilms. Appl Environ Microbiol 69(12):7063–7072. https://doi.org/10.1128/AEM.69.12.7063-7072.2003
Hunter WJ (2014) A Rhizobium selenitireducens protein showing selenite reductase activity. Curr Microbiol 68(3):311–316. https://doi.org/10.1007/s00284-013-0474-7
Hunter WJ (2014) Pseudomonas seleniipraecipitans proteins potentially involved in selenite reduction. Curr Microbiol 69(1):69–74. https://doi.org/10.1007/s00284-014-0555-2
Jain R, Jordan N, Weiss S, Foerstendorf H, Heim K, Kacker R, Hübner R, Kramer H, van Hullebusch ED, Farges F, Lens PN (2015) Extracellular polymeric substances govern the surface charge of biogenic elemental selenium nanoparticles. Environ Sci Technol 49(3):1713–1720. https://doi.org/10.1021/es5043063
Kagami T, Narita T, Kuroda M, Notaguchi E, Yamashita M, Sei K, Soda S, Ike M (2013) Effective selenium volatilization under aerobic conditions and recovery from the aqueous phase by Pseudomonas stutzeri NT-I. Water Res 47(3):1361–1368. https://doi.org/10.1016/j.watres.2012.12.001
Kessi J, Hanselmann KW (2004) Similarities between the abiotic reduction of selenite with glutathione and the dissimilatory reaction mediated by Rhodospirillum rubrum and Escherichia coli. J Biol Chem 279(49):50662–50669. https://doi.org/10.1074/jbc.M405887200
Kessi J, Ramuz M, Wehrli E, Spycher M, Bachofen R (1999) Reduction of selenite and detoxification of elemental selenium by the phototrophic bacterium Rhodospirillum rubrum. Appl Environ Microbiol 65(11):4734–4740. https://doi.org/10.1128/AEM.65.11.4734-4740.1999
Khoei NS, Lampis S, Zonaro E, Yrjälä K, Bernardi P, Vallini G (2017) Insights into selenite reduction and biogenesis of elemental selenium nanoparticles by two environmental isolates of Burkholderia fungorum. New Biotechnol 34:1–11. https://doi.org/10.1016/j.nbt.2016.10.002
Kieliszek M, Błażejak S, Gientka I, Bzducha-Wróbel A (2015) Accumulation and metabolism of selenium by yeast cells. Appl Microbiol Biotechnol 99(13):5373–5382. https://doi.org/10.1007/s00253-015-6650-x
Kora AJ (2018) Bacillus cereus, selenite-reducing bacterium from contaminated lake of an industrial area: a renewable nanofactory for the synthesis of selenium nanoparticles. Bioresour Bioprocess 5(1):1–12. https://doi.org/10.1016/j.jenvman.2016.06.029
Kora AJ, Rastogi L (2016) Biomimetic synthesis of selenium nanoparticles by Pseudomonas aeruginosa ATCC 27853: an approach for conversion of selenite. J Environ Manage 181:231–236. https://doi.org/10.1016/j.jenvman.2016.06.029
Lampis S, Zonaro E, Bertolini C, Bernardi P, Butler CS, Vallini G (2014) Delayed formation of zero-valent selenium nanoparticles by Bacillus mycoides SeITE01 as a consequence of selenite reduction under aerobic conditions. Microb Cell Fact 13(1):1–14. https://doi.org/10.1186/1475-2859-13-35
Lampis S, Zonaro E, Bertolini C, Cecconi D, Monti F, Micaroni M, Turner RJ, Butler CS, Vallini G (2017) Selenite biotransformation and detoxification by Stenotrophomonas maltophilia SeITE02: novel clues on the route to bacterial biogenesis of selenium nanoparticles. J Hazard Mater 324:3–14. https://doi.org/10.1016/j.jhazmat.2016.02.035
Li B, Liu N, Li Y, Jing W, Fan J, Li D, Zhang L, Zhang X, Zhang Z, Wang L (2014) Reduction of selenite to red elemental selenium by Rhodopseudomonas palustris strain N. PLoS ONE 9(4):95955. https://doi.org/10.1371/journal.pone.0095955
Li DB, Cheng YY, Wu C, Li WW, Li N, Yang ZC, Tong ZH, Yu HQ (2014) Selenite reduction by Shewanella oneidensis MR-1 is mediated by fumarate reductase in periplasm. Sci Rep 4(1):1–7. https://doi.org/10.1038/srep03735
Lindblow-Kull CAROLE, Kull FJ, Shrift ALEX (1985) Single transporter for sulfate, selenate, and selenite in Escherichia coli K-12. J Bacteriol 163(3):1267–1269. https://doi.org/10.1128/jb.163.3.1267-1269.1985
Losi ME, Frankenberger WT (1997) Reduction of selenium oxyanions by Enterobacter cloacae SLD1a-1: isolation and growth of the bacterium and its expulsion of selenium particles. Appl Environ Microbiol 63(8):3079–3084. https://doi.org/10.1128/aem.63.8.3079-3084.1997
Macaskie LE, Mikheenko IP, Yong P, Deplanche K, Murray AJ, Paterson-Beedle M, Coker VS, Pearce CI, Cutting R, Pattrick RAD, Vaughan D (2010) Today’s wastes, tomorrow’s materials for environmental protection. Hydrometall 104(3–4):483–487. https://doi.org/10.1016/j.hydromet.2010.01.018
Miethke M, Marahiel MA (2007) Siderophore-based iron acquisition and pathogen control. MMBR 71(3):413–451. https://doi.org/10.1128/MMBR.00012-07
Nancharaiah YV, Lens PNL (2015) Ecology and biotechnology of selenium-respiring bacteria. MMBR 79(1):61–80. https://doi.org/10.1128/MMBR.00037-14
Oremland RS, Herbel MJ, Blum JS, Langley S, Beveridge TJ, Ajayan PM, Curran S (2004) Structural and spectral features of selenium nanospheres produced by Se-respiring bacteria. App Environ Microbiol 70(1):52–60. https://doi.org/10.1128/AEM.70.1.52-60.2004
Painter EP (1941) The chemistry and toxicity of selenium compounds, with special reference to the selenium problem. Chem Rev 28(2):179–213
Pearce CI, Coker VS, Charnock JM, Pattrick RA, Mosselmans JFW, Law N, Lloyd JR (2008) Microbial manufacture of chalcogenide-based nanoparticles via the reduction of selenite using Veillonella atypica: an in-situ EXAFS study. Nanotechnology 19(15):155603. https://doi.org/10.1088/0957-4484/19/15/155603
Pearce CI, Pattrick RA, Law N, Charnock JM, Coker VS, Fellowes JW, Lloyd JR (2009) Investigating different mechanisms for biogenic selenite transformations: Geobacter sulfurreducens, Shewanella oneidensis and Veillonella atypica. Environ Technol 30(12):1313–1326. https://doi.org/10.1080/09593330902984751
Pettine M, Gennari F, Campanella L, Casentini B, Marani D (2012) The reduction of selenium (IV) by hydrogen sulfide in aqueous solutions. GCA 83:37–47. https://doi.org/10.1016/j.gca.2011.12.024
Roskova Z, Skarohlid R, McGachy L (2022) Siderophores: an alternative bioremediation strategy? Sci Total Environ https://doi.org/10.1016/j.scitotenv.2022.153144
Shakibaie M, Forootanfar H, Golkari Y, Mohammadi-Khorsand T, Shakibaie MR (2015) Anti-biofilm activity of biogenic selenium nanoparticles and selenium dioxide against clinical isolates of Staphylococcus aureus, Pseudomonas aeruginosa, and Proteus mirabilis. J Trace Elem Med Biol 29:235–241. https://doi.org/10.1016/j.jtemb.2014.07.020
Siwek B, Bahbouth E, Serra M, Sabbioni E, de Pauw-Gillet MC, Bassleer R (1994) Effect of selenium compounds on murine B16 melanoma cells and pigmented cloned pB16 cells. Arch Toxicol 68(4):246–254. https://doi.org/10.1007/s002040050064
Song D, Li X, Cheng Y, Xiao X, Lu Z, Wang Y, Wang F (2017) Aerobic biogenesis of selenium nanoparticles by Enterobacter cloacae Z0206 as a consequence of fumarate reductase mediated selenite reduction. Sci Rep 7(1):1–10. https://doi.org/10.1038/s41598-017-03558-3
Sors TG, Ellis DR, Salt DE (2005) Selenium uptake, translocation, assimilation and metabolic fate in plants. Photosynth Res 86(3):373–389. https://doi.org/10.1007/s11120-005-5222-9
Srivastava P, Braganca JM, Kowshik M (2014) In vivo synthesis of selenium nanoparticles by Halococcus salifodinae BK18 and their anti-proliferative properties against HeLa cell line. Biotechnol Prog 30(6):1480–1487. https://doi.org/10.1002/btpr.1992
Stolz JF, Oremland RS (1999) Bacterial respiration of arsenic and selenium. FEMS Microbiol Rev 23(5):615–627. https://doi.org/10.1111/j.1574-6976.1999.tb00416.x
Stolz J, Basu P, Oremland R (2002) Microbial transformation of elements: the case of arsenic and selenium. Int Microbiol 5(4):201–207. https://doi.org/10.1007/s10123-002-0091-y
Stolz JF, Basu P, Santini JM, Oremland RS (2006) Arsenic and selenium in microbial metabolism. Annu Rev Microbiol 60:107–130. https://doi.org/10.1146/annurev.micro.60.080805.142053
Tan LC, Nancharaiah YV, van Hullebusch ED, Lens PN (2016) Selenium: environmental significance, pollution, and biological treatment technologies. Biotech Adv 34(5):886–907. https://doi.org/10.1016/j.biotechadv.2016.05.005
Tugarova AV, Mamchenkova PV, Dyatlova YA, Kamnev AA (2018) FTIR and Raman spectroscopic studies of selenium nanoparticles synthesised by the bacterium Azospirillum thiophilum. Spectrochim Acta A Mol Biomol Spectrosc 192:458–463. https://doi.org/10.1016/j.saa.2017.11.050
Tugarova AV, Mamchenkova PV, Khanadeev VA, Kamnev AA (2020) Selenite reduction by the rhizobacterium Azospirillum brasilense, synthesis of extracellular selenium nanoparticles and their characterisation. New Biotechnol 58:17–24. https://doi.org/10.1016/j.nbt.2020.02.003
Vaigankar DC, Mujawar SY, Mohanty AK, Dubey SK (2022) Biogenesis of selenium nanospheres using Halomonas venusta strain GUSDM4 exhibiting potent environmental applications. Arch Microbiol 204:372. https://doi.org/10.1007/s00203-022-02977-9
Wadhwani SA, Shedbalkar UU, Singh R, Chopade BA (2016) Biogenic selenium nanoparticles: current status and future prospects. Appl Microbiol Biotechnol 100(6):2555–2566. https://doi.org/10.1007/s00253-016-7300-7
Wang H, Zhang J, Yu H (2007) Elemental selenium at nano size possesses lower toxicity without compromising the fundamental effect on selenoenzymes: comparison with selenomethionine in mice. Free Radic Biol Med 42(10):1524–1533. https://doi.org/10.1016/j.freeradbiomed.2007.02.013
Wang T, Yang L, Zhang B, Liu J (2010) Extracellular biosynthesis and transformation of selenium nanoparticles and application in H2O2 biosensor. Colloids Surf B: Biointerfaces 80(1):94–102. https://doi.org/10.1016/j.colsurfb.2010.05.041
Wang Y, Shu X, Zhou Q, Fan T, Wang T, Chen X, Li M, Ma Y, Ni J, Hou J, Zhao W (2018) Selenite reduction and the biogenesis of selenium nanoparticles by Alcaligenes faecalis Se03 isolated from the gut of Monochamus alternatus (Coleoptera: Cerambycidae). Int J Mol Sci 19(9):2799. https://doi.org/10.3390/ijms19092799
Wells M, McGarry J, Gaye MM, Basu P, Oremland RS, Stolz JF (2019) Respiratory selenite reductase from Bacillus selenitireducens strain MLS10. J Bacteriol 201(7):e00614–e00618. https://doi.org/10.1128/JB.00614-18
Whanger PD (2002) Selenocompounds in plants and animals and their biological significance. J Am Coll Nutri 21(3):223–232. https://doi.org/10.1080/07315724.2002.10719214
Xia X, Wu S, Li N, Wang D, Zheng S, Wang G (2017) Novel bacterial selenite reductase CsrF responsible for Se (IV) and cr (VI) reduction that produces nanoparticles in Alishewanella sp. WH16-1. J Hazard Mater 342:499–509. https://doi.org/10.1016/j.jhazmat.2017.08.051
Xu D, Yang L, Wang Y, Wang G, Rensing C, Zheng S (2018) Proteins enriched in charged amino acids control the formation and stabilization of selenium nanoparticles in Comamonas testosteroni S44. Sci Rep 8(1):1–11. https://doi.org/10.1038/s41598-018-23295-5
Yamada A, Miyashita M, Inoue K, Matsunaga T (1997) Extracellular reduction of selenite by a novel marine photosynthetic bacterium. Appl Microbiol Biotechnol 48(3):367–372. https://doi.org/10.1007/s002530051064
Zawadzka AM, Crawford RL, Paszczynski AJ (2006) Pyridine-2, 6-bis (thiocarboxylic acid) produced by Pseudomonas stutzeri KC reduces and precipitates selenium and tellurium oxyanions. Appl Environ Microbiol 72(5):3119–3129. https://doi.org/10.1128/AEM.72.5.3119-3129.2006
Zhang W, Chen Z, Liu H, Zhang L, Gao P, Li D (2011) Biosynthesis and structural characteristics of selenium nanoparticles by Pseudomonas alcaliphila. Colloids Surf B: Biointerfaces 88(1):196–201. https://doi.org/10.1016/j.colsurfb.2011.06.031
Acknowledgements
Pooja Yadav and Shraddha Pandey are grateful to University Grant Commission, New Delhi for financial support as UGC Non-NET Fellowship. The authors gratefully acknowledge Distinguished Prof. L.C. Rai for providing laboratory facilities. The authors are thankful to Dr. Krishna Kumar Rai for helping in the graphical representation of data. The authors are also thankful to Department of Physics (BHU) for SEM-EDX facility and AIIMS, New Delhi for TEM and SEM facility. The authors are obliged to Department of Chemistry, BHU and Department of Chemical Engineering (IIT, BHU) for instrumentation facility. The authors are thankful to ISLS, BHU for providing Ultra-Centrifuge facility.
Funding
The research work presented in this article is financially supported by Institution of Eminence (IOE) grant, from Banaras Hindu University, Varanasi, India.
Author information
Authors and Affiliations
Contributions
PY Designed experiments, performed, analyzed the data and prepared the draft manuscript. SP Writing and editing of the draft manuscript. SKD Writing, correction and final editing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yadav, P., Pandey, S. & Dubey, S.K. Selenite bioreduction with concomitant green synthesis of selenium nanoparticles by a selenite resistant EPS and siderophore producing terrestrial bacterium. Biometals 36, 1027–1045 (2023). https://doi.org/10.1007/s10534-023-00503-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-023-00503-y