Abstract
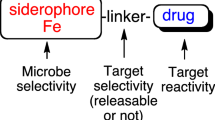
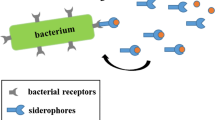
The recent rise in drug resistance found amongst community acquired infections has sparked renewed interest in developing antimicrobial agents that target resistant organisms and limit the natural selection of immune variants. Recent discoveries have shown that iron uptake systems in bacteria and fungi are suitable targets for developing such therapeutic agents. The use of siderophore-drug conjugates as “Trojan Horse” drug delivery agents has attracted particular interest in this area. This review will discuss efforts in our research group to study the salmycin class of “Trojan Horse” antibiotics. Inspired by the natural design of the salmycins, a series of desferridanoxamine-antibiotic conjugates were synthesized and tested in microbial growth inhibition assays. The results of these studies will be related to understanding the role of drug release in siderophore-mediated drug delivery with implications for future siderophore-drug conjugate design.











Similar content being viewed by others
References
Ash C (1996) Antibiotic resistance: the new apocalypse? Trends Microbiol 4:371–372. doi:10.1016/0966-842X(96)30028-0
Benz G, Schröder T, Kurz J, Wünsche C, Karl W, Steffen GJ, Pfitzner J, Schmidt D (1982) Konstitution der deferriform der albomycine δ1, δ2 und ε. Angew Chem Int Ed 94:552–553
Bergeron RJ, Pegram JJ (1988) An efficient total synthesis of desferrioxamine B. J Org Chem 53:3131–3134. doi:10.1021/jo00249a001
Bickel H, Gäumann E, Nussberg G, Reusser P, Vischer E, Voser W, Wettstein A, Zähner H (1960) Stoffwechselprodukte von Actinomyceten. 25 Mitteilung: über die isolierung und charakterisierung der ferrimycine A1 und A2, neuer antibiotika der sideromycin-gruppe. Helv Chim Acta 43:2105–2118. doi:10.1002/hlca.19600430730
Bickel H, Mertens P, Prelog V, Seibl J, Walser A (1965) Constitution of ferrimycin A1. Antimicrob Agents Chemother 5:951–957
Braun V (1999) Active transport of siderophore-mimicking antibacterials across the outer membrane. Drug Resist Updat 2:363–369. doi:10.1054/drup.1999.0107
Braun V, Günthner H, Hantke K, Zimmerman L (1983) Intracellular activation of albomycin in Escherichia coli and Salmonella typhimurium. J Bacteriol 156:308–315
Braun V, Pramanik A, Gwinner T (2008) Use of sideromycins as tools and antibiotics. Paper presented at the 6th international biometals symposium, Santiago de Compostela, Spain, 14–18 July 2008
Brochu A, Brochu N, Nicas TI, Parr TR, Minnick AA, Dolence EK, McKee JA, Miller MJ, Lavoie MC, Malouin F (1992) Modes of action and inhibitory activities of new siderophore-β-lactam conjugates that use specific iron uptake pathways for entry into bacteria. Antimicrob Agents Chemother 36:2166–2175
Budzikiewicz H (2001) Siderophore-antibiotic conjugates used as trojan horses against Pseudomonas aeruginosa. Curr Top Med Chem 1:73–82. doi:10.2174/1568026013395524
Challis GL, Hopwood DA (2003) Synergy and contingency as driving forces for the evolution of multiple secondary metabolite production by Streptomyces species. Proc Natl Acad Sci USA 100:14555–14561. doi:10.1073/pnas.1934677100
Clarke TE, Braun V, Winkelmann G, Tari LW, Vogel HJ (2002) X-ray crystallographic structures of the Eschericia coli periplasmic protein FhuD bound to hydroxamate-type siderophores and the antibiotic albomycin. J Biol Chem 277:13966–13972. doi:10.1074/jbc.M109385200
Codd R (2008) Traversing the coordination chemistry and chemical biology of hydroxamic acids. Coord Chem Rev 252:1387–1408. doi:10.1016/j.ccr.2007.08.001
Dong L, Roosenberg JM, Miller MJ (2002) Total synthesis of deferrisalmycin B. J Am Chem Soc 124:15001–15005. doi:10.1021/ja028386w
Ferguson AD, Braun V, Fiedler HP, Coulton JW, Diederichs K, Welte W (2000) Crystal structure of the antibiotic albomycin in complex with the outer membrane transporter FhuA. Protein Sci 9:956–963
Ferreras JA, Ryu JS, Di Lello F, Tan DS, Quadri LEN (2005) Small molecule inhibition of siderophore biosynthesis in Mycobacterium tuberculosis and Yersinia pestis. Nat Chem Biol 1:219–232. doi:10.1038/nchembio706
Gause GF (1955) Recent studies on albomycin, a new antibiotic. BMJ 12:1177–1179
Guerinot ML (1994) Microbial iron transport. Annu Rev Microbiol 48:743–772. doi:10.1146/annurev.mi.48.100194.003523
Hartmann A, Fiedler HP, Braun V (1979) Uptake and conversion of the antibiotic albomycin by Eschericia coli K-12. Eur J Biochem 99:517–524. doi:10.1111/j.1432-1033.1979.tb13283.x
Grand Challenges in Global Health (2008) Create drugs and delivery systems to limit drug resistance. http://www.gcgh.org/LimitDrugResistance/Topics/DoNotGenerateResistance/Pages/default.aspx. Cited 11 Nov 2008
Heinisch L, Wittmann S, Stoiber T, Berg A, Ankel-Fuchs D, Möllmann U (2002) Highly antibacterial active amnioacyl penicillin conjugates with bis-catecholate siderophores based on secondary diamino acids and related compounds. J Med Chem 45:3032–3040. doi:10.1021/jm010546b
Heinisch L, Wittmann S, Stoiber T, Scherlitz-Hoffmann I, Ankel-Fuchs D, Möllmann U (2003) New tris- and tetrakis-catecholate siderophores based on polyazaalkanoic acids and their β-lactam conjugates. Arzneim-Forschung Drug Res 53:188–195
Hennard C, Truong QC, Desnottes JF, Paris JM, Moreau NJ, Abdallah MA (2001) Synthesis and activities of pyoverdin–quinolone adducts: a prospective approach to a specific therapy against Pseudomonas aeruginosa. J Med Chem 44:2139–2151. doi:10.1021/jm990508g
Ho WH, Wong HNC (1995) Chiral liquid crystalline compounds from d-(+)-glucose. Tetrahedron 51:7373–7388. doi:10.1016/0040-4020(95)00386-M
Huber P, Leuenberger H, Keller-Schierlein W (1986) Danoxamin, der eisenbindende teil des sideromycin-antibioticums danomycin. Helv Chim Acta 69:236–245. doi:10.1002/hlca.19860690128
Jarvis LM (2008) An uphill battle. C E News 86:15–20
Klare I, Heier H, Claus H, Reissbrodt R, Witte W (1995) VanA-mediated high-level glycopeptides resistance in Enterococcus faecium from animal husbandry. FEMS Microbiol Lett 125:165–172. doi:10.1111/j.1574-6968.1995.tb07353.x
Krewulak KD, Vogel HJ (2008) Structural biology of bacterial iron uptake. Biochim Biophys Acta 1778:1781–1804. doi:10.1016/j.bbamem.2007.07.026
Marshall E (2008) The bacteria fight back. Science 321:356–364. doi:10.1126/science.321.5887.356
Miethke M, Marahiel MA (2007) Siderophore-based iron acquisition and pathogen control. Microbiol Mol Biol Rev 71:413–451. doi:10.1128/MMBR.00012-07
Miller MJ, Malouin F (1993) Microbial iron chelators as drug delivery agents: the rational design and synthesis of siderophore-drug conjugates. Acc Chem Res 26:241–249. doi:10.1021/ar00029a003
Miller MJ, Zhu H, Xu Y, Wu C, Walz AJ, Vergne A, Roosenberg JM, Moraski G, Minnick AA, McKee-Dolence J, Hu J, Fennell K, Dolence EK, Dong L, Franzblau S, Malouin F, Möllmann U (2009) Utilization of microbial iron assimilation processes for the development of new antibiotics and inspiration for the design of new anticancer agents. Biometals 22:61–75
Mitscher LA (2008) Coevolution: mankind and microbes. J Nat Prod 71:497–509. doi:10.1021/np078017j
Möllmann U, Dong L, Vertesy L, Miller MJ (2004) Salmycins—natural siderophore-drug conjugates: Prospects for modification and investigation based on successful total synthesis. Paper presented at the 2nd international Biometals symposium, Garmisch-Partenkirchen, Germany, 3–5 Sept 2004
Möllmann U, Heinisch L, Bauernfeind A, Thilo K, Ankel-Fuchs D (2009) Siderophores as drug delivery agents: application of the “Trojan Horse” strategy. Biometals. doi:10.1007/s10534-009-9219-2
Nathan C (2004) Antibiotics at the crossroads. Nature 431:899–902. doi:10.1038/431899a
Neilands JB (1995) Siderophores: structure and function of microbial iron transport compounds. J Biol Chem 270:26723–26726
Neres J, Labello NP, Somu RV, Boshoff HI, Wilson DJ, Vannada J, Chen L, Barry CE, Bennet EM, Aldich CC (2008) Inhibition of siderophore biosynthesis in Mycobacterium tuberculosis with nucleoside bisubstrate analogues: structure–activity relationships of the nucleobase domain of 5′-O-[N-(salicyl)sulfamoyl]adenosine. J Med Chem 51:5349–5370. doi:10.1021/jm800567v
Nikaido H, Nikaido K, Harayama S (1991) Identification and characterization of porins in Pseudomonas aeruginosa. J Biol Chem 266:770–779
Nolan EM, Walsh CT (2008) Investigations of the MceIJ-catalyzed posttranslational modification of the microcin E492 C-terminus: linkage of ribosomal and nonribosomal peptides to form “Trojan Horse” antibiotics. Biochemistry 47:9289–9299. doi:10.1021/bi800826j
Ogawa T, Kaburagi T (1982) Synthesis of a branched d-glucotetraose, the repeating unit of the extracellular polysaccharides of Grifola umbellate, Sclerotinia libertiana, Porodisculus pendulus, and Schizophyllum commune fries. Carbohydr Res 103:53–64. doi:10.1016/S0008-6215(82)80007-4
Pramanik A, Braun V (2006) Albomycin uptake via a ferric hydroxamate transport system of Streptococcus pneumoniae R6. J Bacteriol 188:3878–3886. doi:10.1128/JB.00205-06
Pramanik A, Stroeher UH, Krejci J, Standish AJ, Bohn E, Paton JC, Autenrieth IB, Braun V (2007) Albomycin is an effective antibiotic, as exemplified with Yersinia enterocolitica and Streptococcus pneumoniae. Int J Med Microbiol 297:459–469. doi:10.1016/j.ijmm.2007.03.002
Quadri LEN (2007) Strategic paradigm shifts in the antimicrobial drug discovery process of the 21st century. Infect Disord Drug Targets 7:230–237. doi:10.2174/187152607782110040
Ratledge CL, Dover G (2000) Iron metabolism in pathogenic bacteria. Annu Rev Microbiol 54:881–941. doi:10.1146/annurev.micro.54.1.881
Richmond MH, Clark DC, Wotton S (1976) Indirect method for assessing the penetration of beta-lactamase-nonsusceptible penicillins and cephalosporins in Eschericia coli. Antimicrob Agents Chemother 10:215–218
Rivault F, Liébert C, Burger A, Hoegy F, Abdallah MA, Schalk IJ, Mislin GLA (2007) Synthesis of pyochelin–norfloxacin conjugates. Bioorg Med Chem Lett 17:640–644. doi:10.1016/j.bmcl.2006.11.005
Rook G (2008) Anti-mycobacterial mycobactin-linked glyconanoparticles. In: Grants awarded. Grand Challenges in Global Health. http://www.gcgh.org/explorations/Pages/GrantsAwarded.aspx. Cited 11 Nov 2008
Roosenberg JM, Miller MJ (2000) Total synthesis of the siderophore danoxamine. J Org Chem 65:4833–4838. doi:10.1021/jo000050m
Roosenberg JM, Lin Y-M, Lu Y, Miller MJ (2000) Studies and syntheses of siderophores, microbial iron chelators, and analogs as potential drug delivery agents. Curr Med Chem 7:159–197
Sackmann W, Preusser P, Neipp L, Kradolfer F, Gross F (1962) Ferrimycin A, a new iron containing antibiotic. Antibiot Chemother 12:34–45
Schumann G, Möllmann U (2001) A screening system for xenosiderophores as potential drug delivery agents in mycobacteria. Antimicrob Agents Chemother 45:1317–1322. doi:10.1128/AAC.45.5.1317-1322.2001
Snapper SB, Melton RE, Mustafa S, Kieser T, Jacobs WR (1990) Isolation and characterization of efficient plasmid transformation mutants of Mycobacterium smegmatis. Mol Microbiol 4:1911–1919. doi:10.1111/j.1365-2958.1990.tb02040.x
Thomas X, Destoumieux-Garzón D, Peduzzi J, Afonso C, Blond A, Birlirakis N, Goulard C, Dubost L, Thai R, Tabet JC, Rebuffat S (2004) Siderophore peptide, a new type of post-translationally modified antibacterial peptide with potent activity. J Biol Chem 279:28233–28242. doi:10.1074/jbc.M400228200
Vértesy L, Aretz W, Fehlhaber H-W, Koger H (1995) Salmycin A–D, Antibiotika aus Streptomycese violaceus, DSM 8286, mit siderophore-aminoglycosid-struktur. Helv Chim Acta 78:46–60. doi:10.1002/hlca.19950780105
Wach JY, Bonazzi S, Gademann K (2008) Antimicrobial surfaces through natural product hybrids. Angew Chem Int Ed 47:7123–7126. doi:10.1002/anie.200801570
Walsh C (2003) Antibiotics: actions, origins, resistance. ASM Press, Washington, DC
Winkelmann G, van der Helm D, Neilands JB (1987) Iron transport in microbes, plants, and animals. VCH Press, Weinheim, pp 1–533
Witte W, Cuny C, Braulke C, Heuck D (1994) Clonal dissemination of two MRSA strains in Germany. Epidemiol Infect 113:67–73
Wittmann S, Schnabelrauch M, Scherlitz-Hoffmann I, Möllmann U, Ankel-Fuchs D, Heinisch L (2002) New synthetic siderophores and their β-lactam conjugates based on amino acids and dipeptides. Bioorg Med Chem 10:1659–1670. doi:10.1016/S0968-0896(02)00044-5
Zähner H, Diddens H, Keller-Schierlein W, Nägeli HU (1977) Some experiments with semisynthetic sideromycins. Jpn J Antibiot 30:S201–S206
Zimmermann W (1980) Penetration of β-lactam antibiotics into their target enzymes in Pseudomonas aeruginosa: comparison of a highly sensitive mutant with its parent strain. Antimicrob Agents Chemother 18:94–100
Acknowledgments
We gratefully acknowledge the National Institutes of Health (NIH) research grants RO1 AI054193, NIH AI 030988, and NIH GM025845 for financial support. We thank Irmgard Heinemann and Uta Wohlfeld for their excellent technical assistance with growth promotion and growth inhibition assays at the HKI. MJM gratefully acknowledges the kind hospitality of the HKI and the University of Notre Dame for a sabbatical opportunity in Jena, Germany. TAW gratefully acknowledges the University of Notre Dame Chemistry-Biochemistry-Biology (CBBI) Interface Program and NIH training grant T32GM075762 for a fellowship and the kind hospitality of the HKI for a research internship opportunity.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wencewicz, T.A., Möllmann, U., Long, T.E. et al. Is drug release necessary for antimicrobial activity of siderophore-drug conjugates? Syntheses and biological studies of the naturally occurring salmycin “Trojan Horse” antibiotics and synthetic desferridanoxamine-antibiotic conjugates. Biometals 22, 633–648 (2009). https://doi.org/10.1007/s10534-009-9218-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-009-9218-3