Abstract
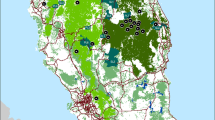
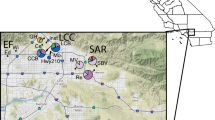
The leopard (Panthera pardus) is facing the threat of continued population decline across its range. In order to inform more effective conservation management programs, genetic information is needed from leopard populations that persist in previously unstudied, isolated and highly fragmented protected areas. The aim of this study was to explore the population structure and genetic diversity of leopard populations across the Mpumalanga province of South Africa. We collected a total of 33 leopard samples from four major locations along a west to east transect across the province. We analysed 17 polymorphic microsatellites and two regions of the mitochondrial genome (NADH-5 and Cytb) to determine the genetic structure of the leopard population in the province. We also calculated genetic diversity indices and explored gene flow in the region. We found that while there is gene flow occurring across the province, the population was genetically structured. We identified two major population units that we describe as ‘West Mpumalanga’ and ‘East Mpumalanga’. Gene flow was moderate between the two populations and we found very high genetic diversity levels compared to other leopard populations previously studied in South Africa. From a conservation perspective, our results show that gene flow is still occurring across seemingly isolated leopard populations that exist in fragmented landscapes, highlighting the importance of all leopard populations in South Africa. Management authorities need to focus conservation efforts on maintaining corridors between regions that are suitable for leopard occupancy and work closely with human settlements to minimise human-leopard conflicts.



Similar content being viewed by others
Data availability
All mtDNA strands have been uploaded to Genbank under the following accession numbers; NADH-5 : OQ132962-OQ132992 & Cytochrome B : OQ117400-OQ117430. Microsatellite data were uploaded to Figshare (https://doi.org/10.6084/m9.figshare.21743852); There are no restrictions on data availability.
References
Abdul-Muneer PM (2014) Application of microsatellite markers in conservation genetics and fisheries management: recent advances in population structure analysis and conservation strategies. Genet Res Int. https://doi.org/10.1155/2014/691759
Anco C, Kolokotronis SO, Henschel P, Cunningham SW, Amato G, Hekkala E (2018) Historical mitochondrial diversity in African leopards (Panthera pardus) revealed by archival museum specimens. Mitochondr DNA A 29:455–473. https://doi.org/10.1080/24701394.2017.1307973
Andersen LW, Fog K, Damgaard C (2004) Habitat fragmentation causes bottlenecks and inbreeding in the European tree frog (Hyla arborea). Proc R Soc B-Biol Sci 271:1293–1302. https://doi.org/10.1098/rspb.2004.2720
Bailey TN (1993) The African leopard: ecology and behavior of a solitary felid. Columbia University Press, New York
Balme GA, Slotow R, Hunter LTB (2010) Edge effects and the impact of non-protected areas in carnivore conservation: leopards in the Phinda-Mkhuze Complex, South Africa. Anim Conserv 13:315–323. https://doi.org/10.1111/j.1469-1795.2009.00342.x
Balme GA, Batchelor A, Britz ND et al (2013) Reproductive success of female leopards Panthera pardus: the importance of top-down processes. Mamm Rev 43:221–237. https://doi.org/10.1111/j.1365-2907.2012.00219.x
Bouckaert R, Heled J, Kuhnert D et al (2014) BEAST 2: a software platform for bayesian evolutionary analysis. PLoS Comput Biol 10:6. https://doi.org/10.1371/journal.pcbi.1003537
Cardillo M, Mace GM, Jones KE et al (2005) Multiple causes of high extinction risk in large mammal species. Science 309:1239–1241. https://doi.org/10.1126/science.1116030
Chakraborty R (1993) Analysis of genetic structure of populations: meaning, methods, and implications. In: Majumder PP (ed) Human population genetics: a centennial tribute to J. B. S. Haldane. Springer, Boston, pp 189–206
Constant NL, Bell S, Hill RA (2015) The impacts, characterisation and management of human-leopard conflict in a multi-use land system in South Africa. Biodivers Conserv 24:2967–2989. https://doi.org/10.1007/s10531-015-0989-2
Darriba D, Taboada GL, Doallo R, Posada D (2012) jModelTest 2: more models, new heuristics and parallel computing. Nat Methods 9:772–772. https://doi.org/10.1038/nmeth.2109
Earl DA, Vonholdt BM (2012) STRUCTURE HARVESTER: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv Genet Resour 4:359–361. https://doi.org/10.1007/s12686-011-9548-7
Epps CW, Castillo JA, Schmidt-Küntzel A, du Preez P, Stuart-Hill G, Jago M, Naidoo R (2013) Contrasting historical and recent gene flow among African buffalo herds in the caprivi strip of Namibia. J Hered 104:172–181. https://doi.org/10.1093/jhered/ess142
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620. https://doi.org/10.1111/j.1365-294X.2005.02553.x
Fattebert J, Dickerson T, Balme G, Slotow R, Hunter L (2013) Long-distance natal dispersal in leopard reveals potential for a three-country metapopulation. S Afr J Wildl Res 43:61–67. https://doi.org/10.3957/056.043.0108
Faubet P, Gaggiotti OE (2008) A new Bayesian method to identify the environmental factors that influence recent migration. Genetics 178:1491–1504. https://doi.org/10.1534/genetics.107.082560
Ferrar AA, Lötter MC (2007) Mpumalanga biodiversity conservation plan handbook. Mpumalanga Tourism and Parks Agency, Nelspruit
Feulner PGD, Bielfeldt W, Zachos FE, Bradvarovic J, Eckert I, Hartl GB (2004) Mitochondrial DNA and microsatellite analyses of the genetic status of the presumed subspecies Cervus elaphus montanus (Carpathian red deer). Heredity 93:299–306. https://doi.org/10.1038/sj.hdy.6800504
Goudet J (1995) FSTAT (Version 1.2): a computer program to calculate F-statistics. J Hered 86:485–486. https://doi.org/10.1093/oxfordjournals.jhered.a111627
Grey JNC, Bell S, Hill RA (2017) Leopard diets and landowner perceptions of human wildlife conflict in the Soutpansberg Mountains, South Africa. J Nat Conserv 37:56–65. https://doi.org/10.1016/j.jnc.2017.03.002
Guillot G, Mortier F, Estoup A (2005) GENELAND: a computer package for landscape genetics. Mol Ecol Notes 5:712–715. https://doi.org/10.1111/j.1471-8286.2005.01031.x
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98
Jacobson AP, Gerngross P, Lemeris JR et al (2016) Leopard (Panthera pardus) status, distribution, and the research efforts across its range. PeerJ 4:28. https://doi.org/10.7717/peerj.1974
Jombart T (2008) adegenet: a R package for the multivariate analysis of genetic markers. Bioinformatics 24:1403–1405. https://doi.org/10.1093/bioinformatics/btn129
Jombart T, Devillard S, Balloux F (2010) Discriminant analysis of principal components: a new method for the analysis of genetically structured populations. BMC Genet. https://doi.org/10.1186/1471-2156-11-94
Keller LF, Waller DM (2002) Inbreeding effects in wild populations. Trends Ecol Evol 17:230–241. https://doi.org/10.1016/s0169-5347(02)02489-8
Kenney J, Allendorf FW, McDougal C, Smith JL (2014) How much gene flow is needed to avoid inbreeding depression in wild tiger populations? Proc R Soc B Biol Sci 281:20133337. https://doi.org/10.1098/rspb.2013.3337
Keyghobadi N (2007) The genetic implications of habitat fragmentation for animals. Can J Zool 85:1049–1064. https://doi.org/10.1139/z07-095
Kock MD, Burroughs REJ (2012) Chemical and physical restraint of wild animals: a training and field manual for African species, 2nd edn. IWVS, Greyton
Kopelman NM, Mayzel J, Jakobsson M, Rosenberg NA, Mayrose I (2015) Clumpak: a program for identifying clustering modes and packaging population structure inferences across K. Mol Ecol Resour 15:1179–1191. https://doi.org/10.1111/1755-0998.12387
Kotze A, Ehlers K, Cilliers DC, Grobler J (2008) The power of resolution of microsatellite markers and assignment tests to determine the geographic origin of cheetah (Acinonyx jubatus) in Southern Africa. Mamm Biol 73:457–462. https://doi.org/10.1016/j.mambio.2007.10.011
Kotze A, Smith RM, Moodley Y et al (2019) Lessons for conservation management: monitoring temporal changes in genetic diversity of Cape mountain zebra (Equus zebra zebra). PLoS ONE 14:14. https://doi.org/10.1371/journal.pone.0220331
Lagendijk DDG, Gusset M (2008) Human-carnivore coexistence on communal land bordering the greater Kruger Area, South Africa. Environ Manag 42:971–976. https://doi.org/10.1007/s00267-008-9204-5
Leigh JW, Bryant D (2015) POPART: full-feature software for haplotype network construction. Methods Ecol Evol 6:1110–1116. https://doi.org/10.1111/2041-210x.12410
Lenormand T (2002) Gene flow and the limits to natural selection. Trends Ecol Evol 17:183–189. https://doi.org/10.1016/s0169-5347(02)02497-7
Lopez JV, Cevario S, Obrien SJ (1996) Complete nucleotide sequences of the domestic cat (Felis catus) mitochondrial genome and a transposed mtDNA tandem repeat (Numt) in the nuclear genome. Genomics 33:229–246. https://doi.org/10.1006/geno.1996.0188
Lötter MC (2015) Technical report for the Mpumalanga biodiversity sector plan—MBSP. Mpumalanga Tourism and Parks Agency, Nelspruit
Maputla NW, Chimimba CT, Ferreira SM (2013) Calibrating a camera trap-based biased mark-recapture sampling design to survey the leopard population in the N’wanetsi concession, Kruger National Park, South Africa. Afr J Ecol 51:422–430. https://doi.org/10.1111/aje.12047
Marsden CD, Woodroffe R, Mills MGL et al (2012) Spatial and temporal patterns of neutral and adaptive genetic variation in the endangered African wild dog (Lycaon pictus). Mol Ecol 21:1379–1393. https://doi.org/10.1111/j.1365-294X.2012.05477.x
McManus JS, Dalton DL, Kotze A, Smuts B, Dickman A, Marshal JP, Keith M (2015) Gene flow and population structure of a solitary top carnivore in a human-dominated landscape. Ecol Evol 5:335–344. https://doi.org/10.1002/ece3.1322
Menotti-Raymond M, David VA, Lyons LA, Schaffer AA, Tomlin JF, Hutton MK, O’Brien SJ (1999) A genetic linkage map of microsatellites in the domestic cat (Felis catus). Genomics 57:9–23. https://doi.org/10.1006/geno.1999.5743
Miller B, Dugelby B, Foreman D, del Rio CM, Noss R, Phillips M (2001) The importance of large carnivores to healthy ecosystems. Endang Species Update 18:202
Miller SM, Harper CK, Bloomer P, Hofmeyr J, Funston PJ (2015) Fenced and fragmented: conservation value of managed metapopulations. PLoS ONE 10:e0144605. https://doi.org/10.1371/journal.pone.0144605
Mills LS, Allendorf FW (1996) The one-migrant-per-generation rule in conservation and management. Conserv Biol 10:1509–1518. https://doi.org/10.1046/j.1523-1739.1996.10061509.x
Mondol S, Bruford MW, Ramakrishnan U (2013) Demographic loss, genetic structure and the conservation implications for Indian tigers. Proc R Soc B-Biol Sci 280:10. https://doi.org/10.1098/rspb.2013.0496
Moodley Y, Russo I-RM, Dalton DL et al (2017) Extinctions, genetic erosion and conservation options for the black rhinoceros (Diceros bicornis). Sci Rep 7:41417. https://doi.org/10.1038/srep41417
Moodley Y, Russo IRM, Robovsky J et al (2018) Contrasting evolutionary history, anthropogenic declines and genetic contact in the northern and southern white rhinoceros (Ceratotherium simum). Proc R Soc B-Biol Sci. https://doi.org/10.1098/rspb.2018.1567
Morris DR, Boardman WS, Swanepoel LH, Simpson G, Coetzee J, Camacho GJ, McWhorter TJ (2021) Population density estimate of leopards (Panthera pardus) in north-western Mpumalanga, South Africa, determined using spatially explicit capture–recapture methods. Mamm Biol. https://doi.org/10.1007/s42991-021-00179-2
Naude VN, Balme GA, O’Riain J, Hunter LTB, Fattebert J, Dickerson T, Bishop JM (2020) Unsustainable anthropogenic mortality disrupts natal dispersal and promotes inbreeding in leopards. Ecol Evol 10:3605–3619. https://doi.org/10.1002/ece3.6089
Peakall R, Smouse PE (2012) GenAlEx 6.5: genetic analysis in Excel. Population genetic software for teaching and research-an update. Bioinformatics 28:2537–2539. https://doi.org/10.1093/bioinformatics/bts460
Porras-Hurtado L, Ruiz Y, Santos C, Phillips C, Carracedo A, Lareu MV (2013) An overview of STRUCTURE: applications, parameter settings, and supporting software. Front Genet 4:98–98. https://doi.org/10.3389/fgene.2013.00098
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959. https://doi.org/10.1093/genetics/155.2.945
Rambaut A, Drummond AJ, Xie D, Baele G, Suchard MA (2018) Posterior summarization in Bayesian phylogenetics using tracer 1.7. Syst Biol 67:901–904. https://doi.org/10.1093/sysbio/syy032
Ripple WJ, Estes JA, Beschta RL et al (2014) Status and ecological effects of the world’s largest carnivores. Science 343:151–162. https://doi.org/10.1126/science.1241484
Ropiquet A, Knight AT, Born C et al (2015) Implications of spatial genetic patterns for conserving African leopards. C R Biol 338:728–737. https://doi.org/10.1016/j.crvi.2015.06.019
Rozas J, Ferrer-Mata A, Sanchez-DelBarrio JC, Guirao-Rico S, Librado P, Ramos-Onsins SE, Sanchez-Gracia A (2017) DnaSP 6: DNA sequence polymorphism analysis of large data sets. Mol Biol Evol 34:3299–3302. https://doi.org/10.1093/molbev/msx248
Schlaepfer DR, Braschler B, Rusterholz HP, Baur B (2018) Genetic effects of anthropogenic habitat fragmentation on remnant animal and plant populations: a meta-analysis. Ecosphere. https://doi.org/10.1002/ecs2.2488
Simpson GB, Badenhorst J, Jewitt GPW, Berchner M, Davies E (2019) Competition for land: the water-energy-food nexus and coal mining in Mpumalanga Province. S Afr Front Environ Sci 7:86. https://doi.org/10.3389/fenvs.2019.00086
Spielman D, Brook BW, Briscoe DA, Frankham R (2004) Does inbreeding and loss of genetic diversity decrease disease resistance? Conserv Genet 5:439–448. https://doi.org/10.1023/B:COGE.0000041030.76598.cd
Spong G, Johansson M, Bjorklund M (2000) High genetic variation in leopards indicates large and long-term stable effective population size. Mol Ecol 9:1773–1782. https://doi.org/10.1046/j.1365-294x.2000.01067.x
Stover BC, Muller KF (2010) TreeGraph 2: combining and visualizing evidence from different phylogenetic analyses. BMC Bioinform 11:7. https://doi.org/10.1186/1471-2105-11-7
Swanepoel LH, Lindsey P, Somers MJ, van Hoven W, Dalerum F (2013) Extent and fragmentation of suitable leopard habitat in South Africa. Anim Conserv 16:41–50. https://doi.org/10.1111/j.1469-1795.2012.00566.x
Swanepoel LH, Lindsey P, Somers MJ, Van Hoven W, Dalerum F (2014) The relative importance of trophy harvest and retaliatory killing of large carnivores: South African leopards as a case study. S Afr J Wildl Res 44:115–134. https://doi.org/10.3957/056.044.0210
Swanepoel LH, Somers MJ, van Hoven W et al (2015) Survival rates and causes of mortality of leopards Panthera pardus in southern Africa. Oryx 49:595–603. https://doi.org/10.1017/s0030605313001282
Swanepoel LH, Balme G, Williams S et al (2016) A conservation assessment of Panthera pardus. In: Child MFRL, Do Linh San E, Raimondo D, Davies-Mostert HT (eds) The red list of mammals of South Africa, Swaziland and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, Midrand, pp 1–13
Teske PR, Golla TR, Sandoval-Castillo J et al (2018) Mitochondrial DNA is unsuitable to test for isolation by distance. Sci Rep 8:8448. https://doi.org/10.1038/s41598-018-25138-9
Thorn M, Green M, Dalerum F, Bateman PW, Scott DM (2012) What drives human-carnivore conflict in the North West Province of South Africa? Biol Conserv 150:23–32. https://doi.org/10.1016/j.biocon.2012.02.017
Uphyrkina O, Johnson WE, Quigley H, Miquelle D, Marker L, Bush M, O’Brien SJ (2001) Phylogenetics, genome diversity and origin of modern leopard, Panthera pardus. Mol Ecol 10:2617–2633. https://doi.org/10.1046/j.0962-1083.2001.01350.x
Van Oosterhout C, Hutchinson WF, Wills DPM, Shipley P (2004) MICRO-CHECKER: software for identifying and correcting genotyping errors in microsatellite data. Mol Ecol Notes 4:535–538. https://doi.org/10.1111/j.1471-8286.2004.00684.x
Wang JL (2004) Application of the one-migrant-per-generation rule to conservation and management. Conserv Biol 18:332–343. https://doi.org/10.1111/j.1523-1739.2004.00440.x
Wolf C, Ripple WJ (2018) Rewilding the world’s large carnivores. R Soc Open Sci 5:172235. https://doi.org/10.1098/rsos.172235
Wright S (1984) Evolution and the genetics of populations, vol 3. Univeristy of Chicago Press, Chicago
Zanin M, Adrados B, González N et al (2016) Gene flow and genetic structure of the puma and jaguar in Mexico. Eur J Wildl Res 62:461–469. https://doi.org/10.1007/s10344-016-1019-8
Acknowledgements
The authors would like to thank the Mpumalanga Tourism and Parks Agency for their support, especially Gerrie Camacho, Delecia Gunn, Ernest Rohm, Gerhardus Sterk and Chris Hobkirk their expertise input, assistance and time sacrificed for leopard captures. They would also like to thank Jannie Du Bruyn, Dr. Hannes Botha, Tommie Steyn, Juan De Beer & Johan Eksteen for their technical support and advice during the project. Thanks to Dr. Philip Stott for his input into the project development. Thanks also to the students Herman Viviers, Tora-Lee Axelson, Max Peters, Parris Jeffries, Marco Gouws and Olivia Rynders who assisted with field work activities. This study was funded from PhD student operating funds available from the School of Animal and Veterinary Science at The University of Adelaide.
Funding
This study was funded from PhD student operating funds available from the School of Animal and Veterinary Science at The University of Adelaide.
Author information
Authors and Affiliations
Contributions
All authors had input to the overall project research design. Field work research and sample collection was performed by DRM, JC, GS, FP & JW. JW contributed genetic samples for analysis. Data analysis was performed by DRM & YM. The paper was written by DRM with large input from YM, TJM & WSJB.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
Ethical approval was received from the University of Adelaide Animal Ethics Committee (S-2016-023) and the permit to conduct research on Loskop Dam Nature Reserve was given by the provincial municipality Mpumalanga Tourism and Parks Agency (TS3/11).
Consent for publication
All authors give their consent to the publication of this article.
Additional information
Communicated by Lilly O. Rodriguez.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Morris, D.R., McWhorter, T.J., Boardman, W.S.J. et al. Gene flow connects key leopard (Panthera pardus) populations despite habitat fragmentation and persecution. Biodivers Conserv 32, 945–963 (2023). https://doi.org/10.1007/s10531-022-02531-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-022-02531-5