Abstract
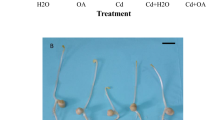
Recent results discovered the protective roles of methane (CH4) against oxidative stress in animals. However, the possible physiological roles of CH4 in plants are still unknown. By using physiological, histochemical and molecular approaches, the beneficial role of CH4 in germinating alfalfa seeds upon copper (Cu) stress was evaluated. Endogenous production of CH4 was significantly increased in Cu-stressed alfalfa seeds, which was mimicked by 0.39 mM CH4. The pretreatment with CH4 significantly alleviated the inhibition of seed germination and seedling growth induced by Cu stress. Cu accumulation was obviously blocked as well. Meanwhile, α/β amylase activities and sugar contents were increased, all of which were consistent with the alleviation of seed germination inhibition triggered by CH4. The Cu-triggered oxidative stress was also mitigated, which was confirmed by the decrease of lipid peroxidation and reduction of Cu-induced loss of plasma membrane integrity in CH4-pretreated alfalfa seedlings. The results of antioxidant enzymes, including ascorbate peroxidase (APX), superoxide dismutase (SOD), catalase (CAT), and guaiacol peroxidase (POD) total or isozymatic activities, and corresponding transcripts (APX1/2, Cu/Zn SOD and Mn-SOD), indicated that CH4 reestablished cellular redox homeostasis. Further, Cu-induced proline accumulation was partly impaired by CH4, which was supported by the alternation of proline metabolism. Together, these results indicated that CH4 performs an advantageous effect on the alleviation of seed germination inhibition caused by Cu stress, and reestablishment of redox homeostasis mainly via increasing antioxidant defence.








Similar content being viewed by others
Abbreviations
- APX:
-
Ascorbate peroxidase
- ASC:
-
Ascorbic acid
- CAT:
-
Catalase
- CH4 :
-
Methane
- FID:
-
Flame ionization detector
- GC:
-
Gas chromatograph
- H2O2 :
-
Hydrogen peroxide
- NBT:
-
Nitroblue tetrazolium
- O2 − :
-
Superoxide anion radicals
- P5C:
-
∆1-pyrroline-5-carboxylate
- P5CS:
-
∆1-pyrroline-5-carboxylate synthetase
- PDH:
-
Proline dehydrogenase
- POD:
-
Guaiacol peroxidase
- PVP:
-
Polyvinylpyrrolidone
- ROS:
-
Reactive oxygen species
- SOD:
-
Superoxide dismutase
- TBA:
-
Thiobarbituric acid
- TBARS:
-
Thiobarbituric acid reactive substances
- TCA:
-
Trichloroacetic acid
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126. doi:10.1016/S0076-6879(84)05016-3
Alaoui-Sossé B, Genet P, Vinit-Dunand F et al (2004) Effect of copper on growth in cucumber plants (Cucumis sativus) and its relationships with carbohydrate accumulation and changes in ion contents. Plant Sci 166:1213–1218. doi:10.1016/j.plantsci.2003.12.032
Anderson CR, Wu WH (2005) Analysis of carbon monoxide in commercially treated tuna (Thunnus spp) and mahi-mahi (Coryphaena hippurus) by gas chromatography/mass spectrometry. J Agric Food Chem 53:7019–7023. doi:10.1021/jf0514266
Ayliffe MA, Mitchell HJ, Deuschle K et al (2005) Comparative analysis in cereals of a key proline catabolism gene. Mol Genet Genomics 274:494–505. doi:10.1007/s00438-005-0048-x
Barker J, Curtis S, Hogsett O et al (1986) Safety in swine production systems. Pork Industry Handbook 104
Bassi R, Sharma SS (1993) Proline accumulation in wheat seedlings exposed to zinc and copper. Phytochem 33:1339–1342. doi:10.1016/0031-9422(93)85086-7
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water stress studies. Plant Soil 39:205–207. doi:10.1007/BF00018060
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287. doi:10.1016/0003-2697(71)90370-8
Bernardi C, Chiesa LM, Soncin S et al (2008) Determination of carbon monoxide in tuna by gas chromatography with micro-thermal conductivity detector. J Chromatogr Sci 46:392–394. doi:10.1093/chromsci/46.5.392
Boros M, Ghyczy M, Érces D et al (2012) The anti-inflammatory effects of methane. Crit Care Med 40:1269–1278. doi:10.1097/CCM.0b013e31823dae05
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. doi:10.1016/0003-2697(76)90527-3
Bruhn D, Møller IM, Mikkelsen TN et al (2012) Terrestrial plant methane production and emission. Physiol Plant 144:201–209. doi:10.1111/j.1399-3054.2011.01551.x
Bruhn D, Mikkelsen TN, Rolsted MMM et al (2014) Leaf surface wax is a source of plant methane formation under UV radiation and in the presence of oxygen. Plant Biol 16:512–516. doi:10.1111/plb.12137
Burkhead JL, Gogolin Reynolds KA, Abdel-Ghany SE et al (2009) Copper homeostasis. New Phytol 182:799–816. doi:10.1111/j.1469-8137.2009.02846.x
Chen CT, Chen TH, Lo KF et al (2004) Effects of proline on copper transport in rice seedlings under excess copper stress. Plant Sci 166:103–111. doi:10.1016/j.plantsci.2003.08.015
Chen Z, Cuin TA, Zhou M et al (2007) Compatible solute accumulation and stress-mitigating effects in barely genotypes contrasting in their salt tolerance. J Exp Bot 58:4245–4255. doi:10.1093/jxb/erm284
Chen O, Ye Z, Cao Z et al (2016a) Methane attenuates myocardial ischemia injury in rats through anti-oxidative, anti-apoptotic and anti-inflammatory actions. Free Radic Biol Med 90:1–11. doi:10.1016/j.freeradbiomed.2015.11.017
Chen Z, Yu L, Wang X et al (2016b) Changes of phenolic profiles and antioxidant activity in canaryseed (Phalaris canariensis L.) during germination. Food Chem 194:608–618. doi:10.1016/j.foodchem.2015.08.060
Conrad R (2005) Quantification of methanogenic pathways using stable carbon isotopic signatures: a review and a proposal. Org Geochem 36:739–752. doi:10.1016/j.orggeochem.2004.09.006
Conrad R (2009) The global methane cycle: recent advances in understanding the microbial processes involved. Environ Microbiol Rep 1:285–292. doi:10.1111/j.1758-2229.2009.00038.x
Cui W, Gao C, Fang P et al (2013) Alleviation of cadmium toxicity in Medicago sativa by hydrogen-rich water. J Hazard Mater 260:715–724. doi:10.1016/j.jhazmat.2013.06.032
Cui W, Qi F, Zhang Y et al (2015) Methane-rich water induces cucumber adventitious rooting through heme oxygenase1/carbon monoxide and Ca2+ pathways. Plant Cell Rep 34:435–445. doi:10.1007/s00299-014-1723-3
Das D, Veziroǧlu TN (2001) Hydrogen production by biological processes: a survey of literature. Int J Hydrogen Energ 26:13–28. doi:10.1016/S0360-3199(00)00058-6
El-Tayeb MA, El-Enany AE, Ahmed NL (2006) Salicylic acid-induced adaptive response to copper stress in sunflower (Helianthus annuus L). Plant Growth Regul 50:191–199. doi:10.1007/s10725-006-9118-2
Foyer CH, Shigeoka S (2011) Understanding oxidative stress and antioxidant functions to enhance photosynthesis. Plant Physiol 155:93–100. doi:10.1104/pp.110.166181
Guenther A, Geron C, Pierce T et al (2000) Natural emissions of non-methane volatile organic compounds, carbon monoxide, and oxides of nitrogen from North America. Atmos Environ 34:2205–2230. doi:10.1016/S1352-2310(99)00465-3
Han Y, Zhang J, Chen X, Gao Z, Xuan W, Xu S, Ding X, Shen WB (2008) Carbon monoxide alleviates cadmium-induced oxidative damage by modulating glutathione metabolism in the roots of Medicago sativa. New Phytol 177:155–166. doi:10.1111/j.1469-8137.2007.02251.x
Hare PD, Cress WA (1997) Metabolic implications of stress-induced proline accumulation in plants. Plant Growth Regul 21:79–102. doi:10.1023/A:1005703923347
He R, Wang L, Zhu J et al (2016) Methane-rich saline protects against concanavalin A-induced autoimmune hepatitis in mice through anti-inflammatory and anti-oxidative pathways. Biochem Biophys Res Commun 470:22–28. doi:10.1016/j.bbrc.2015.12.080
Hellmann H, Funck D, Rentsch D et al (2000) Hypersensitivity of an Arabidopsis sugar signaling mutant toward exogenous proline application. Plant Physiol 122:357–368. doi:10.1104/pp.122.2.357
Hu KD, Hu LY, Li YH et al (2007) Protective roles of nitric oxide on germination and antioxidant metabolism in wheat seeds under copper stress. Plant Growth Regul 53:173–183. doi:10.1007/s10725-007-9216-9
Huang BK, Xu S, Xuan W et al (2006) Carbon monoxide alleviates salt-induced oxidative damage in wheat seedling leaves. J Integr Plant Biol 48:249–254. doi:10.1111/j.1744-7909.2006.00220.x
Inoue H, Date Y, Kohashi K et al (1996) Determination of total hydroxyproline and proline in human serum and urine by HPLC with fluorescence detection. Biol Pharm Bull 19:163–166. doi:10.1248/bpb.19.163
Janas KM, Zielińska-Tomaszewska J, Rybaczek D et al (2010) The impact of copper ions on growth, lipid peroxidation, and phenolic compound accumulation and localization in lentil (Lens culinaris Medic.) seedlings. J Plant Physiol 167:270–276. doi:10.1016/j.jplph.2009.09.016
Jia Z, Cai Z, Xu H et al (2001) Effect of rice plants on CH4 production, transport, oxidation and emission in rice paddy soil. Plant Soil 230:211–221. doi:10.1023/A:1010366631538
Jiang WD, Qu B, Feng L et al (2016) Histidine prevents Cu-induced oxidative stress and the associated decreases in mRNA from encoding tight junction proteins in the intestine of Grass Carp (Ctenopharyngodon idella). PLoS ONE 11:e0157001. doi:10.1371/journal.pone.0157001
Jin Q, Zhu K, Cui W et al (2013) Hydrogen gas acts as a novel bioactive molecule in enhancing plant tolerance to paraquat-induced oxidative stress via the modulation of heme oxygenase-1 signalling system. Plant Cell Environ 36:956–969. doi:10.1111/pce.12029
Keppler F, Boros M, Frankenberg C et al (2009) Methane formation in aerobic environments. Environ Chem 6:459–465. doi:10.1071/EN09137
Ku HM, Tan CW, Su YS et al (2012) The effect of water deficit and excess copper on proline metabolism in Nicotiana benthamiana. Biol Plant 56:337–343. doi:10.1007/s10535-012-0095-1
Lenhart K, Bunge M, Ratering S et al (2012) Evidence for methane production by saprotrophic fungi. Nat Commun 3:1046. doi:10.1038/ncomms2049
Liu DL, An ZG, Mao ZJ et al (2016a) Enhanced heavy metal tolerance and accumulation by transgenic sugar beets expressing Streptococcus thermophilus StGCS-GS in the presence of Cd, Zn and Cu alone or in combination. PLoS ONE 10:e0128824. doi:10.1371/journal.pone.0128824
Liu L, Sun Q, Wang R et al (2016b) Methane attenuates retinal ischemia/reperfusion injury via anti-oxidative and anti-apoptotic pathways. Brain Res 1646:327–333. doi:10.1016/j.brainres.2016.05.037
Lombardi L, Sebastiani L (2005) Copper toxicity in Prunus cerasifera: growth and antioxidant enzymes responses of in vitro grown plants. Plant Sci 168:797–802. doi:10.1016/j.plantsci.2004.10.012
Lopez-Carrion AI, Castellano R, Rosales MA et al (2008) Role of nitric oxide under saline stress: implications on proline metabolism. Biol Plant 52:587–591. doi:10.1007/s10535-008-0117-1
Maggio A, Miyazaki S, Veronese P et al (2002) Does proline accumulation play an active role in stress-induced growth reduction? Plant J 31:699–712. doi:10.1046/j.1365-313X.2002.01389.x
Mani S, Van De Cotte B, Van Montagu M et al (2002) Altered levels of proline dehydrogenase cause hypersensitivity to proline and its analogs in Arabidopsis. Plant Physiol 128:73–83. doi:10.1104/pp.010572
Messenger DJ, McLeod AR, Fry SC (2009) Reactive oxygen species in aerobic methane formation from vegetation. Plant Signal Behav 4:629–630. doi:10.1111/j.1365-3040.2008.01892.x
Metwally A, Finkemeier I, Georgi M et al (2003) Salicylic acid alleviates the cadmium toxicity in barley seedlings. Plant Physiol 132:272–281. doi:10.1104/pp.102.018457
Patui S, Clincon L, Peresson C et al (2014) Lipase activity and antioxidant capacity in coffee (Coffea arabica L.) seeds during germination. Plant Sci 219–220:19–25. doi:10.1016/j.plantsci.2013.12.014
Planchet E, Jagadis Gupta K, Sonoda M et al (2005) Nitric oxide emission from tobacco leaves and cell suspensions: rate limiting factors and evidence for the involvement of mitochondrial electron transport. Plant J 41:732–743. doi:10.1111/j.1365-313X.2005.02335.x
Renwick GM, Giumarro C, Siegel SM (1964) Hydrogen metabolism in higher plants. Plant Physiol 39:303–306. doi:10.1104/pp.39.3.303
Rockel P, Strube F, Rockel A et al (2002) Regulation of nitric oxide (NO) production by plant nitrate reductase in vivo and in vitro. J Exp Bot 53:103–110. doi:10.1093/jexbot/53.366.103
Seiler W, Holzapfel-Pschorn A, Conrad R et al (1983) Methane emission from rice paddies. J Atmos Chem 1:241–268. doi:10.1007/BF00058731
Sharma SS, Dietz K (2009) The relationship between metal toxicity and cellular redox imbalance. Trends Plant Sci 14:43–50. doi:10.1016/j.tplants.2008.10.007
Sheldon AR, Menzies NW (2005) The effect of copper toxicity on the growth and root morphology of Rhodes grass (Chloris gayana Knuth.) in resin buffered solution culture. Plant Soil 278:341–349. doi:10.1007/s11104-005-8815-3
Tarr MA, Miller WL, Zepp RG (1995) Direct carbon monoxide photoproduction from plant matter. J Geophys Res 100:11403–11413. doi:10.1029/94JD03324
Tripathi BN, Singh V, Ezaki B et al (2013) Mechanism of Cu- and Cd-induced proline hyperaccumulation in triticum aestivum (wheat). Plant Growth Regul 32:799–808. doi:10.1007/s00344-013-9343-7
Verbruggen N, Hermans C (2008) Proline accumulation in plants: a review. Amino Acids 35:753–759. doi:10.1007/s00726-008-0061-6
Wang SH, Zhang H, Zhang Q et al (2011) Copper-induced oxidative stress and responses of the antioxidant system in roots of Medicago sativa. J Agron Crop Sci 197:418–429. doi:10.1111/j.1439-037X.2011.00476.x
Wang Y, Li L, Cui W et al (2012) Hydrogen sulfide enhances alfalfa (Medicago sativa) tolerance against salinity during seed germination by nitric oxide pathway. Plant Soil 351:107–119. doi:10.1007/s11104-011-0936-2
Wang ZP, Chang SX, Chen H et al (2013) Widespread non-microbial methane production by organic compounds and the impact of environmental stresses. Earth-Sci Rev 127:193–202. doi:10.1016/j.earscirev.2013.10.001
Wishkerman A, Greiner S, Ghyczy M et al (2011) Enhanced formation of methane in plant cell cultures by inhibition of cytochrome c oxidase. Plant Cell Environ 34:457–464. doi:10.1111/j.1365-3040.2010.02255.x
Xie Y, Ling T, Han Y et al (2008) Carbon monoxide enhances salt tolerance by nitric oxide-mediated maintenance of ion homeostasis and up-regulation of antioxidant defence in wheat seedling roots. Plant Cell Environ 31:1864–1881. doi:10.1111/j.1365-3040.2008.01888.x
Xu WF, Shi WM, Liu F et al (2008) Enhanced zinc and cadmium tolerance and accumulation in transgenic Arabidopsis plants constitutively overexpressing a barley gene (HvAPX1) that encodes a peroxisomal ascorbate peroxidase. Botany 86:567–575. doi:10.1139/B08-025
Xu S, Zhu S, Jiang Y et al (2013) Hydrogen-rich water alleviates salt stress in rice during seed germination. Plant Soil 370:47–57. doi:10.1007/s11104-013-1614-3
Yamamoto Y, Kobayashi Y, Matsumoto H (2001) Lipid peroxidation is an early symptom triggered by aluminum, but not the primary cause of elongation inhibition in pea roots. Plant Physiol 125:199–208. doi:10.1104/pp.125.1.199
Yonar ME, Ispir U, Mise Yonar S et al (2015) Effect of copper sulphate on the antioxidant parameters in the rainbow trout fry, Oncorhynchu mykiss. Cell Mol Biol (Noisy-le-grand) 62:55–58
Yruela I (2005) Copper in plants. Braz J Plant Physiol 17:145–156. doi:10.1590/S1677-04202005000100012
Zhang CS, Lu Q, Verma DPS (1995) Removal of feedback inhibition of ∆1-pyrroline-5-carboxylate synthetase, a bifunctional enzyme catalyzing the first two steps of proline biosynthesis in plants. J Biol Chem 270:20491–20496. doi:10.1074/jbc.270.35.20491
Zhang H, Hu LY, Hu KD et al (2008) Hydrogen sulfide promotes wheat seed germination and alleviates oxidative damage against copper stress. J Integr Plant Biol 50:1518–1529. doi:10.1111/j.1744-7909.2008.00769.x
Zhang HJ, Zhang N, Yang RC et al (2014) Melatonin promotes seed germination under high salinity by regulating antioxidant systems, ABA and GA4 interaction in cucumber (Cucumis sativus L.). J Pineal Res 57:269–279. doi:10.1111/jpi.12167
Zhu K, Cui W, Dai C et al (2016) Methane-rich water alleviates NaCl toxicity during alfalfa seed germination. Environ Exper Bot 129:37–47. doi:10.1016/j.envexpbot.2015.11.013
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (J1210056 and J1310015) and the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
Author information
Authors and Affiliations
Corresponding author
Additional information
Muhammad Kaleem Samma and Heng Zhou have contributed equally to this article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Samma, M.K., Zhou, H., Cui, W. et al. Methane alleviates copper-induced seed germination inhibition and oxidative stress in Medicago sativa . Biometals 30, 97–111 (2017). https://doi.org/10.1007/s10534-017-9989-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-017-9989-x