Abstract
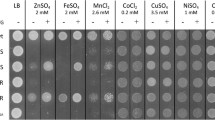
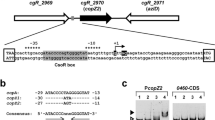
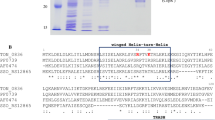
The pathway of copper entry into Escherichia coli is still unknown. In an attempt to shed light on this process, a lux-based biosensor was utilized to monitor intracellular copper levels in situ. From a transposon-mutagenized library, strains were selected in which copper entry into cells was reduced, apparent as clones with reduced luminescence when grown in the presence of copper (low-glowers). One low-glower had a transposon insertion in the comR gene, which encodes a TetR-like transcriptional regulator. The mutant strain could be complemented by the comR gene on a plasmid, restoring luminescence to wild-type levels. ComR did not regulate its own expression, but was required for copper-induction of the neighboring, divergently transcribed comC gene, as shown by real-time quantitative PCR and with a promoter-lux fusion. The purified ComR regulator bound to the promoter region of the comC gene in vitro and was released by copper. By membrane fractionation, ComC was shown to be localized in the outer membrane. When grown in the presence of copper, ∆comC cells had higher periplasmic and cytoplasmic copper levels, compared to the wild-type, as assessed by the activation of the periplasmic CusRS sensor and the cytoplasmic CueR sensor, respectively. Thus, ComC is an outer membrane protein which lowers the permeability of the outer membrane to copper. The expression of ComC is controlled by ComR, a novel, TetR-like copper-responsive repressor.






Similar content being viewed by others
References
Ausubel RM, Brent R, Kingston RE, Moore DD, Smith JA, Struhl K (1995) Current protocols in molecular biology. John, New York
Bishop RE, Gibbons HS, Guina T, Trent MS, Miller SI, Raetz CR (2000) Transfer of palmitate from phospholipids to lipid A in outer membranes of gram-negative bacteria. EMBO J 19:5071–5080
Bordoli L, Kiefer F, Arnold K, Benkert P, Battey J, Schwede T (2009) Protein structure homology modeling using SWISS-MODEL workspace. Nat Protoc 4:1–13
Brown CT, Callan CG Jr (2004) Evolutionary comparisons suggest many novel cAMP response protein binding sites in Escherichia coli. Proc Natl Acad Sci USA 101:2404–2409
Changela A, Chen K, Xue Y, Holschen J, Outten CE, O’Halloran TV, Mondragon A (2003) Molecular basis of metal-ion selectivity and zeptomolar sensitivity by CueR. Science 301:1383–1387
Egler M, Grosse C, Grass G, Nies DH (2005) Role of the extracytoplasmic function protein family sigma factor RpoE in metal resistance of Escherichia coli. J Bacteriol 187:2297–2307
Franke S, Grass G, Rensing C, Nies DH (2003) Molecular analysis of the copper-transporting efflux system CusCFBA of Escherichia coli. J Bacteriol 185:3804–3812
Grass G, Thakali K, Klebba PE, Thieme D, Muller A, Wildner GF, Rensing C (2004) Linkage between catecholate siderophores and the multicopper oxidase CueO in Escherichia coli. J Bacteriol 186:5826–5833
Harrison MD, Jones CE, Solioz M, Dameron CT (2000) Intracellular copper routing: the role of copper chaperones. Trends Biochem Sci 25:29–32
Kloosterman TG, van der Kooi-Pol MM, Bijlsma JJ, Kuipers OP (2007) The novel transcriptional regulator SczA mediates protection against Zn2+ stress by activation of the Zn2+-resistance gene czcD in Streptococcus pneumoniae. Mol Microbiol 65:1049–1063
Maurer LM, Yohannes E, Bondurant SS, Radmacher M, Slonczewski JL (2005) pH regulates genes for flagellar motility, catabolism, and oxidative stress in Escherichia coli K-12. J Bacteriol 187:304–319
Munson GP, Lam DL, Outten FW, O’Halloran TV (2000) Identification of a copper-responsive two-component system on the chromosome of Escherichia coli K-12. J Bacteriol 182:5864–5871
Nies DH (2003) Efflux-mediated heavy metal resistance in prokaryotes. FEMS Microbiol Rev 27:313–339
Orth P, Schnappinger D, Hillen W, Saenger W, Hinrichs W (2000) Structural basis of gene regulation by the tetracycline inducible Tet repressor-operator system. Nat Struct Biol 7:215–219
Outten FW, Outten CE, Hale J, O’Halloran TV (2000) Transcriptional activation of an Escherichia coli copper efflux regulon by the chromosomal MerR homologue. CueR J Biol Chem 275:31024–31029
Outten FW, Huffman DL, Hale JA, O’Halloran TV (2001) The independent cue and cus systems confer copper tolerance during aerobic and anaerobic growth in Escherichia coli. J Biol Chem 276:30670–30677
Parkhill J, Ansari AZ, Wright JG, Brown NL, O’Halloran TV (1993) Construction and characterization of a mercury-independent MerR activator (MerRAC): transcriptional activation in the absence of Hg(II) is accompanied by DNA distortion. EMBO J 12:413–421
Petersen C, Moller LB (2000) Control of copper homeostasis in Escherichia coli by a P-type ATPase, CopA, and a MerR-like transcriptional activator, CopR. Gene 261:289–298
Pomposiello PJ, Bennik MH, Demple B (2001) Genome-wide transcriptional profiling of the Escherichia coli responses to superoxide stress and sodium salicylate. J Bacteriol 183:3890–3902
Rae TD, Schmidt PJ, Pufahl RA, Culotta VC, O’Halloran TV (1999) Undetectable intracellular free copper: the requirement of a copper chaperone for superoxide dismutase. Science 284:805–808
Rensing C, Grass G (2003) Escherichia coli mechanisms of copper homeostasis in a changing environment. FEMS Microbiol Rev 27:197–213
Rensing C, Fan B, Sharma R, Mitra B, Rosen BP (2000) CopA: an Escherichia coli Cu(I)-translocating P-type ATPase. Proc Natl Acad Sci USA 97:652–656
Richmond CS, Glasner JD, Mau R, Jin H, Blattner FR (1999) Genome-wide expression profiling in Escherichia coli K-12. Nucleic Acids Res 27:3821–3835
Rogowsky PM, Close TJ, Chimera JA, Shaw JJ, Kado CI (1987) Regulation of the vir genes of Agrobacterium tumefaciens plasmid pTiC58. J Bacteriol 169:5101–5112
Rosen J, Hellenas KE (2002) Analysis of acrylamide in cooked foods by liquid chromatography tandem mass spectrometry. Analyst 127:880–882
Singh SK, Grass G, Rensing C, Montfort WR (2004) Cuprous oxidase activity of CueO from Escherichia coli. J Bacteriol 186:7815–7817
Stoyanov JV, Hobman JL, Brown NL (2001) CueR (YbbI) of Escherichia coli is a MerR family regulator controlling expression of the copper exporter CopA. Mol Microbiol 39:502–512
Stoyanov JV, Magnani D, Solioz M (2003) Measurement of cytoplasmic copper, silver, and gold with a lux biosensor shows copper and silver, but not gold, efflux by the CopA ATPase of Escherichia coli. FEBS Lett 546:391–394
Towbin H, Staehelin T, Gordon J (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci USA 76:4350–4354
Yoshida Y, Furuta S, Niki E (1993) Effects of metal chelating agents on the oxidation of lipids induced by copper and iron. Biochim Biophys Acta 1210:81–88
Yu Z, Reichheld SE, Savchenko A, Parkinson J, Davidson AR (2010) A comprehensive analysis of structural and sequence conservation in the TetR family transcriptional regulators. J Mol Biol 400:847–864
Zhang XS, Garcia-Contreras R, Wood TK (2007) YcfR (BhsA) influences Escherichia coli biofilm formation through stress response and surface hydrophobicity. J Bacteriol 189:3051–3062
Zheng M, Wang X, Templeton LJ, Smulski DR, LaRossa RA, Storz G (2001) DNA microarray-mediated transcriptional profiling of the Escherichia coli response to hydrogen peroxide. J Bacteriol 183:4562–4570
Acknowledgments
We thank Thomas Weber for expert technical assistance and the National BioResource Project (NIG, Japan) for providing strain JD27538. This work was supported by Grant 3100A0_122551 from the Swiss National Foundation and grants from the International Copper Association and the Swiss State Secretary for Education & Research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Mélanie Mermod and David Magnani have contributed equally to this work
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mermod, M., Magnani, D., Solioz, M. et al. The copper-inducible ComR (YcfQ) repressor regulates expression of ComC (YcfR), which affects copper permeability of the outer membrane of Escherichia coli . Biometals 25, 33–43 (2012). https://doi.org/10.1007/s10534-011-9510-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-011-9510-x